Abstract
Plasmodium vivax reemerged in the Republic of Korea (ROK) in 1993, and is likely to continue to affect public health. The purpose of this study was to measure levels of anti-P. vivax antibodies using indirect fluorescent antibody test (IFAT) in border areas of ROK, to determine the seroprevalence of malaria (2003-2005) and to plan effective control strategies. Blood samples of the inhabitants in Gimpo-si, Paju-si, and Yeoncheon-gun (Gyeonggi-do), and Cheorwon-gun (Gangwon-do) were collected and kept in Korea Centers for Disease Control and Prevention (KCDC). Out of a total of 1,774 serum samples tested, the overall seropositivity was 0.94% (n=17). The seropositivity was the highest in Paju-si (1.9%, 7/372), followed by Gimpo-si (1.4%, 6/425), Yeoncheon-gun (0.67%, 3/451), and Cheorwon-gun (0.19%, 1/526). The annual parasite incidence (API) in these areas gradually decreased from 2003 to 2005 (1.69, 1.09, and 0.80 in 2003, 2004, and 2005, respectively). The highest API was found in Yeoncheon-gun, followed by Cheorwon-gun, Paju-si, and Gimpo-si. The API ranking in these areas did not change over the 3 years. The seropositivity of Gimpo-si showed a strong linear relationship with the API of 2005 (r=0.9983, P=0.036). Seropositivity data obtained using IFAT may be useful for understanding malaria prevalence of relevant years, predicting future transmission of malaria, and for establishing and evaluating malaria control programs in affected areas.
INTRODUCTION
Plasmodium vivax malaria is the second most serious form of human malaria after Plasmodium falciparum. Malaria affects several hundred million individuals annually and is a major public health problem in most tropical regions, as well as in many temperate countries [1]. In the Korean peninsula, the first scientific documentation of malaria was in 1913, although malaria had already been prevalent throughout the area for several centuries [2,3]. The incidence of vivax malaria in Republic of Korea (ROK) has rapidly decreased in the 1960's [4,5], because of a national malaria eradication program involving the World Health Organization (WHO). In fact, it was thought that vivax malaria was eradicated in the ROK in the late 1970s, although 2 sporadic cases were detected in the 1980s [6]. In 1993, a case was diagnosed as vivax malaria among ROK soldiers serving in the northern part of Gyeonggi-do [7]. Subsequently, 2 cases were reported among civilians [8]. Thereafter, many cases have been reported near the demilitarized zone (DMZ), centering on the cities and counties of Paju-si, Yeoncheon-gun, Cheorwon-gun, Gimpo-si, Ganghwa-gun, Goyang-si, and Dongducheon-si. There is considerable concern that malaria will be re-established in the region and will then expand geographically [9].
The malaria research team at the Korea Centers for Disease Control and Prevention (KCDC) has developed several diagnostic methods to support pathological examinations. In particular, an indirect fluorescent antibody test (IFAT) was developed for detecting antibodies in blood samples. Such serological data can provide evidence regarding the extent and degree of malaria endemicity and can reflect the period of infection [10,11]. Serological data provides valuable epidemiological information, especially in areas of low endemicity [12-14]. The rate of parasitemia has been used as a classical method of measuring the endemicity of malaria, but may be insufficient to adequately describe the epidemiology of malaria in a given population. When the incidence of malaria is low, mass blood film examination does not yield results commensurate with the work involved [15,16]. In the present study, the IFAT serological test was compared with the index of annual parasite incidence (API) to evaluate potential relationships between the test results and the index in areas with a high malaria risk in ROK.
MATERIALS AND METHODSInformation of blood sample collectionThe study was conducted in Gimpo-si, Paju-si, and Yeoncheon-gun (Gyeonggi-do) and Cheorwon-gun (Gangwon-do) from November to December of 2004. Blood samples were collected from 1,774 inhabitants and kept in KCDC. Blood smears were prepared for microscopic examinations. Sera were separated and stored at -20℃ for antibody analysis. Informed consent was obtained from each of the individuals. All samples were collected using protocols that were reviewed and approved by the Human Ethics Committee of the KCDC.
Microscopic examinationsThin blood films were prepared to determine the infectivity of blood samples. Each film was methanol-fixed and stained with Giemsa, using buffered water at pH 7.2, to emphasize the parasite inclusions in RBCs. Compared with thick film examination, the fixed monolayer of RBCs in this procedure makes the parasitemia easier to count and morphological identification of the parasite to the species level much more accurate [17]. To estimate the densities of blood-stage parasites by microscopy, we counted the number of asexual parasites observed relative to 200 WBCs and then multiplied the parasite:WBC ratio by 8,000, that is, the assumed number of WBCs per microliter of blood [18].
Indirect fluorescent antibody test (IFAT)To test for antibodies against malaria, IFAT was performed using fixed blood-stage parasites of P. vivax [14]. Briefly, malaria-infected blood was collected from P. vivax patients. After removal of plasma, RBCs were suspended in PBS (pH 7.2) and centrifuged for 5 min at 2,500 rpm. Supernatants were discarded, and the RBCs were suspended in fresh PBS and washed, as described, 3 more times. Finally, an appropriate amount of PBS was added to the RBCs, and the solution was added to each well of Teflon-coated slides. The slides were dried at room temperature for 12 hr, and stored at -70℃ until use. To determine antibody titers against P. vivax, antigen slides were fixed in precooled acetone (-20℃) for 10 min and washed with PBS, and then 20 µl of each test sera titrated from 1:32 to 1:8,192 (vol/vol with PBS) was added to the wells. Positive and negative controls were added to each slide at the same dilutions as the test sera, and the slides incubated for 30 min in a humidified chamber at 37℃. The slides were washed by immersion in PBS for 6 min and dried at room temperature. Diluted FITC-conjugated anti-human IgG (Sigma, St. Louis, Missouri, USA), 1:32 vol/vol in PBS, was then added to the wells, and the slides were incubated and washed using the methods described above. Several drops of buffered glycerol were added to the samples, and the slides were covered with coverslips. Slides were examined by a fluorescence microscope (×200 and/or ×600, Nikon ECLIPSE TE2000-S).
Calculation of the APIMalaria is classified as one of the Group III communicable diseases that should be controlled by the Korean Government. The cases of malaria detected in private hospitals or clinics are reported to the local Public Health Center (PHC). The data collected by the PHCs is provided periodically to the Division of Infectious Disease Surveillance (DIDS), KCDC. Therefore, APIs of 2003, 2004, and 2005 were obtained from DIDS. The API was calculated as the number of malaria-positive patients per 1,000 inhabitants for each of the study site as determined by microscopy: API=(number of microscopically proven malaria cases/1,000 population/year).
Pearson correlation analysisThe relationship between the seropositivity and API of P. vivax in a given year was analyzed using Pearson's correlation. The data were analyzed using SPSS software (version 17.0, SPSS Inc.) (Chicago, Illinois, USA). A P-value of less than 0.05 was considered statistically significant. The degree of correlation was interpreted as none (0.0-0.09), small (0.1-0.3), medium (0.3-0.5), or strong (0.5-1.0) [19].
RESULTSBlood collectionA map of the study locations is shown in Fig. 1. All areas are located near the DMZ, a known high-risk area for malaria transmission. Blood samples were collected from participants residing in 23 villages of 3 cities (Gimpo-si, Paju-si, and Yeoncheon-gun) located in Gyeonggi-do and 6 villages in Cheorwon-gun, Gangwon-do. A total of 1,774 blood samples (1.9%) were collected and tested. The total number of inhabitants in the study areas in 2004 was 92,246.
Overview of malaria transmission in study areasThe criteria for a positive IFAT result had been established in a previous experiment [20]. Briefly, sera from 86 normal uninfected individuals showed reactivity at serum dilutions below 1:16 (range, 0-1:16), while sera from 58 patients infected with P. vivax showed reactivity at serum dilutions over 1:256 (range, 1:256-1:4,096). Therefore, a positive antibody response against vivax malaria was defined as a serum dilution ≥ 1:32. Additionally, positive and negative sera were applied on each slide at the same dilutions as the tested sera for quality control (Fig. 2). When sera showed an equivocal reaction between positive and negative reactions, the tests were repeated until clear results were obtained. Samples from 17 of the 1,774 study subjects (0.94%) showed positive reactions in IFAT. Samples from Paju-si (Fig. 1B) showed the highest positivity rate (7/372, 1.9%), followed by Gimpo-si (Fig. 1A, 6/425, 1.4%), Yeoncheon-gun (Fig. 1C, 3/451, 0.67%), and Cheorwon-gun (Fig. 1D, 1/526, 0.19%). The range of API results was consistent in Gimpo-si, Yeoncheon-gun, and Cheorwon-gun during the 3 years from 2003 to 2005. Yeoncheon-gun showed the highest API over the 3 years, followed by Cheorwon-gun, Paju-si, and Gimpo-si (unpublished data). The seropositivity of IFAT showed a negative linear relationship with the API of 2005 (r=-0.5682, P=0.432), 2003 (r=-0.4492, P=0.551), and 2004 (r=-0.3823, P=0.618) (Table 1). However, it failed to detect parasitic infections among seropositive individuals.
Malaria transmission in Gimpo-siBlood samples were collected from Haseong-myeon, Yangchon-myeon, and Wolgot-myeon in Gimpo-si. In Wolgot-myeon, samples from 4 of 166 inhabitants (2.4%) yielded positive results in IFAT. Wolgot-myeon is located in the northwest part of Gimpo-si and is close to South Hwanghae-do in North Korea, which is an area with epidemic malaria. The IFAT results showed that Wolgot-myeon had the highest positive rate (4/166, 2.4%), followed by Yangchon-myeon (2/206, 0.97%) which is located in the middle of Gimpo-si, and Haseong-myeon (0/53, 0%) which is in the northeastern part of Gimpo-si and is also close to South Hwanghae-do. Wolgot-myeon showed the highest API during all 3 years. Haseong-myeon showed the second-highest API in 2003 and 2004, but there were no patients in this region in 2005. Yangchon-myeon had the third-highest API in 2003 and 2004 and second-highest API in 2005 (unpublished data). The seropositivity detected by IFAT showed a strong positive linear relationship with the API of 2005 (r=0.9984, P=0.036) and 2004 (r=0.8206, P=0.387), and a small weak positive relationship with the API of 2003 (r=0.2385, P=0.847) (Table 2).
Malaria transmission in Paju-siBlood samples were collected from Munsan-eup and Papyeong-myeon in Paju-si. In Papyeong-myeon, samples from 3 of 140 inhabitants (2.1%) showed a positive response in IFAT (Fig. 1d). Papyeong-myeon had a higher positive rate than Munsan-eup (4/232, 1.7%, Fig. 1e). During all 3 years, Papyeong-myeon had a higher API than Munsan-eup (unpublished data). Thus, positive IFAT results in both areas showed a strong positive linear relationship with APIs of 2003, 2004, and 2005 (r=1.0000; Table 3).
Malaria transmission in Yeoncheon-gunBlood samples were collected from Wangjing-myeon, Baekhak-myeon, and Misan-myeon in Yeoncheon-gun. In Misan-myeon, which is located in the middle of Yeoncheon-gun and is close to the Hantan River, a sample from 1 of 75 inhabitants (1.3%) showed a positive IFAT result (Fig. 1h). Samples from Baekhak-myeon (Fig. 1f), which is located in the northwest and close to the DMZ, showed the second-highest positive IFAT results (2/265, 0.75%). The next was from Wangjing-myeon (Fig. 1g; 0/111, 0%), which is located in the northwest of Yeoncheon-gun. Baekhak-myeon showed the highest API in 2003 (5.36) and 2004 (3.69) and then gradually dropped to third place in 2005 (1.34). Misan-myeon showed the second-highest API in 2003 (1.68) and 2004 (2.79) but ranked first in 2005 (2.79). Wangjing-myeon showed the lowest API in 2003 (1.60) and 2004 (1.60) and then ranked second in 2005 (2.40) (unpublished data). The seropositivity of IFAT showed a strong positive linear relationship with the API of 2004 (r=0.6266, P=0.569), a small positive linear relationship with the API of 2005 (r=0.1881, P=0.880), and no relationship with the API of 2003 (r=0.0922, P=0.941) (Table 4).
Malaria transmission in Cheorwon-gunBlood samples were collected from inhabitants of Gimhwa-eup (Fig. 1i), Seo-myeon (Fig. 1j), Geunnam-myeon (Fig. 1l), and Cheorwon-eup (Fig. 1k) in Cheorwon-gun. Only 1 sample obtained from 142 inhabitants (0.7%) in Cheorwon-eup yielded positive IFAT results. No positive results were seen in Gimhwa-eup (0/115, 0%), Seo-myeon (0/126, 0%), or Geunnam-myeon (0/143, 0%). The API of Cheorwon fluctuated by area during the 3 years. Geunnam-myeon showed the highest API in 2003 (2.67), followed by Cheorwon-eup (1.90), Gimhwa-eup (1.85), and Seo-myeon (0.99) (unpublished data). However, Seo-myeon (1.98) had the highest malaria prevalence in 2004, followed by Gimhwa-eup (1.54), Cheorwon-eup (0.86), and Geunnam-myeon (0.45). Gimhwa-eup showed the highest API in 2005 (2.46), followed by Geunnam-myeon (2.23), Seo-myeon (0.49), and Cheorwon-eup (0.35). The seropositivity of IFAT showed a strong negative linear relationship with the API of 2005 (r=-0.6163, P=0.384), a medium negative linear relationship with the API of 2004 (r=-0.3389, P=0.661), and no relationship with the API of 2003 (r=0.0461, P=0.954) (Table 5).
DISCUSSIONTo evaluate malaria transmission in a given geographical region, many factors such as temperature, mosquito density, vectorial capacity, climate, rainfall, and humidity need to be considered [21]. However, collecting these data is costly and time-consuming. A simple and inexpensive tool is therefore needed to give rapid and accurate assessment of malaria epidemiology and transmission potential. Such a tool may also be useful for researchers initiating malaria studies in areas where no previous data are available. Although parasitemia is a useful means for measuring malaria endemicity, it cannot provide adequate information on malaria prevalence. In areas, such as ROK where has a low incidence of malaria and where patients have long incubation periods, or in areas with many asymptomatic patients, parasitemia as a diagnostic measure is limited [16]. The KCDC malaria research team had previously suggested that IFAT had the capacity to compensate for some of the disadvantages inherent in estimates of parasitemia [15]. This is supported by works in China, which showed that IFAT could help determine the prevalence of malaria within a population [16]. A preliminary study in Ganghwa-gun showed that 16.7% (4/24) of the seropositive IFAT experienced malaria in the following year (authors' unpublished data). In another study in Gimpo-si in 1999, PCR analysis yielded positive results for 16 of 125 seropositive IFAT patients (12.8%) [22]. This result suggests that seropositive individuals may include asymptomatic patients who must have had a history of exposure to malaria parasites.
Some, but not all, human malaria parasites can survive in the liver as hypnozoites. P. vivax parasites cannot survive for long periods in human blood due to the host immunity but sequester in the liver, where they can evade the immune system [22,23]. The incidence of malaria in ROK peaks in August, after the rainy season, and declines to baseline by mid-October. Blood collection was carried out between mid-November and mid-December, when the active anopheline population had diminished. Antibody detection can therefore provide useful information regarding P. vivax infection in a previously naïve population and is useful for identifying asymptomatic patients. Furthermore, the mean incubation period of P. vivax has been reported to be as long as 279±41 days (range, 153-452 days) [24]. Another study estimated the percentages of patients with short and long incubation periods as 25% and 75%, respectively [6].
In the present study, the seropositivity of IFAT was compared with the APIs of the previous (2003, 1,164 cases), relevant year (2004, 864 cases), and following year (2005, 1,311 cases) to assess the relationship. We chose the blood samples which were collected in 2004, because it was the first time to reach the bottom line of malaria incidence during 2 decades after reemerging in ROK and then went up again in 2005 [25]. Malaria incidence of west side (Paju-si and Gimpo-si) was decreased during 3 years, but that of east side (Cheorwon-gun) was decreased in 2004 and then increased again in 2005 (Table 1). After that, there were 2 more deeps in 2008 (1,009 cases) and 2012 (555 cases) [25]. Interestingly, the malaria situation was vice versa, malaria incidence of east side (Gangwon-do) decreased rapidly during 2010-2012 (184, 94, and 13 cases, respectively), but that of west side (Incheon) was reduced and then increased (256, 124, and 143 cases, respectively) [25]. The fluctuation of malaria incidence and areas might be influenced by the malaria situation of North Korea. The other word, it shows border malaria characters.
The seropositivity for Gimpo-si showed a strong linear relationship with the API of 2005 (r=0.9983, P=0.036). The other areas and years showed medium and strong correlations between seropositivity and API but this was not statistically significant. This correlation might be improved by increasing the number of study areas in the future. Interestingly, the seropositivity of all 4 high-risk areas showed a higher relationship with the API of the following year (2005) than that of the relevant year (2004) and previous (2003) years. This finding suggests that the relationship between seropositivity in IFAT and API is strongly influenced by the long incubation period for P. vivax malaria. In ROK, patients infected in a year may experience disease after the winter season in the following year. In other words, malaria transmission could potentially be predicted from the seropositivity in the previous year. Many factors such as the density of mosquitoes, vectorial capacity, ratio of long- to short- incubation periods among patients, symptomatic to asymptomatic patient ratio, differences in rainfall and temperature, and community immunity may all affect these results. It would require considerable time and efforts to elucidate the effects and relationships between these factors on malaria transmission in ROK. However, the present study shows clearly that antibody detection is closely related to API values. Thus, antibody detection will help us to understand the basis of malaria transmission in a given area, even in the absence of more comprehensive information [13,22].
The classical measurements of parasitemia, to determine malaria endemicity cannot provide a complete understanding of malaria prevalence, in areas such as ROK, where many patients have long incubation periods or are asymptomatic. The present study shows that antibody detection was closely related to API values; thus, antibody detection methods such as IFAT might be more efficient and cost-effective than microscopic examinations for the study of malaria transmission in ROK.
Korean National Institute of Health, National Research Foundation Grant2011-0028135
Inha University Research Grant ACKNOWLEDGMENTSWe are grateful to all the blood donors and to the staff members of Public Health Centers in Gimpo-si, Paju-si, Yeoncheon-gun, and Cheorwon-gun. We appreciate the help provided by Ms. Yeon-Joo Kim for the preparation of IFAT antigen slides. This work was supported by the Korean National Institute of Health, National Research Foundation Grant (NRF, no. 2011-0028135) funded by the Korean Government and by an Inha University Research Grant.
REFERENCES1. Mendis K, Sina BJ, Marchesini P, Carter R. The neglected burden of Plasmodium vivax malaria. Am J Trop Med Hyg 2001;64:97-106. PMID: 11425182.
2. Chu JK. A century history of parasitology in Korea. Seoul, Korea. Kookje Kyoyok Munwha. 1986, pp 1-144 (monograph in Korean).
3. Chun CH, Kim JJ. Malaria in Korea. Korean Med 1959;2:63-66 (in Korean).
4. National Malaria Eradication Service, Ministry of Health and Social Affairs, Republic of Korea. Malaria pre-eradication programme in Korea, 1961-1965. Seoul, Korea. Progress Report. 1966, pp 44-70 (in Korean).
6. Soh CT, Lee KT, Im KI, Min DY, Ahn MH, Kim JJ, Yong TS. Current status of malaria in Korea. Yonsei Rep Trop Med 1985;16:11-18.
7. Chai IH, Lim GI, Yoon SN, Oh WI, Kim SJ, Chai JY. Occurrence of tertian malaria in a male patient who has never been abroad. Korean J Parasitol 1994;32:195-200 (in Korean). PMID: 7953245.
8. Cho SY, Kong Y, Park SM, Lee JS, Lim YA, Chae SL, Kho WG, Lee JS, Shim JC, Shin HK. Two vivax malaria cases detected in Korea. Korean J Parasitol 1994;32:281-284. PMID: 7834248.
9. Lee JS, Kho WG, Lee HW, Seo M, Lee WJ. Current status of vivax malaria among civilians in Korea. Korean J Parasitol 1998;36:241-248. PMID: 9868889.
10. Jeffery GM, McWilson W, Collins WE, Lobel H. Application of the indirect fluorescent antibody method in a study of malaria endemicity in Mato Grosso, Brazil. Am J Trop Med Hyg 1975;24:402-411. PMID: 1098491.
11. Vafa M, Israelsson E, Maiga B, Dolo A, Doumbo OK, Troye-Blomberg M. Relationship between immunoglobulin isotype response to Plasmodium falciparum blood stage antigens and parasitological indexes as well as splenomegaly in sympatric ethnic groups living in Mali. Acta Trop 2009;109:12-16. PMID: 18831954.
12. Razakandrainibe R, Thonier V, Ratsimbasoa A, Rakotomalala E, Ravaoarisoa E, Raherinjafy R, Andrianantenaina H, Voahanginirina O, Rahasana TE, Carod JF, Domarle O, Menard D. Epidemiological situation of malaria in Madagascar: baseline data for monitoring the impact of malaria control programmes using serological markers. Acta Trop 2009;111:160-167. PMID: 19376076.
13. Curado I, Dos Santos Malafronte R, de Castro Duarte AM, Kirchgatter K, Branquinho MS, Bianchi Galati EA. Malaria epidemiology in low-endemicity areas of the Atlantic Forest in the Vale do Ribeira, São Paulo, Brazil. Acta Trop 2006;100:54-62. PMID: 17126279.
14. Cerutti C Jr, Boulos M, Coutinho AF, Hatab MC, Falqueto A, Rezende HR, Duarte AM, Collins W, Malafronte RS. Epidemiologic aspects of the malaria transmission cycle in an area of very low incidence in Brazil. Malar J 2007;6:33. PMID: 17371598.
15. Collins WE, Skinner JC. The indirect fluorescent antibody test for malaria. Am J Trop Med Hyg 1972;21:690-695. PMID: 4627546.
16. Duo-Quan W, Lin-Hua T, Zhen-Cheng G, Xiang Z, Man-Ni Y. Application of the indirect fluorescent antibody assay in the study of malaria infection in the Yangtze River Three Gorges Reservoir, China. Malar J 2009;8:199. PMID: 19678949.
17. Moody A. Rapid diagnostic tests for malaria parasites. Clin Microbiol Rev 2002;15:66-78. PMID: 11781267.
18. McKenzie FE, Prudhomme WA, Magill AJ, Forney JR, Permpanich B, Lucas C, Gasser RA Jr, Wongsrichanalai C. White blood cell counts and malaria. J Infect Dis 2005;192:323-330. PMID: 15962228.
19. Cohen J. Statistical Power Analysis for the Behavioral Sciences. 2nd ed. Psychology Press. 1988.
20. Lee HW, Lee JS, Lee WJ, Cho SH, Lee HS. The evaluation of recombinant circumsporozoite protein in malaria diagnosis. Korean J Microbiol 2000;36:142-149 (in Korean).
21. Snow RW, Gilles HM. The Anopheles vector. Essential Malariology. 4th ed. London, UK. Arnold. 2002, pp 59-84.
22. Lee WJ, Kim HH, Hwang SM, Park MY, Kim NR, Cho SH, In TS, Kim JY, Sattabongkot J, Sohn Y, Kim H, Lee JK, Lee HW. Detection of an antibody against Plasmodium vivax in residents of Gimpo-si, South Korea, using an indirect fluorescent antibody test. Malar J 2011;10:19. PMID: 21281481.
23. Park CG, Chwae YJ, Kim JI, Lee JH, Hur GM, Jeon BH, Ko HS, Han JE, Lee SJ, Park JW, Kaslow DC, Strickman D, Roh CS. Serologic responses of Korean soldiers serving in malaria-endemic areas during a recent outbreak of Plasmodium vivax. Am J Trop Med Hyg 2000;62:720-725. PMID: 11304063.
24. Kho WG, Jang JY, Hong ST, Lee HW, Lee WJ, Lee JS. Border malaria characters of reemerging vivax malaria in the Republic of Korea. Korean J Parasitol 1999;37:71-76. PMID: 10388264.
25. Kim TS, Kim JS, Na BK, Lee WJ, Kim HC, Youn SK, Gwack J, Kim HS, Cho PY, Ahn SK, Cha SH, Park YK, Lee SK, Kang YJ, Sohn Y, Hong Y, Lee HW. Decreasing incidence of Plasmodium vivax in the Republic of Korea during 2010-2012. Malar J 2013;12:309. PMID: 24010412.
Fig. 1Study areas. (A) Gimpo-si, (B) Paju-si, (C) Yeoncheon-gun, (D) Cheorwon-gun. a, Haseong-myeon; b, Wolgot-myeon; c, Yangchon-myeon; d, Papyeong-myeon; e, Munsan-eup; f, Baekhak-myeon; g, Wangjing-myeon; h, Misan-myeon; i, Gimhwa-eup; j, Seo-myeon; k, Cheorwon-eup; l, Geunnam-myeon; DPRK, Democratic People's Republic of Korea; ROK, Republic of Korea; DMZ, demilitarized zone. 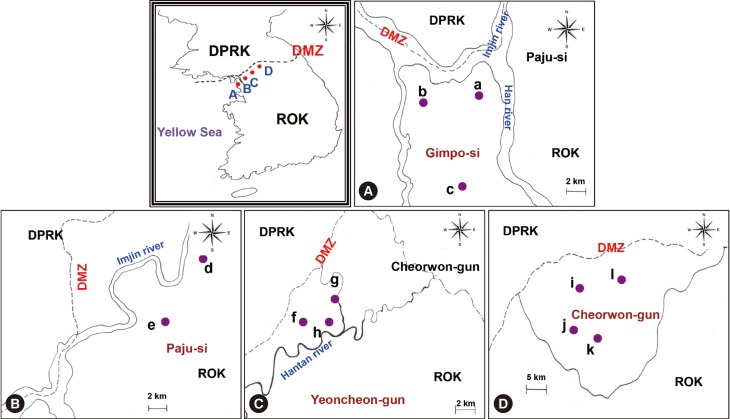
Fig. 2Immune responses between serum of vivax malaria patient (A) or normal person (B) and RBCs infected with Plasmodium vivax parasites. →; representative positive reaction. 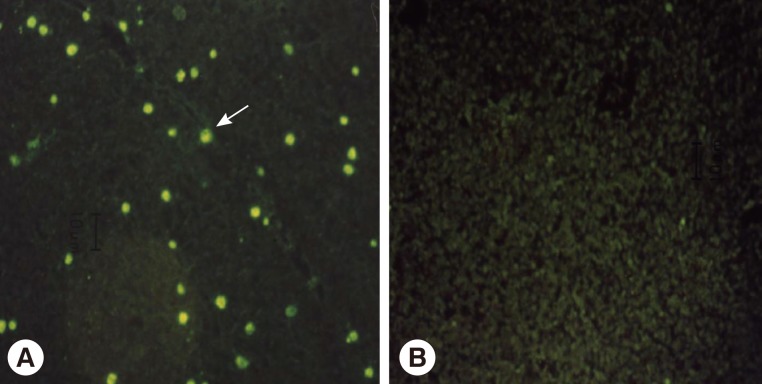
Table 1.Overall positive rates obtained from the data for the indirect fluorescent antibody test and annual parasite incidence for Gimpo-si, Paju-si, Yeoncheon-gun, and Cheorwon-gun Table 2.Positive rates obtained from the data for the indirect fluorescent antibody test and annual parasite incidence for Gimpo-si Table 3.Positive rates obtained from the data for the indirect fluorescent antibody test and annual parasite incidence for Paju-si
Table 4.Positive rates obtained from the data for the indirect fluorescent antibody test and the annual parasite incidence for Yeoncheon-gun Table 5.Positive rates obtained from the data for the indirect fluorescent antibody test and annual parasite incidence for Cheorwon-gun |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||