AbstractAlthough schistosomicidal drugs and other control measures exist, the advent of an efficacious vaccine remains the most potentially powerful means for controlling this disease. In this study, native fatty acid binding protein (FABP) from Fasciola gigantica was purified from the adult worm's crude extract by saturation with ammonium sulphate followed by separation on DEAE-Sephadex A-50 anion exchange chromatography and gel filtration using Sephacryl HR-100, respectively. CD1 mice were immunized with the purified, native F. gigantica FABP in Freund's adjuvant and challenged subcutaneously with 120 Schistosoma mansoni cercariae. Immunization of CD1 mice with F. gigantica FABP has induced heterologous protection against S. mansoni, evidenced by the significant reduction in mean worm burden (72.3%), liver and intestinal egg counts (81.3% and 80.8%, respectively), and hepatic granuloma counts (42%). Also, it elicited mixed IgG1/IgG2b immune responses with predominant IgG1 isotype, suggesting that native F. gigantica FABP is mediated by a mixed Th1/Th2 response. However, it failed to induce any significant differences in the oogram pattern or in the mean granuloma diameter. This indicated that native F. gigantica FABP could be a promising vaccine candidate against S. mansoni infection.
INTRODUCTIONSchistosomiasis is a chronic disease affecting human beings through 76 countries in several parts of the world and is arguably the most important human parasitic disease in terms of mortality. It is estimated that 207 million people are already infected by various species of schistosomes [1]. There are 779 millions exposed to the risk of infection [2] and the disease is responsible for 280,000 deaths per year in endemic areas [3].
Despite the existence of the highly effective antischistosomal drug praziquantel, drug treatment and other existing control measures are costly and have failed to eliminate the incidence of infection, morbidity, and mortality due to schistosomiasis infection [4]. This is in part due to the inability of chemotherapy to prevent new infection and failure of previously infected individuals to develop an effective immune response against the parasite [5]. Therefore, a long-term disease control strategy is needed combining mass chemotherapy with a protective vaccine. The development of an effective vaccine against schistosome is a critical stage, despite the fact that progress has been relatively slow, the successful use in animals of attenuated vaccines combined with recent recombinantly derived schistosome antigens, suggested that development of a safe and effective vaccine is feasible [6].
Vaccines in schistosomiasis using homologous antigens have been studied broadly in experimentally infected mammalian hosts than that of heterologous antigens. Numerous authors using a variety of experimental models have demonstrated crossed immunity. Native Fasciola hepatica fatty acid binding protein (FABP) (=nFh12) was shown to be a potential vaccine against both Schistosoma mansoni and F. hepatica [7]. C57/BL6 mice immunized with nFh12 had a significant reduction in Schistosoma bovis worm burden recoveries (96 and 87% reductions over control in 2 separate experiments) [8]. MAbs developed against Fasciola gigantica FABP all cross react with S. mansoni whole worms extract [9].
This work aimed to evaluate the efficacy of native F. gigantica FABP as a prophylactic agent against S. mansoni infection in CD1 mice.
MATERIALS AND METHODSAnimalsSix to 8-week-old female albino CD1 mice (24±2 g) were bred and maintained at the Schistosome Biological Supply Program, Theodor Bilharz Research Institute, Giza, Egypt (SBSP/TBRI). They were kept under standard laboratory care. Animal experiments have been carried out according to the internationally valid guidelines and ethical conditions [10].
ParasitesAdult F. gigantica worms were obtained from the bile ducts and gall bladders of naturally infected cattle livers. The flukes were washed 4 times with PBS (pH 7.4) at room temperature to remove all traces of blood and bile. Cercariae of an Egyptian strain of S. mansoni were obtained from SBSP/TBRI, and used for infection immediately after shedding from Biomphalaria alexandrina snails.
Purification of F. gigantica fatty acid binding protein (FABP)FABP was purified from the crude extracts by a combination of 3 different methods: ammonium sulphate precipitation, ion exchange chromatography on diethylaminoethyl (DEAE) sephadex A-50, and gel filtration using sephacryl HR-100, respectively, according to Timanova et al. [11].
Adult F. gigantica were homogenized at 4℃, in 2 volumes of 20 mM Tris-HCL buffer (BDH Chemicals, England) containing 5 mM phenylmethylsulfonyl fluoride (PMSF) as a protease inhibitor (Sigma Aldrich, St. Louis, Missouri, USA) at 20,000 rpm using IKA T20 homogenizer (IKA, Staufen, Germany). The homogenate was centrifuged at 30,000 rpm for 30 min at 4℃ and the supernatant was subjected to purification with 70% ammonium sulfate saturation. The post-saturation supernatant was then dialyzed overnight against 3 changes of 40 volumes of 10 mM Tris HCl buffer (pH 6.5), the dialysate was loaded on to a DEAE Sephadex A-50 column that had previously been equilibrated with the same buffer. The substance of interest (FABP) was washed out of the gel and not adsorbed under these conditions. The eluate was concentrated and subjected to sephacryl HR-100 gel filtration column equilibrated with 10 mM Tris HCl buffer (pH 8.2). The elution is carried out under gravity, 1 ml fractions were collected and absorbance was measured at 280 nm. The fractions were then analyzed by SDS-PAGE.
SDS-PAGETo analyze samples at different purification steps, SDS-PAGE 0.75-mm thick 12% vertical slab gels was performed under reducing conditions as described by Laemmli [12]. Samples were mixed with an equal volume of sample buffer (0.125 M Tris Hcl, 4% [w/v] SDS, 20% [v/v] glycerol, 10% [v/v] mercaptoethanol, 0.1% [w/v] bromophenol blue as a tracking dye) and immediately boiled for 5 min. All reagents used were of electrophoresis grade (Bio-Rad Laboratories, Richmond, California, USA). Low Mw kit standard protein (Bio-Rad) was prepared in parallel. The gels were stained with coomassie brilliant blue 0.05%.
Immunization and challenge infectionA batch of 55 mice was divided into 4 groups. Group I: Normal healthy controls (10 mice). Group II: Infected control group (15 mice). Mice were subcutaneously (SC) injected with 120 S. mansoni cercariae. Group III: Immunized group (10 mice). Mice were intraperitoneally (IP) injected with 100 µg of FABP emulsified in complete Freund's adjuvant (CFA) (Pierce, Rockford, Illinois, USA). Booster doses (50 µg/ml in equal volume of incomplete Freund's adjuvant (IFA, Pierce) was administered at week 2 and 3 after the initial dose. Group IV: Immunized infected group (20 mice). Mice were IP injected with 100 µg of FABP emulsified in CFA. Booster doses (50 µg/ml in equal volume of IFA was administered at week 2 and 3 after the first dose. At 1 week after the last boost, all animals were challenged through the SC injection with 120 S. mansoni cercariae. At week 8 post-infection (PI), all mice groups were sacrificed and subjected to examinations on the following parameters:
Parasitological parametersWorm burdenHepatic and portomesenteric vessels were perfused [13] to recover worms for subsequent counting. Following perfusion, numbers and sex of the adult worms were determined. They were counted either by direct visualization or under a stereomicroscope. Protection was calculated comparing the number of worms recovered from each group of mice with their respective controls, using the formula below:
Tissue egg loadThe number of eggs per gram of tissues (liver and intestine) was studied according to the procedure achieved by Cheever [14].
Oogram patternThe percentages of immature, mature, and dead ova in the small intestines were computed from a total of 100 eggs per intestinal segment and classified according to the categories previously defined by Pellegrino et al. [15].
ELISATotal and subclasses of anti-FABP IgM and IgG (H+L chains), IgG1, IgG2a, IgG2b, and IgG3 were measured using indirect ELISA based on the method of Engvall and Perlman [16]. ELISA microtitre plates were coated with 100 µl/well of 3 µg/ml FABP in carbonate/bicarbonate buffer. Sera were diluted to 1:250. Alkaline phosphatase (AKP)-labeled anti-mouse IgM (µ chain specific) and IgG (H+L chains) specific conjugate solution (Promega, Madison, Wisconsin, USA), were used at dilution 1:5,000 and 1:2,500, respectively. For the isotype determination experiment, isotype specific AKP-labeled anti-mouse mAb to mouse IgG1 (1:1,000), IgG2a (1:1,000), AKP-labeled rat mAb to mouse, IgG2b (1:3,000) and IgG3 (1:1,000) conjugates were used. The reaction was measured at optical density (OD) values 405 nm using an ELISA reader (Bio-Rad Microplate Reader).
Histopathological proceduresLivers of mice were fixed in 10% buffered formalin, processed into paraffin blocks, serially cut at 4 µm thickness and stained with hematoxylin and eosin. Hepatic granuloma measurements were done according to von Lichtenberg [17] using an ocular micrometer for those containing a central ovum only. Counting was carried out in 5 successive microscopic fields (10×10) in serial tissue sections of more than 250 µm apart.
Statistical analysisData are presented as mean±SD. The means of different groups were compared globally using 1 way analysis of variance (ANOVA), the mean difference between the 2 groups was calculated using the Student's t-test. Data were considered significant if P-value was <0.05. SPSS computer program (version 12 windows) was used for data analysis.
RESULTSFABP purificationThe profile of F. gigantica adult worm crude extract (lane 2) and its purified FABP (lanes 3 and 5) was shown in Fig. 1. The washed fractions obtained after purification by DEAE sephadex A-50 ion exchange column chromatography was assayed by 12% SDS. FABP was represented by a single band at 14.5 kDa with traces of other proteins at 34, 50, and 80 kDa (Fig. 1, lane 3). The protein content of these fractions was 2.5 mg/ml, and the other fractions were found to be a 17 kDa protein (Fig. 1, lane 4).
The fractions containing FABP were further purified through a Sephacryl HR-100 gel filtration column. The eluted FABP in the collected fraction was assayed by 12% SDS-PAGE, and the purified FABP was represented by a single band at 14.5 kDa (Fig. 1, lane 4). The protein content of FABP by these methods was 1.5 mg/ml as a purification result from starting protein content of crude extracts of F. gigantica adult worms (20 mg/ml).
Parasitological parametersImmunization of mice with F. gigantica FABP antigen induced a high significant reduction in the mean total number of worm burden on comparing the immunized infected group to the infected control group (P<0.001), with percent reduction (PR) 72.3% (Table 1). Moreover, A highly significant reduction in the mean total number of hepatic and intestinal egg loads was observed on comparing the immunized infected group to the infected control group (P<0.001), with PR of 81.3% and 80.8%, respectively (Table 1). The FABP antigen administration had insignificant effects on the oogram pattern (mature, immature, and dead eggs).
Immunological parametersWe investigated which type of immune response is induced by multiple immunizations with FABP in Freund's adjuvant. The detection of IgG1 as a Th2 marker and IgG2a/2b as a Th1 marker was performed by ELISA. Multiple immunizations with FABP in Freund's adjuvant induced a highly significant increase in total IgG, IgG1, and IgG2b antibodies in immunized groups and infected immunized group (P<0.001), with IgG1 isotype predominating, while no significant increase was seen in IgG2a and IgG3 antibodies. There was also a significant increase in IgM levels in immunized and infected immunized groups (Table 2).
Histopathological parametersThe mean number of hepatic granuloma of infected control group recorded 16.2±3.7 which was reduced in the immunized infected group after multiple administration of FABP antigen to be 10.0±3.5 with PR=42%, showing a highly significant reduction (P<0.001) compared to infected control group. In contrast, the mean hepatic granuloma diameter (MGD) in infected control mice group recorded 180.0±5.6. Repeated administration of FABP dose induced no significant reduction in the MGD in the infected mice (165.0±6.5) (Fig. 2).
DISCUSSIONSchistosomiasis is the most important human helminth infection in terms of morbidity and mortality. Despite 2 decades of wide spread chemotherapy with safe and effective drugs, the number of individuals with active schistosomiasis worldwide remains at about 200 million. Generally, severe symptoms of the disease appear only in people who harbor large numbers of parasites, so if a vaccine is effective in reducing a patient's burden of worms by 50% or more, this will be reflected in a reduction in the numbers of eggs retained in the tissues. Thus it can dramatically reduce the number of severe cases of the disease and morbidity [18]. Consequently, vaccine strategies represent an essential component for the future control of schistosomiasis as an adjunct to chemotherapy. Since morbidity, rather than sterile immunity is the target, only a partial protective vaccine is required.
The development of an effective vaccine against schistosomes is a critical stage, despite the fact that progress have been relatively slow, the successful use in animals of attenuated vaccines combined with recent recombinant derived Schistosoma antigens and partial immunity developed against schistosomiasis in individuals living in endemic areas suggest that the development of a safe and effective vaccine is possible [4,19]. Vaccines in schistosomiasis using homologous antigens have been studied extensively in experimentally infected mammalian hosts; vaccines using heterologous antigens have received comparatively less attention [20]. Cross immunity has been demonstrated among Schistosoma species and different parasitic trematodes [21-25]. One of the most studied cross reactive antigens is the native Fh12 and recombinant rFh15 F. hepatica FABP [20]. Sirisriro et al. [9] developed mAb against rFABP of F. gigantica. These mAb reacted with at least two isoforms of native proteins at Mw 14.5 kDa that were present in the tegumental antigen, crude worm extracts, and the excretory-secretory (ES) product and cross-reacted with a 14.5 kDa antigen present in the whole body extract of S. mansoni. Whether this can be translated into cross reactive immunoprophylaxis against both S. mansoni and F. gigantica is to be determined. So, our idea was to evaluate the efficacy of the native F. gigantica FABP as a prophylactic agent against S. mansoni infection in CD1 mice.
The result of the present study proved that native heterologous F. gigantica FABP significantly protects CD1 mice against challenge infection with S. mansoni. Multiple immunizations with native heterologous F. gigantica FABP via IP route in Freund's adjuvant had a strong immunoprophylactic effect against challenge infection with S. mansoni in CD1 mice. The protective effects were evidenced by reductions in the mean worm burden, ova count, and granuloma count in immunized infected group as compared to infected control group. Multiple immunizations of mice with FABP recorded a high significant reduction in worm burden (72.3%), which is the gold standard for anti-schistosomal vaccine development as it also had an adverse effect on the liver and intestinal egg count which results in lower number of eggs recovered from liver and intestinal tissues. Administration of FABP resulted in a very high and significant PR in liver and intestinal egg count (81.3% and 80.8%, respectively). This reduction reduces the number of eggs trapped in the hepatic tissues and hence decreases the number of hepatic granuloma by 42%. In contrast, it failed to induce any significant differences in the oogram pattern or in the mean granuloma diameter. These results were in agreement with those obtained by Abáne et al. [8] who reported that native F. hepatica FABP (Fh12) could elicit protective immunity in C57/BL6 mice. In 2 separate experiments, reductions in worm burden (96 and 87%) were found when Fh12 was inoculated with Freund's adjuvant; however, no protection was obtained in NRMI and BALB/c mice. The recombinant form of the antigen (rFh15) administrated in Freund's also induced significant levels of protection against heterologous challenge with S. bovis in C57/BL6 mice, 72% less worms than controls, and milder liver lesions was observed [8]. Furthermore, Hamed [26] reported that F. hepatica ES products in Freund's adjuvant significantly protect mice against S. mansoni infection evidenced by the significant reduction in worm burden, ova count, granuloma size, and numbers. FABP is considered as one of the major components of the helminth ES products.
In the humoral response, the change of immunoglobulin isotypes is regulated by the cytokines that segregate the T helper cells. IL-4 induces the change from IgM to IgG1 and IgE in B lymphocytes, a Th2 antibody response. On the other hand, IFN-γ required for Th1 T cell-dependent IgG2a/2b antibodies responses [27,28].
The highly significant increase in IgM and IgG antibodies, and more specifically IgG1 and IgG2b isotypes could indicate that the immunoprophylactic effect of the native F. gigantica FABP induced an immune response mediated by both cell-mediated and humoral (Th1/Th2) mechanisms which impede or interrupt the development of the infection with S. mansoni in some way in this strain of mice.
The protection obtained with 14.5 kDa native heterologous F. gigantica FABP was higher than that obtained with other homologous candidate antigens for anti-Schistosoma vaccines. The percentage protection using native F. gigantica FABP was greater (70%) in terms of the reduction of worm burden which were never achieved using homologous antigens of S. mansoni, IrV5 (50%-70%) [29], Sm28GST (40%-50%) [30], Sm14 (65%) [31], paramyosin (26-33%) [32], Sm29 (51%) [33], and Sm-p80 (39%-57%) [34]. The reduction in worm burden leads to a high reduction in liver and intestinal egg count which exceed 80%, this ratio was greater than the anti-fecundity effects of Sm28- and Sh28-GST (50% reduction in tissue egg load) and consequently there was a less number of liver granuloma (42% reduction in granuloma number). Further studies should be directed toward the use of recombinant form of F. gigantica FABP in experimental infection of S. mansoni and Schistosoma haematobium.
REFERENCES1. King CH. Toward the elimination of schistosomiasis. N Engl J Med 2009;360:106-109. PMID: 19129524.
2. Steinmann P, Keiser J, Bos R, Tanner M, Utzinger J. Schistosomiasis and water resources development: systematic review, meta-analysis, and estimates of people at risk. Lancet Infect Dis 2006;6:411-425. PMID: 16790382.
3. van der Werf MJ, de Vlas SJ, Brooker S, Looman CW, Nagelkerke NJ, Habbema JD, Engels D. Quantification of clinical morbidity associated with schistosome infection in sub-Saharan Africa. Acta Trop 2003;86:125-139. PMID: 12745133.
4. McManus DP, Loukas A. Current status of vaccines for schistosomiasis. Clin Microbiol Rev 2008;21:225-242. PMID: 18202444.
5. Capron A, Riveau G, Capron M, Trottein F. Schistosomes: the road from host-parasite interactions to vaccines in clinical trials. Trends Parasitol 2005;21:143-149. PMID: 15734662.
6. Wilson RA, Coulson PS. Schistosome vaccines: a critical appraisal. Mem Inst Oswaldo Cruz 2006;101:13-20. PMID: 17308743.
7. Hillyer GV. Fasciola antigens as vaccines against fascioliasis and schistosomiasis. J Helminthol 2005;79:241-247. PMID: 16153318.
8. Abán JL, Ramajo V, Arellano JL, Oleaga A, Hillyer GV, Muro A. A fatty acid binding protein from Fasciola hepatica induced protection in C57/BL mice from challenge infection with Schistosoma bovis. Vet Parasitol 1999;83:107-121. PMID: 10392967.
9. Sirisriro A, Grams R, Vichasri-Grams S, Ardseungneon P, Pankao V, Meepool A, Chaithirayanon K, Viyanant V, Tan-Ariya P, Upatham ES, Sobhon P. Production and characterization of a monoclonal antibody against recombinant fatty acid binding protein of Fasciola gigantica. Vet Parasitol 2002;105:119-129. PMID: 11900926.
10. Nessim NG, Hassan SI, William S, el-Baz H. Effect of the broad spectrum anthelmintic drug flubendazole upon Schistosoma mansoni experimentally infected mice. Arzneimittelforschung 2000;50:1129-1133. PMID: 11190780.
11. Timanova A, Müller S, Marti T, Bankov I, Walter RD. Ascaridia galli fatty acid-binding protein, a member of the nematode polyprotein allergens family. Eur J Biochem 1999;261:569-576. PMID: 10215871.
12. Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970;227:680-685. PMID: 5432063.
13. Duvall RH, DeWitt WB. An improved perfusion technique for recovering adult schistosomes from laboratory animals. Am J Trop Med Hyg 1967;16:483-486. PMID: 4952149.
14. Cheever AW. Conditions affecting the accuracy of potassium hydroxide digestion techniques for counting Schistosoma mansoni eggs in tissues. Bull World Health Organ 1968;39:328-331. PMID: 4881073.
15. Pellegrino J, Oliveira CA, Faria J, Cunha AS. New approach to the screening of drugs in experimental Schistosomiasis mansoni in mice. Am J Trop Med Hyg 1962;11:201-215. PMID: 14484966.
16. Engvall E, Perlmann P. Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry 1971;8:871-874. PMID: 5135623.
17. von Lichtenberg . Host response to eggs of S. mansoni. I. Granuloma formation in the unsensitized laboratory mouse. Am J Pathol 1962;41:711-731. PMID: 13930476.
18. Cherfas J. New hope for vaccine against schistosomiasis. Science 1991;251:630-631. PMID: 1992516.
19. McManus DP. A vaccine against Asian schistosomiasis: the story unfolds. Int J Parasitol 2000;30:265-271. PMID: 10719119.
20. Hillyer GV. Comparison of purified 12 kDa and recombinant 15 kDa Fasciola hepatica antigens related to a Schistosoma mansoni fatty acid binding protein. Mem Inst Oswaldo Cruz 1995;90:249-253. PMID: 8531666.
21. Janecharut T, Kitikoon V, Usawattanakul W, Sornmani S. Investigation on immunity induced by Schistosoma spindale against S. mekongi in experimental mice. Southeast Asian J Trop Med Public Health 1988;19:123-129. PMID: 3406801.
22. Agnew AM, Murare HM, Doenhoff MJ. Specific cross-protection between Schistosoma bovis and S. haematobium induced by highly irradiated infections in mice. Parasite Immunol 1989;11:341-349. PMID: 2506508.
23. Dean DA, Mangold BL, Harrison RA, Ricciardone MD. Homologous and heterologous protective immunity to Egyptian strains of Schistosoma mansoni and S. haematobium induced by ultraviolet-irradiated cercariae. Parasite Immunol 1996;18:403-410. PMID: 9229394.
24. Rabia I, Ali E, Hassan E, Salah F, El-Dafrawy T. Intraperitoneal injection of Echinostome antigens confers resistance to schistosomal infection in murine model. New Egypt J Med 2007;36:25-31.
25. Siles-Lucas M, Uribe N, López-Abán J, Vicente B, Orfao A, Nogal-Ruiz JJ, Feliciano AS, Muro A. The Schistosoma bovis Sb14-3-3zeta recombinant protein cross-protects against Schistosoma mansoni in BALB/c mice. Vaccine 2007;25:7217-7223. PMID: 17707955.
26. Hamed MA. Excretory-secretory product of Fasciola hepatica worm protects against Schistosoma mansoni infection in mice. Indian J Exp Biol 2006;44:554-561. PMID: 16872044.
27. Snapper CM, Mond JJ. Towards a comprehensive view of immunoglobulin class switching. Immunol Today 1993;14:15-17. PMID: 8442856.
28. Pfeifhofer C, Gruber T, Letschka T, Thuille N, Lutz-Nicoladoni C, Hermann-Kleiter N, Braun U, Leitges M, Baier G. Defective IgG2a/ 2b class switching in PKC alpha-/- mice. J Immunol 2006;176:6004-6011. PMID: 16670309.
29. Nascimento E, Leão IC, Pereira VR, Gomes YM, Chikhlikar P, August T, Marques E, Lucena-Silva N. Protective immunity of single and multi-antigen DNA vaccines against schistosomiasis. Mem Inst Oswaldo Cruz 2002;97:105-109. PMID: 12426603.
30. Dupré , Herv M, Schacht AM, Capron A, Riveau G. Control of schistosomiasis pathology by combination of Sm28GST DNA immunization and praziquantel treatment. J Infect Dis 1999;180:454-463. PMID: 10395862.
31. Ramos HR, Miyasato PA, Ramos CR, de Mattos Aréas AP, Kawano T, Ho PL. A genetic fusion between Sm14 and CTB does not reduce Schistosoma mansoni worm burden on intranasally immunized BALB/c mice. J Vaccines Vaccin 2010;1:111.
32. Pearce EJ, James SL, Hieny S, Lanar DE, Sher A. Induction of protective immunity against Schistosoma mansoni by vaccination with schistosome paramyosin (Sm97), a nonsurface parasite antigen. Proc Natl Acad Sci U S A 1988;85:5678-5682. PMID: 3135553.
33. Cardoso FC, Macedo GC, Gava E, Kitten GT, Mati VL, de Melo AL, Caliari MV, Almeida GT, Venancio TM, Verjovski-Almeida S, Oliveira SC. Schistosoma mansoni tegument protein Sm29 is able to induce a Th1-type of immune response and protection against parasite infection. PLoS Negl Trop Dis 2008;2:e308. PMID: 18827884.
34. Siddiqui AA, Phillips T, Charest H, Podesta RB, Quinlin ML, Pinkston JR, Lloyd JD, Pompa J, Villalovos RM, Paz M. Enhancement of Sm-p80 (large subunit of calpain) induced protective immunity against Schistosoma mansoni through co-delivery of interleukin-2 and interleukin-12 in a DNA vaccine formulation. Vaccine 2003;21:2882-2889. PMID: 12798631.
Fig. 2Hepatic granuloma diameter. (A) Hepatic sections of S. mansoni infected control animals at week 8 PI showed that granulomas were large, scattered of both cellular and fibrocellular types. Mild degree of hydropic degeneration was observed in many of the hepatocytes. (B) Hepatic sections of immunized infected group showed milder fibrosis, and chronic perivascular inflammation was still observed. 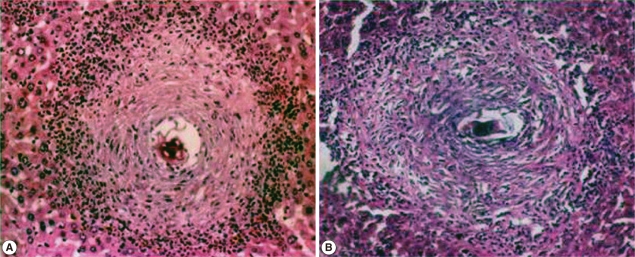
Table 1.Different parasitological parameters detected at week 8 PI in different animal groups
Table 2.Serum IgM, IgG (H+L), and various IgG isotypes levels against FABP antigen in different mice groups
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||