INTRODUCTION
Leishmaniasis is endemic in 88 countries throughout Africa, Asia, Europe, and North and South Americas. There are an estimated 12 million cases worldwide, with 1.5 to 2 million new cases each year (WHO, 1984, 1990). Leishmania and Donovan, working separately, first described the Leishmania protozoan in 1903 (Herwaldt, 1999). Since then, this organism has been found to be a complex group of species, at least 20 of which cause infections in humans (Markele and Khaldoun, 2004). Some species cause visceral leishmaniasis, some cause cutaneous leishmaniasis, and some cause both. Cutaneous leishmaniasis (CL) occurs in the new world and in the old world. In the old world, the disease is primarily caused by Leishmania tropica in urban areas (dry type) and Leishmania major (wet type) in dry desert areas (Markele and Khaldoun, 2004).
The parasite enters the human host with the bite of the sandfly and is pulled into macrophages by ingestion. Leishmania parasites are able to survive the acidic environment of the lysosome and become amastigote forms. These forms are obligate, intracellular, nonmotile, and about 2-7 µm in diameter. This amastigote form causes disease in humans and affects cellular immunity. CL begins as an erythematous papule at the site of sandfly bite on exposed parts of the body. Eventually, a sandfly will pick up this form while feeding, and it will develop into the promastigote form again in the insect (Kafetzis, 2003; Markele and Khaldoun, 2004).
Cutaneous leishmaniasis (CL) is still considered an important health problem in many parts of the world, especially the Mediterranean region, some countries of Africa, and almost all countries of the Middle East, including Iran (Momeni, 1994; Ozbel et al., 1995;Khoury et al., 1996; Alimohammadian et al., 1999). The prevalence of infection is high in some provinces of Iran, including Isfahan (Nadim and Faghih, 1968;Salimi, 2000), Shiraz (Moaddeb et al., 1993), Khorasan (Javadian et al., 1967), and Khozestan and Kerman (Nadim and Seyedi, 1971). Isfahan is a well known endemic area of zoonotic cutaneous leishmaniasis (ZCL). In north east of Isfahan, the disease incidence is high, especially in rural areas (Nadim and Faghih, 1968; Salimi, 2000). In spite of some reports about increased rate of CL in north area of Isfahan Province, however, there have been no previous reports of endemicity of ZCL around Kashan, 230 km north of Isfahan.
Although this disease does not cause mortality, but because of cosmetic disfiguration, a prolonged period of lesions, great expense of treatment, and side effects of available drugs, it has created many problems (Edrissian et al., 1982; Momeni, 1994). During last years, valuable studies have been performed in Iran. According to those studies, the outbreaks of CL in Isfahan were 2.5% (Nadim and Faghih, 1968; Salimi, 2000). This study was performed in order to determine the abundance and characteristics of cutaneous leishmaniasis in the childhood patients. We aimed here to describe the epidemio-clinical profiles, therapeutic characteristics, and outcome data of CL in 3 areas of Kashan, Iran.
MATERIALS AND METHODS
This descriptive study was carried out on all patients clinically suspected of having CL who were referred to the central laboratory in Kashan, for parasitologic diagnosis during the period from 2001 to 2003. Kashan is an ancient city; the area has a desert climate, very hot in summer and cold in winter. The city is located 230 km north of Isfahan and 230 km south of Tehran.
The diagnosis of CL was based on clinical presentations and a positive parasitic smear. For each case having cutaneous lesions (ulcers or scars), a questionnaire was completed to record the necessary information such as name, age, sex, sites of ulcer on the body, address, data and place of acquiring the disease, previous travel history, or location of workplace.
A total of 1,625 patients with skin lesions, suspected of CL, from villages in 3 areas, i.e., Kashan, Abouzeidabad, and Aran-Bidgol, were selected for study. Skin scrapings from the edge of the lesion were obtained from each patient. The remaining skin scraping portion was smeared on a slide for staining with Giemsa and examined microscopically for presence of amastigotes. The χ2-test was used to determine statistically significant differences in the disease patterns and the prevalence between boys and girls.
RESULTS
During this 3-year period, information on 1,625 children showed that the prevalence of ulcerative localized CL was 7.2% (117 cases), and cases with scar and ulcer were 4.2% (68 cases), 3.0% (49 cases) were with active ulcers. Parasites were isolated in smears from the skin. Infections with Leishmania parasites were recorded in 53 girls (45.3%) and 64 boys (54.7%). The highest rates in both genders were recorded in the age group of 6-8 years (45.3%). The lowest rates (20.5%) were recorded in the age group of 12-15 years.
The overall scar rate was 4.2% and the prevalence of ulcers was 3%. This means a medium endemicity of the disease, which has recently become endemic, otherwise a higher scar rate should have been observed. Single lesions were seen in 50.9% of patients, appearing as a round papular plaque with a diameter of 4-80 mm. Double lesions were observed in 24.6% of patients and 29.4% of patients showed multiple (3-15) lesions. Lesions were mainly located on the face and neck (47.0%), compared with hands and arms (23.0%), and legs (16.3%). Multiple lesions appeared on 2 or more parts of the body in 17% of the cases. Detailed distribution of lesion and scar according to the number of lesions and scars were shown in Table 1.
The face and neck was the most commonly affected site (47.0%). On the face and neck, the cheek was the site of highest involvement (45.5%) and the chin was the site of lowest involvement (5.5%) (Table 2). The duration of the disease at the time of presentation ranged from 20 days to 5 years with an average duration of 8-12 mo.
All slides, which have been prepared by scraping the edges of ulcers of all patients contained parasites morphologically compatible with L. major (a large vacuole in the cytoplasm). The affected children had an average age of 9.75 years. The age range of the patients was 6-15 years. The sex ratio (boy 64: girl 53) was 1.2. The age groups and sex distribution of CL are shown in Table 3.
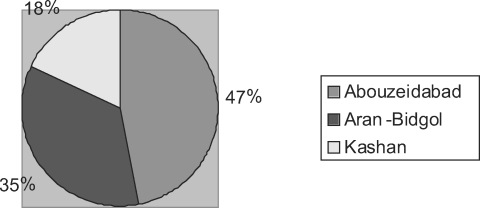
Residence, workplace, travel history of patients and the time of appearance of the lesions were important data for determining the locations where the infections might have taken place. The distribution of positive cases in the villages of Abouzeidabad, Kashan, and Aran-Bidgol is showed in Fig. 1. The highest infection rate (47.0%) was recorded in patients who are living or traveling in the villages at the area of Abouzeidabad. The lowest rate (18.0%) was recorded in patients living in the villages at the area of Kashan
We encountered 5 clinical forms of CL; papulonodular (27.4%), ulcer (60.7%), sporotrichoid (6.0%), impetiginous (2.5%), and erysipeloid (3.4%). Treatment and follow-up were well documented in 93 (79.5%) of 117 patients. Twenty-two patients were lost to follow-up and 2 child healed spontaneously. Two patients (1.7%) underwent treatment with intralesional meglumine antimoniate therapy weekly until recovery. Meglumine antimoniate was well tolerated without any side effects.
In follow-up of 93 patients, all CL lesion healed within an average period of 2 to 14 mo. Hyperpigmented scars were seen in 25.6%, atrophic scars in 4.3%, and hypopigmented scars in 3.4% of the patients.
DISCUSSION
This study reports cases of human CL from 3 different regions in southeast of Kashan, Iran. The prevalence rate of 3.0% reported in this study, seems to be rather high, but could reflect the occurrence of an almost severe outbreak of CL in this area. In clinical aspects, 3% of people under survey showed single or multiple ulcers, and 4.2% of them showed scars on their face-neck, hand, arm, legs, and other parts of the body. The overall rate of scars only was 1.4%, and the prevalence of ulcers was 3%. The frequency of lesions was almost twice as high as that of scars only, indicating the increasing progression of the outbreak during last years. The majority of ulcers or scars were seen on the face and neck, and upper parts of the body, which may be due to slipping out of the covered area on the terraces or house yards without using bednets during summer. The duration of active lesions was restricted to less than a year in most cases, while healing occurred in 79.5% of the patients within months. The endemic nature of the cases in these areas indicates the recent spread of the disease to these areas. Clustering of cases was recorded in endemic areas of Kashan and Aran-Bidgol; the highest number of cases was reported mostly in Abouzeidabad. The prevalence of CL was equal in the countryside of Kashan and Aran-Bidgol. Several previous studies reported on the endemicity of CL on both sides of Kashan and Aran-Bidgol (Yaghoobi et al., 2001, 2003, 2004).
The disease is endemic for many years in Abouzeidabad, and in the villages of Kashan area. The endemicity of the disease has been maintained due to the presence of the vector Phlebotomus papatasi and the reservoir host Rhombomys opimus in those areas (Alimohammadian et al., 1999). The predilection of the infection to boys (54.7%) than girls (45.6%) was also reported by other workers. This difference might be explained by more frequent exposure of boys to sandfly bites during their travel and work activities in the endemic areas (Kharfi et al., 2004). Significant differences were observed between infected males and females (P < 0.05). The χ2-test also showed non significant differences of host ages on scar/lesion rates.
The lesions were most frequently seen on the face and neck (47.0%), hands and arms (19.7%), legs (16.3%), and other parts of the body (17.0%). Customs and the geographic location of Kashan, presence of contaminated centers in Badrood and Ardestan, and traverse of susceptible hosts, especially the migration of Afgahans to these areas, are most important factors in expanding the disease (Javadian et al., 1967; Salimi, 2000). Phlebotomus longipes can not bite over the clothing, but bite the exposed areas of the body to suck the blood, so the lesions mainly appear in hands, face and legs (Nadim and Seyedi, 1971). Considering the fact that about 48.7% of patients had one or more skin lesions, we concluded that a sandfly, because of its physiologic characteristics, bites the host more than one time, and from every area of the bite, parasites enter the blood (Ahmadiyazdi et al., 2004; Yaghoobi et al., 2002; Ajdary et al., 2000).
The transmission cycle of Leishmania organisms requires an arthropod vector and a mammalian reservoir. The arthropod vectors are females of sandfly species. Adult female sandflies inject saliva containing Leishmania organisms into the wound. In the vector, the parasites are flagellated and extracellular; the organism is in its flagellated, promastigote form. Once inside the human, canine, or rodent host, the parasites are internalized by dermal macrophages. Here, they exit as intracellular, non-flagellated amastigotes. The cycle is completed when the sandfly feeds on an infected host, and the parasite moves to the gut where metamorphosis into the promastigote form takes place (Hanafi et al., 2002). As shown in our study, childhood CL mostly afffects school-age children having a history of stay in an endemic area. Even infants can be affected. Most of our patients were living in the southeast of Kashan, which may be associated with the geographical situation of Isfahan.
CL lesions can be misdiagnosed, especially in children, as impetigo, prurigo and folliculitis. Diagnosis is easily performed by the parasitology smear, but in endemic areas the diagnosis is established on clinical examinations (Berman, 1997). We used intramuscular meglumine antimoniate in cases of resistance to local treatments, multiplicity of the lesions (over 5), proximity to the cartilage and joint, and locations on the leg and foot. Our patients received the drug at the dose of 20-30 mg/kg/day with no side effects. Some authors reported success by using a dose of 10 mg/kg/day in children in order to prevent cardiac side effects (Selim et al., 1990; Talari et al., 1999; Talari and Sadr, 2005; Sadr and Talari, 1998).
In conclusion, childhood CL was frequent in the surveyed areas of Iran. The disease had the characteristics of endemic leishmaniasis. The standard therapy for multiple lesions by intramuscular injection with meglumine antimoniate was satisfactory, although further prospective studies are needed.