AbstractAdult gnathostomes were discovered in the stomach of the Jeju weasel, Mustela sibilica quelpartis, road-killed in Jeju-do (Province). Their morphological characters were examined to identify the species. Total 50 gnathostome adults were collected from 6 out of 10 weasels examined. In infected weasels, 4-6 worms were grouped and embedded in each granulomatous gastric tumor, except 1 weasel. Male worms were 25.0×1.4 mm in average size, and had a tail with pedunculate papillae, a spicule, and minute tegumental spines. Females were 40.0×2.5 mm in average size, and had a tail without tegumental spines. Pointed and posteriorly curved hooklets were arranged in 8-10 rows on the head bulb. Tegumental spines were distributed from behind the head bulb to the middle portion of the body. The spines were different in size and shape by the distribution level of the body surface. Fertilized eggs were 65.5×38.9 µm in average size, and had a mucoid plug at 1 pole. These gnathostomes from Jeju weasels were identified as Gnathostoma nipponicum Yamaguti, 1941. By the present study, it was confirmed for the first time that G. nipponicum is distributed in Jeju-do, the Republic of Korea, and the Jeju weasel, M. sibilica quelpartis, plays a crucial role for its definitive host.
Nematode parasites of the genus Gnathostoma are clinically important, causing food-borne zoonoses in humans. More than 10 species have been reported in various parts of Asia, Europe, Oceania, and Americas. In the Republic of Korea, there had been no reports on gnathostome infections before a paper by Kim in 1973 [1]. He detected 2 Gnathostoma spinigerum larvae in the abdominal muscle of a snakehead, Channa argus argus, from Kimhae, Gyeongsangnam-do (Province). After that time, third stage larvae of Gnathostoma nipponicum and Gnathostoma hispidum were found in the imported Chinese loaches [2,3]. In addition, G. hispidum larvae were detected in pit-viper snakes, Agkistrodon brevicaudus, caught in Korea, and also in red banded odd-tooth snakes, Dinodon rufozonatum rufozonatum, from China [4,5]. However, adult gnathostomes had never been reported. In the present study, we found adult gnathostomes in the stomach of the Jeju weasel, Mustela sibilica quelpartis, road-killed in Jeju-do (Province), the Republic of Korea, and identified them as G. nipponicum on the basis of their morphological characters.
A total of 10 Jeju weasels, M. sibilica quelpartis, road-killed, were donated from the party of environmentalists of Jeju-do from April 2008 to April 2010 (Fig. 1). Their whole visceral organs were isolated to investigate parasite infections. Adult nematodes were collected from the stomach of the weasels. Some collected worms were fixed with 10% formalin, cleared in alcohol-glycerin solution, mounted in glycerin-jelly, and observed under a light microscope. To observe the surface ultrastructure, some worms were washed several times with 0.2 M cacodylate buffer (pH 7.2) and fixed with 2.5% glutaraldehyde at 4℃. After washing 3 times with the buffer, they were dehydrated through a graded alcohol series (50%, 70%, 80%, 90%, 95%, and absolute alcohol), dried with hexamethyldisilazane, coated (JFC-1100E ion sputtering device) with gold, and observed with a scanning electron microscope (Philips XL-30S, Einthoven, The Netherlands) at an accelerating voltage of 15 kV.
Total 50 gnathostomes were collected from 6 out of 10 weasels examined, and they were mainly from weasels caught in Jeju-si (Table 1). Except in 1 weasel, 4-6 worms were grouped and embedded in each granulomatous gastric tumor of weasels (Fig. 2). Pointed and posteriorly curved hooklets were arranged in 8-10 rows on the head bulb (Fig. 3A, 3B). Male worms were 22.0-27.0×1.2-1.9 (25.0×1.4) mm in size, and had a characteristic head bulb and a tail with pedunculate papillae, a spicule, and minute tegumental spines (Fig. 3C, 3D). Females were 35.0-45.0×2.3-2.8 (40.0×2.5) mm in size, and had a head bulb and a tail without tegumental spines. Fertilized eggs were 63.8-67.5×37.5-40.0 (65.5×38.9) µm in size, and had a mucoid plug at 1 pole (Fig. 4). Tegumental spines were distributed only on the anterior half of the body. They were different in size and shape by the distribution level of the body surface. Slender 3-4 tipped spines (31.4×11.6 µm in average size) were densely distributed just behind of the head bulb (Fig. 5A). Broader 3 tipped spines (44.6×18.3 µm in average size) with a long middle tip (25.0 µm in average length) were broadly distributed at the anterior half of the middle surface. Slender 3 tipped ones (34.0×11.8 µm in average size) and 2-3 tipped small pointed spines (about 20 µm in length) were distributed on the posterior half of the middle surface (Fig. 5B, 5C, 5D). No tegumental spines were distributed on the posterior half of the body surface, especially in females (Fig. 5E). In males, minute tegumental spines were distributed only at the tail portion.
It has been known that G. nipponicum is a common parasite found in esophageal tumors of weasels, Mustela sibirica itasi and Mustela sibirica coreana, only in Japan. This nematode was first found by Yoshida in 1931 [6] in the esophagus of a weasel from the middle part of Honshu in Japan, and erroneously named as G. spinigerum. Subsequently, by many Japanese workers, it was revealed that this parasite is widely distributed in more than 23 Prefectures in Japan [7-15]. By the present study, it was confirmed for the first time that G. nipponicum is distributed also in Jeju-do, the Republic of Korea, and the Jeju weasel, M. sibilica quelpartis, plays a crucial role for a definitive host.
Detailed morphological features of adult G. nipponicum have been described by Yamaguti in 1941 and later by Miyazaki and Umetani in 1950 [9,16]. Miyazaki [17] reviewed 10 valid species of Gnathostoma, i.e. G. spinigerum, G. hispidum, G. doloresi, G. nipponicum, G. turgidum, G. procyonis, G. miyazakii, G. malaysiae, G. vietnamicum, and G. americanum [17]. According to this review [17], the distribution and shape of tegumental (cuticular) spines on the body surface were treated as the most useful landmarks for species differentiation of Gnathostoma. In 6 species, G. spinigerum, G. nipponicum, G. turgidum, G. miyazakii, G. vietnamicum, and G. americanum, their tegumental spines are covered at the anterior half of the body, while in remaining 4 species, G. hispidum, G. doloresi, G. procyonis, and G. malaysiae, the tegumental spines are densely distributed on the whole body surfaces. However, G. nipponicum can be discriminated from other 5 species, including G. spinigerum by the morphology of tegumental spines and eggs. Broader 3 tipped spines with a long middle tip, which are broadly distributed at the anterior half of the middle surface, are most characteristic and comparable features for species identification. Additionally, the tegumental ultrastructures of adult G. nipponicum are clearly shown by the present study.
It has been known that G. nipponicum adults are found in esophageal tumors of weasels, M. sibirica itasi and M. sibirica coreana, while most of other Gnathostoma spp. are usually found in the stomach of definitive hosts, including cats, dogs, and pigs [17]. However, in the present study, the worms were detected in the stomach of Jeju weasels, M. sibilica quelpartis. It is interesting that habitats of G. nipponicum in the definitive hosts are different between weasels from Japan and from Korea. Molecular genetic studies may be needed to know what is differential points between Japanese and Korean worms, although they are morphologically identical.
ACKNOWLEDGEMENTSWe thank the party of environmentalists of Jeju-do (Province) for their help in collection of road-killed weasels.
Conflict of interestThe authors have no conflicts of interest concerning the work reported in this paper.
REFERENCES1. Kim YK. A study on Gnathostoma (1) An investigation into the geographical distribution of larvae on the second and third stage in Gyeongsang Nam do. Bull Pusan Nat Univ (Natur Sci) 1973;15:111-116.
2. Sohn WM, Kho WG, Lee SH. Larval Gnathostoma nipponicum found in the imported Chinese loaches. Korean J Parasitol 1993;31:347-352. PMID: 8297892.
3. Sohn WM, Lee SH. Identification of larval Gnathostoma obtained from imported Chinese loaches. Korean J Parasitol 1996;34:161-167. PMID: 8843692.
4. Sohn WM, Lee SH. The first discovery of larval Gnathostoma hispidum (Nematoda: Gnathostomidae) from a snake host, Agkistrodon brevicaudus. Korean J Parasitol 1998;36:81-89. PMID: 9637825.
5. Cho SH, Kim TS, Kong Y, Na BK, Sohn WM. Larval Gnathostoma hispidum detected in the red banded odd-tooth snake, Dinodon rufozonatum rufozonatum, from China. Korean J Parasitol 2007;45:191-198. PMID: 17876164.
6. Yoshida S. Gnathostoma spinigerum Owen, 1836, causing the esophageal tumor of the Japanese weasel. Nisshin Igaku 1931;20:1604-1618 (in Japanese).
7. Yoshida S. Observations on Gnathostoma spinigerum Owen, 1836, cause of esophageal tumor of the Japanese mink Luteola itatsi itatsi (Temmink, 1844), with especial reference to its life history. Jap J Zool 1934;6:113-122 (in Japanese).
8. Yoshida S. Contribution to the study on Gnathostoma spinigerum Owen, 1836, cause of esophageal tumor of the Japanese mink, with especial reference to its life history. Trans 9th Congress FEATM Nanking 1934;1:625-630 (in Japanese).
9. Miyazaki I, Umetani T. A morphological study of Gnathostoma nipponicum parasitic in the weasel in Kyushu. Jap J Clin Exp Med 1950;27:112-116 (in Japanese).
10. Katagiri S, Oturu M. Distribution of Gnathostoma nipponicum in Hokuriku and Ou districts, Japan (Nematoda: Gnathostomatidae). Med Biol 1957;43:199-202 (in Japanese).
11. Ashizawa H, Kugi G, Nosaka D, Tateyama S, Yanai T. Natural infection of weasels with Gnathostoma nipponicum in Oita Prefecture, Japan. Bull Fac Agr Miyazaki Univ 1978;25:85-92 (in Japanese).
12. Gyoten J, Nishida H. 1978. On the Gnathostoma nipponicum in Kagawa Prefecture. Jpn J Parasitol 1978;27:411-416 (in Japanese).
13. Koga M, Ishii Y. Larval gnathostomes found in reptiles in Japan and experimental life cycle of Gnathostoma nipponicum. J Parasitol 1981;67:565-570.
14. Ando K, Tanaka H, Ohkawa C. A survey of geographical distribution of Gnathostoma nipponicum in Mie, Nara, Kyoto and Shiga Prefectures, Japan. Jpn J Parasitol 1988;37:263-267 (in Japanese).
15. Oyamada T, Kudo N, Sakashita H, Yoshikawa T. The first report of Gnathostoma nipponicum in Aomori Prefecture. Jpn J Parasitol 1995;44:128-132.
16. Yamaguti S. Studies on the helminth fauna of Japan. Part 35. Mammalian nematodes II. Jap J Zool 1941;9:409-440.
17. Miyazaki I. Section III. Nematode Zoonoses. 33. Gnathostomiasis. An Illustrated Book of Helminthic Zoonoses. 1991, Tokyo, Japan. International Medical Foundation of Japan. pp 368-409.
Fig. 1The Jeju weasel, Mustela sibilica quelpartis, a new definitive host for Gnathostoma nipponicum, which was photographed by Hong-Shik Oh, one of the authors. 
Fig. 2Adult worms of G. nipponicum (arrow marks) embedded in the granulomatous gastric tumor of a weasel. Scale bar=2 cm. 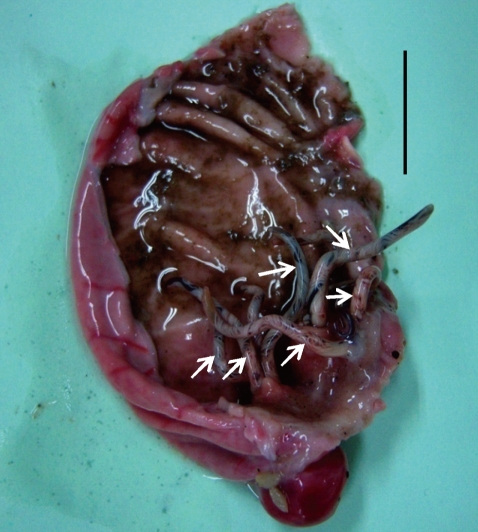
Fig. 3Morphological characters of G. nipponicum adults recovered from a Jeju weasel. Light (A) and scanning electron microscopic (SEM) (B) views of the head bulb with 10 rows of hooklets. Light (C) and SEM (D) views of the tail portion of a male with pedunculate papillae, a spicule, and minute tegumental spines. Scale bar=0.2 mm. 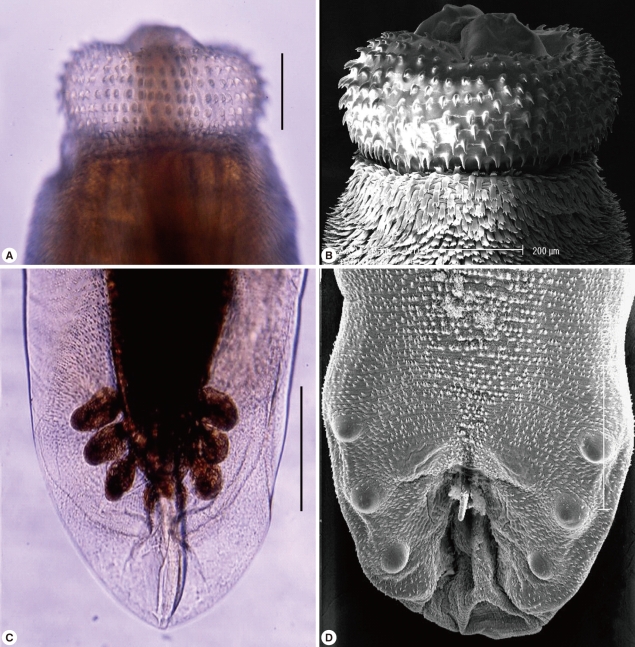
Fig. 4An egg of G. nipponicum collected from the uterus of an adult female (A). Developing eggs from the cultivating petri dish with freshwater at 28℃ are seen (B & C). Scale bar=25 µm. 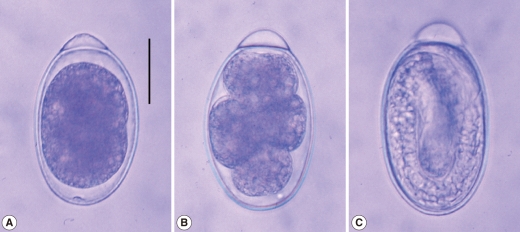
Fig. 5Tegumental ultrastructures of a G. nipponicum adult recovered from a Jeju weasel. 3-4 tipped slender cuticular spines distributed at the behind surface of the head bulb (A); 3 tipped broad spines with a middle long tip broadly distributed on the anterior half of middle surface (B); 3 tipped spines with a middle long tip (C) and 2-3 tipped small spines on the posterior half of middle surface (D); tegument without spines on the posterior half of body (E). Scale bar=25 µm. 
Table 1.The road-killed Jeju weasel, Mustela sibilica quelpartis, examined and the result of worm recovery
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||