INTRODUCTION
Malaria is one of the most serious diseases to affect peoples in developing countries with tropical and subtropical climates. Although large amounts of investment have been made in attempts to develop a vaccine against malaria, none have been developed so far. The major problem in vaccine development is the antigenic diversity of the vaccine candidates among parasites; thus, the variation of the immunodominant region is a serious obstacle (Berzins and Anders, 1999; Bold and Berzins, 2000). Therefore, the genetic variation study for the antigens of potential vaccine candidate, e.g. CSP (circumsporozoite protein), MSP (merozoite surface protein), and DBP (Duffy binding protein), is very important and continues to grow steadily in both Plasmodium faliciparum and P. vivax (Rich et al., 2000).
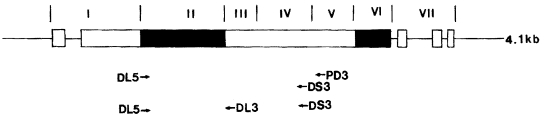
Erythrocytes of Duffy-negative individuals are resistant to invasion by P. vivax (Miller et al., 1976). The P. vivax DBP (PvDBP) has been identified as the parasite ligand for the Duffy-glycoprotein receptor in human erythrocytes (Barnwell et al., 1989). This fact could certainly compromise the potential of PvDBP as an asexual blood-stage malaria vaccine candidate (Adams et al., 1990). The PvDBP is divided into seven regions: a leader peptide sequence (region I), cysteine rich regions (region II and VI), hydrophobic regions (region III to V), and a transmembrane domain (region VII). Among them the region II, amino terminal cysteine-rich region, has been proven that it is implicated in the binding process to human erythrocytes (Barnwell et al., 1989; Ranjan and Chitnis, 1999). Therefore the genetic polymorphism in the region should be considerable factor in the rational design of vaccines against asexual blood stages of P. vivax. However, the only available information has been made from the Papua New Guinean (PNG) and Colombian isolates (Tsuboi et al., 1994; Ampudia et al., 1996). To add useful information for rational design of vaccine the polymorphism of PvDBP region II in the Korean isolates was investigated in this study. The polymorphism of region IV where the length polymorphism has been reported in the PNG isolates (Tsuboi et al., 1994) also observed in the Korean isolates.
In addition, we have attempted oligonucleotide hybridization, which is economical and rapid compared to DNA sequencing, to identify the genotype for P. vivax of Korean isolates.
MATERIALS AND METHODS
Isolation of parasite genomic DNA
Thirty blood samples were collected from P. vivax patients who were detected in Yonchon-gun, Kyonggi-do in 1998. All patients were diagnosed by microscopic examination at the Institute of Malariology, Inje University.
As described previously, parasite DNA was extracted from 0.1 ml of EDTA treated blood by proteinase K digestion and then followed by two rounds of phenol/chloroform extraction (Kho et al., 1999). The supernatant was mixed with 0.1 volume of 3 M sodium acetate (pH 5.2) and 3 volumes of ethanol, and incubated at -70℃ for 1 hour. The parasite DNA recovered were dissolved in 50 µl of sterilized water and used as template for PCR.
Semi-nested polymerase chain reaction (PCR)
Three different primers were synthesized for amplification of DBP gene (Fig. 1). The first set of primers was DL5 (5'-GGGAAAGAGATTGGGACTGT-3') and PD3 (5'-GCCCCGTTCTTTTCCGTGTC-3'). The second set of primers was DL5 and DS3 (5-AGTATCACCCGTAGCAGAGC-3). Amplification by the PCR of the fragment encoding regions II, III, and VI of the gene was carried out in 20 µl reaction mixture containing extracted P. vivax DNA, 200 µM dNTP each, 0.5 pM primer each, 10 mM Tris-HCl, pH 8.0, 50 mM KCl, 1.0 mM MgCl2, and 2.5 units of Taq polymerase (Perkin Elymer, Norwalk, CT, USA). The reaction was subjected to 35 cycles of denaturation at 94℃ for 1 min, annealing at 58℃ for 1 min, and extension at 72℃ for 1 min 45 sec, and one at 72℃ for 5 min in a thermal cycler (GeneAmp PCR system 9700, Perkin-Elmer). A sample of 1/104 µl of the first PCR product was transferred to a new tube, followed by the addition of the second set of primers, fresh Taq polymerase and fresh dNTPs. The semi-nested PCR reaction was performed as described above.
Sequencing and gene analysis
The 1.5 kb DNA fragments obtained by amplification of thirty blood samples were separated on a 0.8% agarose gel. The amplified DNA was purified from agarose gel using a QIAEX II gel extraction kit (Qiagen, Valencia, CA, USA) following the manufacturer's instruction. The nucleotide sequence was determined by dideoxynucleotide chain termination method using a sequenase kit (ABI PRISM Dye Terminator Cycle Sequencing Core Kit, Perkin Elmer) and an automated DNA sequencer (Applied Biosystems model 377A, Perkin Elmer). The DL5, DS3, and DL3 (5'-TCATTCTCAAAAGCCACCTC-3') primers were used for direct sequencing. In order to detect any possible nucleotide mis- incorporation due to false Taq polymerase activity, two of each amplified products were sequenced.
The DNA sequencing data were analyzed using DNASIS (Hitachi, Ver. 2.5, Japan) and the BLAST program of the NCBI databases (NIH, Bethesda, MD, USA). Our sequence data were also compared with published sequences of P. vivax isolates (Tsuboi et al., 1994; Ampudia et al., 1996).
Oligonucleotide hybridization
To distinguish the difference between SK-1 and SK-2 genotypes, two different oligonucleotide probes were used in this study. Two oligonucleotides, D4-AF1 (5'-TGATAGCG ATGGACC-3') and D4-BF3 (5'-GAATTTGC AGAATCTACGAAAT-3'), were designed and synthesized to detect 6-bp insert of the Sal-1 and 27-bp insert of the PNG isolate, respectively. The oligonucleotides were radiolabeled with 32P-dCTP by 5'-end labeling kit (Amersham-Pharmacia Biotech, Uppsala, Sweden) following the manufacturer's instruction. Unincorporated nucleotides were removed from the labeling mixture by a spin column (Sephadex G-25) chromatography.
One µg of PCR product for PvDBP gene was separated by 0.8% agarose gel electrophoresis and transferred to Hybond N+ membrane (Amersham-Pharmacia Biotech) by capillary action. These southern blots were hybridized with the radio-labelled oligonucleotides. The prehybridization and hybridization were carried out using Hybrid sol I (Oncer, Gaitherberg, MD, USA) in a hybridization incubator (Robbinson Scientific, Sunnyvale, CA, USA). After hybridization overnight at 42℃, the membrane was washed with 1 × SSC (10 × SSC; 1.5 M NaCl and 0.15 M sodium citrate) containing 0.2% SDS for 10 min at 50℃, then with 0.5 × SSC/0.2% SDS for 10 min at 55℃. The membrane was exposed to Kodak XAR-5 film overnight at -70℃ with intensifying screens.
RESULTS
PCR products of PvDBP gene
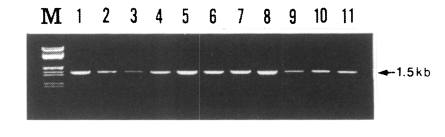
PCR amplification of the gene of PvDBP gene was attempted for 30 blood samples obtained from patients in the Republic of Korea. The primary PCR products showed a specific band in about 83% of samples (25/30 isolates), whereas the second round PCR showed a specific band pattern in all of the samples (30/30 isolates). The size of PCR product was 1.5 kb which includes the fragment encoding regions II (partial), III, and IV (partial) of the PvDBP gene (Fig. 1). There was no difference in size, but a slight difference in band intensity was observed (Fig. 2). There was not any specific product from un-infected donor (data not shown).
Variation of the region-II of PvDBP gene
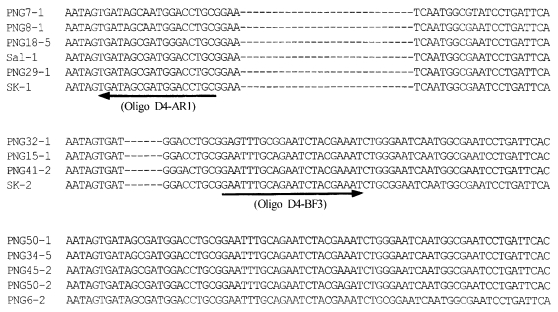
The DNA sequences of the 30 isolates were determined and deduced by amino acid sequences (Fig. 3). Two genotypes, SK-1 (Genbank accession number AF215738) and SK-2 (Genbank accession number AF215738), were identified on the basis of grouping mutations in the nucleotides and their corresponding amino acids. Of 30 isolates, 10 isolates showed SK-1 genotype and 20 isolates SK-2 genotype. Most of the predicted amino acid sequence in the region II of DBP were conserved between the Korean isolates and the Belem strain. The amino acid substitutions occurred in the middle of the amino cysteinerich region (region II-2). Compared with the region II-2 of the Belem strain, 4 variations in SK-1 and 3 variations in SK-2 among 107 amino acids were found. Two of these variations in positions 54 (Lys/Glu) and 55 (Asn/Lys) were shared in SK-1 and SK-2. Variations at 40 (Glu/Lys) and 53 (Gly/Asp) were unique in SK-1 and variation at 59 (Gln/His) was found only in SK-2.
Oligonucleotide hybridization
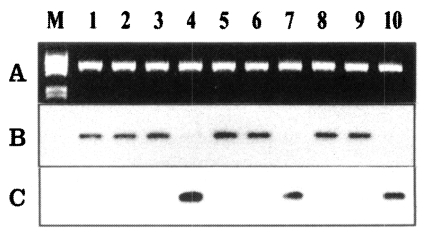
In the region IV of DBP, a 6-bp insert that shows the PNG allele type was found in SK-1, and a 27-bp insert that shows the Sal-1 allele type was found in SK-2 (Fig. 4). The oligonucleotide probes (D4-AF1 and D4-BF3) were designed to identify the sequences of the two different types, SK-1 and SK-2. The D4-AF1 oligonucleotide probe was specific to SK-1 type, which characterized the 6-bp insert of the Sal-1 type, and hybridized to 10 of the 30 isolates (Fig. 5B). The D4-BF3 oligonucleotide probe specific to SK-2 type hybridized to 20 of the 30 isolates (Fig. 5C).
DISCUSSION
The central binding residues of PvDBP lay in 170 amino acids stretch between cysteines 4 and 7 of region II, between region II-1 and II-3, and participate in the binding process to human erythrocyte (Ranjan and Chitnis, 1999). A previous study showed that the amino acid changes mainly observed in the middle of the cysteine-rich region (region II-2 Tsuboi et al, 1994). However, it is also demonstrated that three variants of the Duffy binding domain bind to RBCs in an identical fashion (Chitnis et al., 1994). It seems more likely that the changes are a result of immune pressure or selection. Therefore, the conserved residues of this region are less likely to be subjected to immune pressure and possibly involved as a core part in erythrocyte binding.
In this study, part of region II including the central binding domain was studied in the Korean isolates. Most of the predicted amino acids in the region II of PvDBP were conserved between the Korean isolates and the Belem strain. Of the total 225 amino acids (part of region II), variations at 5 positions were found, corresponding to a polymorphism of 2.2% compared with the Belem strain (Fig. 3). These 5 variations of the Korean isolates were also found in a previous study in PNG or Colombia (Tsuboi et al., 1994; Ampudia et al., 1996). It is proved that the variations in these positions do not affect erythrocyte specificity (Barnwell et al., 1989; Chitnis et al., 1994). Therefore, the variations of the PvDBP region II found in SK-1 and SK-2 genotypes are considered not to affect the binding affinity of PvDBP to RBCs.
Tsuboi et al. (1994) classified the DNA sequence of region IV into three groups based on the inserted nucleotides: the first group has a 6-bp insert and no 27-bp insert; the second group has a 27-bp insert and no 6-bp insert; the third group has both 6-bp and 27-bp inserts. According to the Tsubois' criteria, SK-1 and SK-2 genotypes in region IV were identical to the first and second group, respectively. Results from this study support the postulation that the variations of PvDBP region IV are formed by the tandem repeats (Tsuboi et al 1994). The tandem-repeat polymorphism was identified in the merozoite microneme protein-1 (MP-1) genes of P. knowlesi (Prickett et al., 1994; Chitnis et al., 1996) and P. falciparum (Ware et al., 1993). This result suggests that the Plasmodium MP-1 family has a common mechanism for genetic exchange among different genotypes in a given species. To distinguish SK-1 and SK-2 genotypes, two different oligonucleotide probes were designed on the basis of PvDBP region IV nucleotide sequence. As shown in Fig. 5, the two genotypes of SK-1 and SK-2 can be easily distinguished by the hybridigatin using these probes. The DNA sequencing data of the 30 specimens coincided very well with the hybridization results (data not shown). Therefore, the hybridization with these probes can be useful in distinguishing the two genotypes of P. vivax found in Korea instead of using the gene sequencing technique.
In addition to vaccine development, the information of the genetic variation of an antigen can be utilized in various fields including genetics, and epidemiology. The genetic variation of some genes for Plasmodium sp. is geographically related. Therefore, the information of genetic variation may be used in geophylogenetic studies and tracking the origin of Plasmodium. For this purpose, we have reported the genotypes (SK-A and SK-B) of P. vivax circumsporozoite (PvCSP) of the Korean isolates (Kho et al., 1999). The analysis of P. vivax genotype may demonstrate geographical linkage, which will help to find the origin of parasites. However, the geographical characteristic linkage of the PvDBP gene is unknown because it is purely understood at the molecular level. To date, the genetic study of PvDBP has only been done in PNG, Columbia, and Korea (Tsuboi et al., 1994; Ampudia et al., 1996). The PvDBP regions II and IV DNA sequences of the Korean isolates were found to have no identical sequence when compared with known data. To understand the PvDBP correlation according to the geographical distribution, further investigations of the isolates of Southeast Asia, China, and neighboring countries are required.
Concerning the extent of genetic polymorphism of PvDBP according to the geographical region, two genotypes were found from 30 patients in Korea while 12 genotypes from 50 patients in PNG and 17 genotypes from 20 patients in Colombia were found (Tsuboi et al., 1994; Ampudia et al., 1996). In this study, the number of positions where the change occurred was five from 255 amino acids while genetic variations were found at 37 positions from a total of 300 amino acids, corresponding to a polymorphism of 12% in PNG, and 19 positions from a total of 383 amino acids showed polymorphism (5%) in Colombia, respectively (Tsuboi et al., 1994; Ampudia et al., 1996). The difference both in the number of genotypes and positions changed in amino acid sequences between Korea and two other countries may be to the difference in the intensity of malaria transmission.
Recently, attempts are being made to evaluate the transmission intensity using genetic information; genetic variation in hyperendemic areas have been found to be larger than in hypo-endemic areas (Babiker and Walliker, 1997; Paul et al., 1998). Previous studies of the genetic variation within the amino terminal of the cysteine rich region in PvDBP show several patterns among isolates, also observed is a large genetic variation in the high transmission intensity areas (Tsuboi et al., 1994). Because the variations may occur mainly due to a continuous immune pressure or selection. The both of two countries, PNG and Columbia, are malaria endemic areas and have shown high transmission intensity for a long time.
Although the recent intensity of malaria transmission in the Republic of Korea has not been scientifically measured, it is believed that the opportunity of malaria being transmitted by mosquitoes might be strictly limited since the transmission is possible during the limited season from May to October and the transmission ability of vector mosquito may be extremely low (Sim et al., 1997; Ree, 2000). When considering the epidemic characteristics of malaria, the genetic change may occur more slowly in Korea than in the high-endemic areas. In the Republic of Korea, one case of indigenous vivax malaria, which had disappeared since 1984, had been reported in 1993 and the number of cases has increased exponentially year after year. Currently about four thousand cases are reported annually (Chai et al., 1994; NIH, 2000). The result of this study may also be utilized as baseline data for monitoring the genetic change in an endemic area, because new malaria endemic recently begins in Korea. Therefore, it is thought that the results of this study in Korea may assist in elucidating the mechanism of polymorphisms due to immune pressure or selection and the mechanism of its spread in the gene.
In this study, we show that the PvDBP of Korean isolates has little polymorphism and that two genotypes of P. vivax coexist in the endemic areas of the Republic of Korea. The result shown in this study is significant in that it is the first report for PvDBP in the Far East Asia.