INTRODUCTION
Human sparganosis is a food-borne zoonosis mainly caused by the plerocercoid (spargana) belonging to the genus Spirometra [1]. Human sparganosis has been reported in many countries but East Asia is the most common place [2]. The infection is more frequently observed in Korea than in other countries. In humans, the infection is usually due to oral ingestion of larva-containing water or direct ingestion of raw amphibians or reptiles [3]. The most common clinical sign of sparganosis is a subcutaneous mass of body including abdominal or chest wall, scrotum, lower extremity [4]. The spargana are released into the intestine, penetrate the intestinal wall and migrate to subcutaneous and other tissues. The mass may be mistaken for a malignant tumor, thereby causing difficulty in terms of diagnosis and treatment [4]. The infection rate of parasites in Korea has been decreasing recently, but Korean surgeons still occasionally encounter cases of sparganosis. Given the rarity of the disease, the principles of diagnosis and treatment, as well as clinical characteristics, are not well understood. Here, we report 2 cases of sparganosis with masses located on the abdomen, which were first suspected as lipomas.
CASE REPORT
Case 1
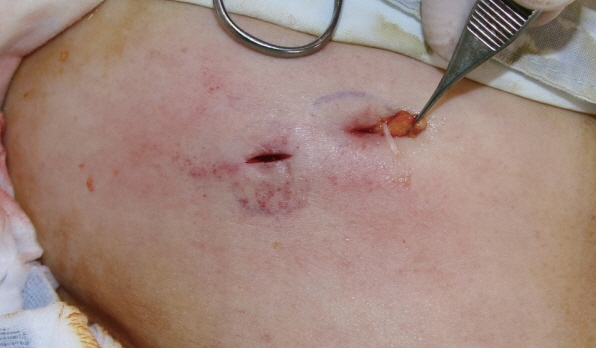
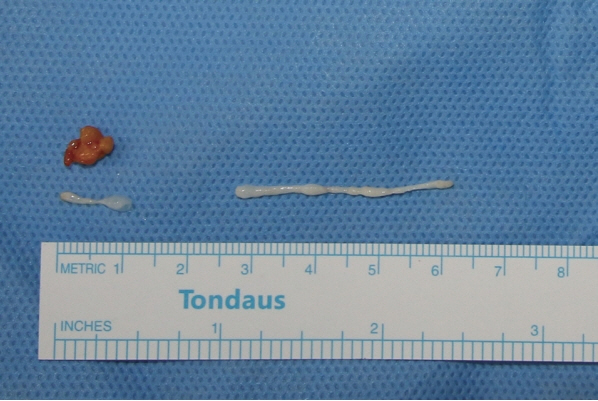
A 66-year-old woman visited our clinic for the removal of a lipoma-like abdominal mass that was movable, hard, and painless. Excision and biopsy were planned. Given the small size of the mass, we suspected a lipoma and did not perform any other preoperative tests. Intraoperatively, small incisions were made to explore the abdomen, but no pathologic lesions were identified (Fig. 1). The subcutaneous fat tissue appeared normal, and we identified 2 white masses, one measuring 0.2×4 cm and another measuring 0.2×1 cm (Fig. 2). A parasitic infection was suspected, and a biopsy was performed. Pathology results indicated the white mass was a parasite. We requested further information from the division of infectious diseases to identify the exact species of parasite. An antibody screening test was performed, and the results tested positive for sparganum. Following surgery, our patient has not experienced any postoperative complications. She recalled having eaten a raw frog approximately 60 years before, but besides that episode, she denied any history of having eaten the raw flesh of snakes or frogs and infected water.
Case 2
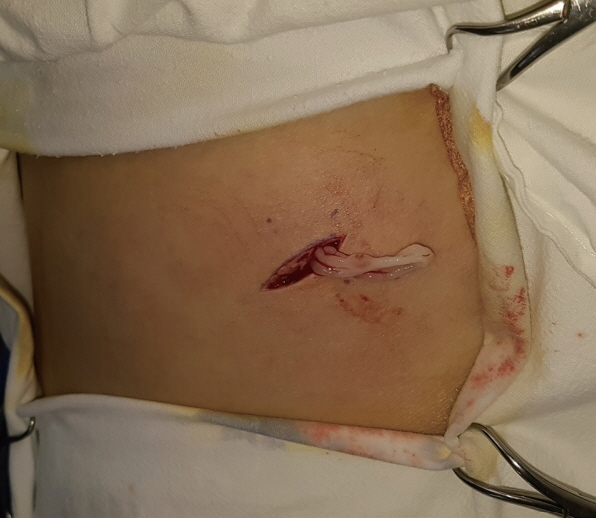
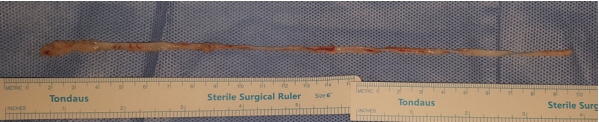
A 35-year-old woman presented to our clinic with an asymptomatic nodule on her abdomen that had been detected a few weeks previously. Physical examination revealed a solitary, moveable subcutaneous nodule, approximately 4 cm in diameter, and we suspected a lipoma. While performing an excisional biopsy of the lesion and while the incision was being made, a parasite emerged through the incision (Fig. 3). A 24 cm sized white, flat parasite emerged that was contracting and moving its body, and it was removed (Fig. 4). A histopathological study confirmed the parasite was a sparganum. Following surgery, our patient has had no complications. She denied consuming snake flesh or any kind of relevant high-risk food including well water, but she previously lived in North Korea where incidents of consuming high-risk food have been recorded as being more common.
An anti-helminthic drug (Praziquantel) was prescribed for prophylaxis, and the postoperative course was uneventful. One year postoperatively, this patient had no symptoms and showed no signs of recurrence of any lesion.
DISCUSSION
Sparganosis is defined as an infection caused by the larvae of parasitic tapeworms of the Spirometra species. The first case of human sparganosis in Korea was incidentally confirmed by Uemura (1917) in muscle fascia during surgery for a lower extremity amputation in a Korean farmer [5]. The most common location of sparganosis is in subcutaneous tissues [6]. When the spargana is released into intestine, it passes through intestinal wall and moves into subcutaneous tissues, as shown in examinations performed in rats. Humans are an intermediate host for the parasite. Infection occurs via several ways, including drinking water containing infected copepods; eating amphibian or rodent meat; and using the flesh of these creatures as a traditional self-treatment according to ritual belief. These risk behaviors have been identified in many reported cases and comprised 41.2% of reported cases, for example, in Thailand [2].
Park et al. [7] reported that a history of eating raw frog or snake is the most important risk factor for sparganosis, with an odds ratio of 3.1. Sparganosis manifests as a migrating subcutaneous nodule in the abdominal wall, chest wall, lower extremity, or scrotum. It has been reported that ingested spargana can invade various organs, such as the eye, subcutaneous tissues, abdominal wall, brain, spinal cord, lung, or breast. In the case of the genitourinary system, this invasion can include the epididymis, spermatic cord, penis, retroperitoneum, and the ureter. Clinically, patients with sparganosis present with vague or indeterminate symptoms, and distinguishing sparganosis from malignancy remains challenging [4].
The subcutaneous lesion appears as a clear, soft, irregular nodule with a diameter between 0.3 and 5 cm, similar to a fibroma, lipoma, or other neoplasm. With no specific symptom reflecting the presence of larvae in the peritoneum, subcutaneous masses may be the first sign of the disease when the larvae emerge from the peritoneal cavity. Subcutaneous masses are associated with clinical signs of inflammation or they may mimic a soft tissue neoplasm.
Serologic test like ELISA is best tool of diagnosis. Sonographic findings may also be helpful for the preoperative diagnosis of sparganosis. Serpiginous and anechoic tubular tracts with some internal echogenicity are important characteristics. Magnetic resonance imaging characteristics of sparganosis include multiple tubular tracts with low on T1-weighted images and high on T2-weighted images. However, these imaging studies provide limited evidence for making a diagnosis.
Repeated serological tests at appropriate intervals after removal can confirm the removal of the entire sparganum. If the mass is not suitable for surgical therapy, localized chemotherapy with 40% ethanol procaine or α-chymotrypsin can be administered. For patients who have pericardial or pleural effusion without a pleural or pericardial nodule, or who have visceral sparganosis, surgical treatment is not possible [1].
Surgeons should be recognize the possible diagnosis and potential longevity of the spargana. Although the survival duration time of spargana in human skin is not known precisely, there have been case reports of finding a live worm between 8 and 9 years after the development of a subcutaneous mass. We encountered an interesting case of sparganosis that was asymptomatic and latent for 60 years (case 1), and the sparganum was very long (24 cm in size) and was present within the subcutaneous fat layer (case 2). To our knowledge, our report presents the longest latent-occurring sparganosis and reports a patient who was treated for the removal of a very long-living sparganum.
While the incidence rate of sparganosis has decreased with economic development and advancements in sanitation, surgeons still encounter patients with sparganosis in the clinical setting. Therefore, a careful history taking is required in order to diagnose sparganosis.
In conclusion, sparganosis of the subcutaneous tissue is an uncommon disease and is seldom encountered in plastic surgery. Complete surgical removal of sparganosis is the best choice and provides a definite diagnosis. Recurrence depends on the location and the possibility of incomplete removal of the parasite. Once a confirmatory diagnosis is made, a thorough examination of the outer organs, including the abdominal wall and extremities, should be required to detect any concurrent involvement in these locations. Alternatively, a postoperative ELISA test may prevent non-detection of any additional worms.