INTRODUCTION
Trichuriasis is a common soil-transmitted helminthiasis in tropical and subtropical regions with an estimated of 604-795 million infected people and a global prevalence of 22-44.9% [1,2]. In Latin America and Caribbean countries, it is estimated that 100 million people are infected with Trichuris trichiura (prevalence of 19%), where 66% of those infected were over 15 years of age [2,3]. In addition, the landscape of geohelminthiasis has also changed because the incidence has decreased significantly not only due to appropriate control measures by government but also by migration of rural people to urban zones [2,4]. The etiological agents of trichuriasis are Trichuris suis in swine, Trichuris vulpis in canids, and T. trichiura in man. Although T. vulpis or T. suis are rarely found in humans, they can cause an uncommon zoonosis. Light Trichuris infections are often asymptomatic but in heavy infections, some patients develop eosinophilia, dysentery syndrome, anemia, mucosal swelling, tenesmus, and rectal prolapse. The first case of T. vulpis in man was reported in 1956 by Hall and Sonnenberg [5] in a 4-year-old child. Since then, several cases have been reported in the literature as single infections in some communities [6-8] [6-13]. In addition, mixed infections by T. trichiura and T. vulpis were reported in Mexico [9,14].
The diagnosis of trichuriasis is based on the identification of eggs by coproparasitologic analysis. Trichuris eggs have particular morphological characteristics which, in routine analysis, technicians diagnose them only as T. trichiura eggs. However, T. vulpis eggs can occasionally be confused with T. trichiura eggs. Therefore, it is important to identify accurately T. vulpis eggs in stool examinations to perform a suitable diagnosis and hence to carry out appropriate actions. In the present study, we report a case of T. vulpis infection in a Mexican child.
CASE RECORD
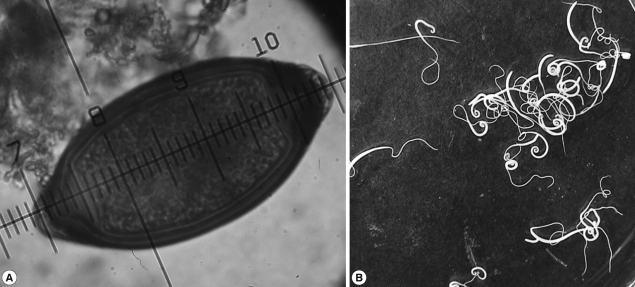
A 9-years-old girl was presented at the medical services with epistaxis lasting 2 years. The patient had nasal bleeding 4 times a day for 5 days without an apparent cause. Physical examination showed normal pharynx and moderate rhinorrhea. The x-ray did not show cardiopulmonary complications, and there were no symptoms of abdominal discomfort. The diagnosis was rhinitis. After that, the patient was presented at the medical services with a clinical history of abdominal discomfort and new epistaxis. Laboratory tests showed mild eosinophilia in a blood test. The patient submitted a fecal sample which was concentrated and examined following standard laboratory protocols. There were no worms in the feces but the examination with iodine wet mount showed the presence of yellowish-brown eggs with 2 polar plugs that resemble Trichuris eggs. The average size of the eggs was 90 µm in length and 44 µm in width (range 86-99 µm by 38-47 µm) (Fig. 1A).
Based on these findings, this case was diagnosed as T. vulpis (Froelich, 1789) infection. The infection probably did not occur at home, since no eggs of T. vulpis were detected in the coproparasitologic studies of their 2 pets, but in 292 fecal samples of dogs collected in a period of 5 days in the peri-domicile, prevalence of hookworms (28.1%), Toxocara canis (11.8%), and Trichuris vulpis (3.5%) was found. The patient was treated with mebendazole 100 mg per os twice daily for 3 days. From the first day of treatment, whitish worms were released in the feces (Fig. 1B). Numerous pieces of worms immersed in the semisolid feces were observed. The number of intact adult specimens collected were 315 and their morphology resembled T. vulpis. The size was 7.4±1.2 cm and the thinnest parts (esophagus) were reddish but soon they became whitish. The ratio of males and females was 1:4. In follow-up stool examinations, neither adult worms nor eggs of T. vulpis were found in there feces.
DISCUSSION
Micrometry is a useful tool in the identification of different stages of parasites. In many laboratories, the lack of an ocular micrometer makes these organisms misidentified during routine stool examinations. The eggs of T. trichiura measure 50 to 55 µm length by 20 to 25 µm width (Fig. 1A), while T. vulpis eggs measure 72 to 90 µm length by 32 to 40 µm width (Fig. 1A). However, occasionally a small proportion of large T. trichiura eggs were found which can measure up to 78 µm length and 30 µm width [10,15], while in T. vulpis, the majority of eggs are bigger (90 µm long and 74 µm wide). These differences have also been demonstrated by other authors [11,12,16,17]. There has been an approach using molecular tools, which could help identifying different species of Trichuris sp. [13,14,18,19], but their application on many laboratories is difficult, particularly in underdeveloped countries. Since T. vulpis eggs were not found in the feces of the family pet, we analyzed the feces of dogs at the peri-domicile. The frequency of T. vulpis was 3.5%. Therefore, the infection probably occurred through contact with dogs that are close to home.
T. vulpis have been found to cause visceral larva migrans [15,16,20,21]. The eosinophilia found in this patient may have been caused by migration of larval forms in early stages of the disease, so the rhinitis could be associated with the presence of T. vulpis. The nasal mucosa might have been involved and rhinitis was caused for a long period of time. This clinical presentation gradually decreased after the antiparasitic treatment.
The treatment of trichuriasis in humans is based on the use of mebendazole as a drug of choice [17,22], but the prevention and control measures are different when other species are found. So, it is imperative to achieve an accurate diagnosis in order to prevent further infections or reinfections, particularly considering the nature of T. vulpis as a zoonotic agent and the role of dogs as a source of infection. Due to a variety in the size of T. trichiura eggs in human feces, we recommend routine use of micrometry of eggs for an accurate identification of Trichuris species.