The nutria (Myocastor coypus) or coypu rat is a large rodent originally from South America [1]. It is now present in North America, Europe, Africa, and Asia (including Korea) and is used as a source of fur and meat [2]. A total of 100 nutrias were first imported to Korea from France in 1985, but they all died due to insufficient preparation of the environment required for them to adapt to the Korean winter. In 1987, a further 60 nutrias were imported from Bulgaria and were successfully bred. However, many nutrias were eventually released into the wild because there was no sufficient demand for them and the maintenance cost was high (personal communication). Although nutrias were considered to be maladapted to the Korean winter, they actually survived and spread to the south-eastern part of the Korean peninsula, parallel to the Nakdong-gang (River).
Nutrias are herbivores, eating as much as 25% of their body weight per day. They have been known to damage crops, because of their burrowing habits, as well as water-saving systems such as reservoirs. In addition, they are hosts to several zoonotic diseases caused by viruses, bacteria, and parasites [1]. They are known as a host of parasites that can be transmitted to other animals, including humans; Strongyloides myopotami, Schistosoma mansoni, Giardia lamblia, Taenia spp., and Fasciola hepatica [1]. Until now, no efforts have been made to monitor these diseases in Korea, particularly those transmitted by parasites. The present study examined the helminthic fauna of feral nutrias, in particular, a nematode, Strongyloides myopotami.
Total 10 adult nutrias (Fig. 1) were trapped near Gimhae-si (City), Gyeongsangnam-do (Province) in April 2013 and transported alive to our laboratory where they were euthanized with ether. Their entire digestive tract, respiratory organs, and subcutaneous tissues were examined by necropsy. The contents of the organs were washed and filtered through 200 µm meshes to recover worms, which were washed several times with saline and fixed in 10% formalin or 70% ethanol. For morphological studies, the specimens were transferred to a mixture of glycerin and 70% ethanol. When the ethanol evaporated entirely, they were observed and measured by light microscopy, and a scanning electron microscope was used (SEM, LEO-1530, Oberkochen, Germany), following Sato's method [3], to observe the type of the stoma (=mouth part).
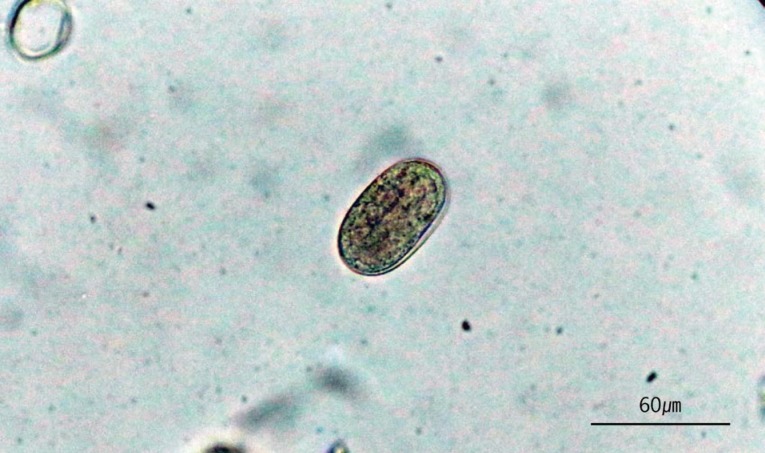
A total of 1,300 nematodes, all belonging to the same species, were isolated from all (100%) nutrias examined (n=10). The observed morphological characteristics of the worms were as follows (n=20): body small, thin, and elongated, 3.7-4.7 (4.0±0.36) mm in length, 30-40 (33±3) µm in width; oral opening complex, 8-lobed; nerve ring, 122-165 (150±16) µm from the anterior extremity; excretory pore, 150-202 (180±18) µm from the anterior extremity; esophagus long, divided into 2 parts on the basis of the nerve ring; muscular and glandular part, 820-1,162 (1,024±117) µm; uterus short, didelphic, 300-625 (451±100) µm; vulva with prominent lips, 976-1,503 (1,114±150) µm from the posterior extremity; ovary directly recurrent type; anus subterminal, 43-60 (53±5) µm from the posterior extremity; conical tail with blunt extremity (Fig. 2); eggs oval-shaped, embryonate in feces, 51-66 (58.0±4.5) µm in length and 21-27 (241.9) µm in width (Fig. 3).
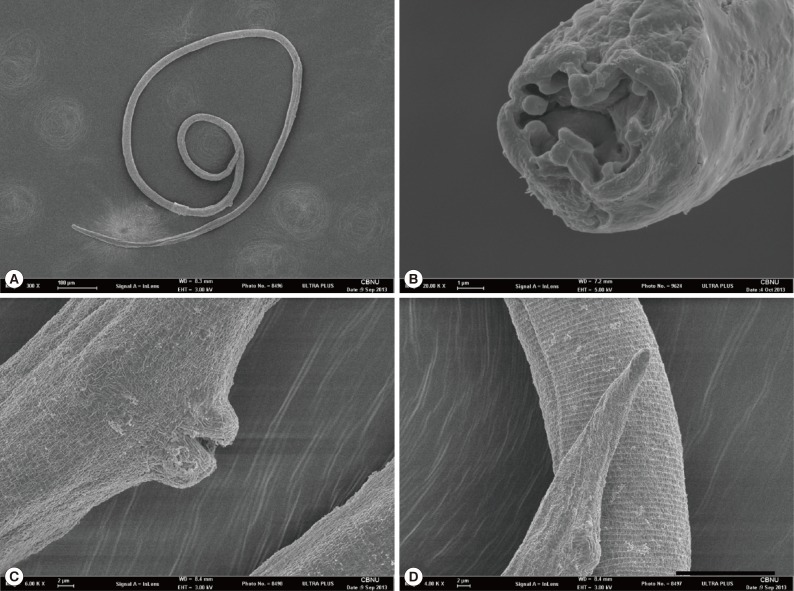
The genus Strongyloides has 5 major identifying characteristics, which include the shape of the stoma, the type of ovary, the shape of the tail, the number of lobes on the circumoral elevation of the parasitic female, and the shape of eggs by coproscopical examinations [4,5]. The body length of the female can also be used as a feature for identification along with other key characteristics [5]. S. myopotami has an ornate stoma with a circumoral elevation including 2 low, 2 broad, and 2 lateral lobes, an ovary running straight through the intestine, and the blunted conical tail [4]. In 2008, Sato et al. [3] re-defined the circumoral elevation of S. myopotami by using scanning electron microscopy, and described the inner surface as having 4 projections of different shapes; (1) 2 lateral prominent projections, (2) 1 dorsal, (3) 1 ventral, and (4) 2 subdorsal and 2 subventral projections. The specimens analyzed in this study displayed characteristics that were in line with those of S. myopotami, including size variation and prominent lips of the vulva (Fig. 4; Table 1). The morphological characteristics of our specimens were in agreement with those described in previous studies [4,5,6,7,8], confirming that these parasites are S. myopotami.
Infection rates of S. myopotami were as follows: 35.9-62.5% with 2-42 worms per host in Louisiana [6], 29 out of 108 nutrias (26.8%) [12], 11 out of 42 Ctenomys talarum (26.1%) in Argentina, and 1 out of 40 C. pearsoni (2.5%) harbored S. myopotami in Uruguay [8]. In the present study, at least 100 S. myopotami worms were recovered from each of the 10 nutrias analyzed (100%). Although these worms can be easily overlooked due to their small body buried deep in the mucosa [12], our results show that Korean nutrias have high infection rates and worm burdens higher than those reported in previous studies.
S. myopotami can cause parasitic dermatitis, also known as "nutria itch", "marsh itch", "swimmer's itch", or "creeping eruption". In 1960, Burks and Jung [9] reported the first case of suspected S. myopotami infection in 4 patients in Louisiana. Following this, in 1966, Little [10] performed experiments on a human volunteer by exposing him to S. myopotami and S. procyonis larvae, noting that dermatitis developed following exposure to infective larvae of S. myopotami and S. procyonis. This was in agreement with observations made by Burks and Jung [10] on cases of naturally acquired infections. Additional cases were reported in South Louisiana with positive enzyme immunoassay (EIA) titers confirmed [11]. Symptoms of the disease included severe pruritic rash, maculopapular erythmatous rash, and creeping eruption [9,10,11]. For all human cases of S. myopotami infection, it was confirmed that the patients had entered wetlands populated by nutrias before the occurrence of symptoms [9,10,11]. Although there have been no reports of human S. myopotami infection outside the USA, numerous nutrias have spread into south-eastern areas of the Korean peninsula, which has an abundance of stagnant marshes, such as rice fields. Accordingly, it is expected that infection rates and nutria populations would be high in Korea. Experiments in a human volunteer have shown that outbreaks of severe dermatitis tend to occur following repeated exposure to the pathogen [10]. In Korea, farmers who work in the rice fields have probably been exposed to these nematodes during the farming season. In this study, we report the first outbreak of S. myopotami in nutrias in Korea, although it is likely that this disease has already occurred without being identified.
The nutria is known as one of the reservoir hosts of several parasites that can infect humans, livestock, and pets. In 1961, Bert et al. [6] made a checklist of all the major references of helminthic parasites transmitted by the nutria between 1916 and 1959. This list included zoonotic helminthes, such as Fasciola gigantica, F. hepatica, Dicrocoelium lanceolatum, Schistosoma mansoni (experimental infection), Taenia metacestodes, Echinococcus granulosus, Capillaria hepatica, and Strongyloides spp. Furthermore, a study carried out in Argentina between December 2009 and June 2010 identified various zoonotic parasites, including nematodes, trematodes, and protozoans, transmitted by the nutria, namely, S. myopotami, C. hepatica, D. lanceolatum, F. hepatica, Cryptosporidium spp., and Giardia spp. [12]. S. myopotami [7,13] and Fasciola sp. [7] were also reported in Japan. In the present study, we found only 1 parasite species from 10 nutrias. However, considering their lifestyle and habits, they are strongly suspected to play a role as a natural definitive host for other zoonotic parasites, like F. hepatica in Korea.
In the present study, we report a zoonotic nematode (S. myopotami) infection in feral nutrias from Korea. Though we examined an insufficient number of samples, we confirmed high infection rates of S. myopotami. More epidemiological studies on this public issue are being considered to be necessary.