INTRODUCTION
Cutaneous leishmaniasis (CL) is a protozoan infection caused by protozoa of the genus Leishmania. This disease is characterized by chronic skin lesions and leaves permanent scars with deformation of the infected area [1]. CL is a public health problem at a global level because of affecting annually 1.5 million people worldwide [2]. Both epidemiological forms of this skin disease are present In Iran; anthroponotic CL (ACL) and zoonotic CL (ZCL) caused by Leishmania tropica and Leishmania major, respectively [3]. Current first choice chemotherapy of CL with antimonial drugs such as meglumine antimoniate and sodium stibogluconate is a challenge because of limited efficacy, toxic side effects, and drug resistance [4]. Since there is no effective vaccine for prevention, maintenance, and improvement of existing treatment regimens, combined with new drug discovery initiatives, appear to be the only ways to guarantee continued control of this important tropical disease.
Since ancient time, plant extracts and plant-derived compounds, due to having fewer side effects, low cost, and high availability, are valuable sources that are commonly used to treat a wide range of disease conditions including infectious diseases [5]. Myrtus communis L. (Myrtaceae) with the common name "myrtle" is native to Southern Europe, North Africa, and Asia. Different parts of this plant such as its berries, branches, leaves, and fruits have been used extensively as a folk medicine for the treatment of diarrhea, peptic ulcers, bleeding, headache, palpitation, leucorrhoea, urethritis, conjunctivitis, and pulmonary and skin diseases [6]. Moreover, reviews have reported M. communis as having anti-inflammatory, antinociceptive, antioxidant, neuro-protective, anti-diabetic, and antimicrobial effects [6-9]. Previous studies have also shown that the main constituents of the essential oil of M. communis are terpenoid compounds (1,8- cineole, α-pinene, myrtenyl acetate, limonene, linalool, and α-terpinolene) [6]. However, composition of the essential oil is quite variable depending on the geographic region of production, the season of harvest, and the length of distillation [10]. The present study aimed to evaluate leishmanicidal effects of methanolic extract and essential oil of M. communis against promastigote and amastigote forms of L. tropica and their cytotoxic activities against J774-A1 cells on an in vitro model.
MATERIALS AND METHODS
Chemicals
All the chemicals and solvents used were of the highest purity commercially available. Meglumine antimoniate (MA) (Glucantime) as a control was purchased from Aventis, France. Penicillin and streptomycin were obtained from Alborz Pharmacy, Karaj, Iran, and were stored at room temperature (25˚C) until testing. MTT powder [3-(4.5-dimethylthiazol-2-yl)-2.5-diphenyl tetrazolium bromide)], fetal calf serum (FCS), RPMI-1640 medium with L-glutamine, and Tween 20 were prepared from Sigma-Aldrich (St. Louis, Missouri, USA).
Parasite and cell culture
Standard strain of L. tropica (MHOM/IR/2002/Mash2) was kindly prepared from Center for Research and Training in Skin Diseases and Leprosy (Tehran, Iran). The parasites were cultured in NNN medium and subcultured in RPMI 1640 supplemented with penicillin (200 IU/ml), streptomycin (100 μg/ml) and 15% heat-inactivated fetal calf serum (FCS). Murine macrophage cells (J774-A1) were obtained from Pasteur Institute of Iran (Tehran, Iran). The cells were cultured and maintained in Dulbecco's modified eagle's medium (DMEM) supplemented with 10% FCS at 37˚C in 5% CO2.
Collection of plant materials
The leaves of M. communis were collected from rural regions of Baft district of Kerman province in September 2013. The identity was confirmed by the botanist at the Botany Department of Shahid Bahonar University, Kerman, Iran. A voucher specimen of the plant materials was deposited at the Herbarium of Department of Pharmacognosy of School of Pharmacy, Kerman University of Medical Science, Kerman, Iran (KF1356).
Preparing methanolic extract
The dried aerial parts of the plant (100 g) were extracted by percolation method with methanol (80%) successively for 72 hr in room temperature. The extracts were passed through filter paper (Whatman No. 3, Sigma, Germany) to remove plant debris. Finally, they were concentrated in vacuum at 50˚C using a rotary evaporator (Heidolph, Germany) and stored at -20˚C until use.
Phytochemical analysis of the methanolic extract
The preliminary phytochemical analysis of the M. communis methanolic extract was carried out to determined the presence of tannins, saponins, alkaloids, phenols, and glycosides as described elsewhere [11].
Isolation of the essential oil
Air-dried plant materials (200 g) were subjected to hydro-distillation for 3 hr using an all-glass clevenger-type apparatus. The essential oil obtained was dried over anhydrous sodium sulfate and stored in darkness at 4˚C in air-tight glass vials closed under nitrogen gas until testing.
Gas chromatography/mass spectrometry (GC/MS) analysis of essential oil
GC analysis: GC analysis was carried out by a Hewlett-Packard 6890 (Hewlett-Packard, Palo Alto, California, USA) with a HP-5MS column (30m×0.25 mm, film thickness 0.25 mm). The column temperature was maintained at 55˚C for 3 min and programmed to 280˚C at a rate of 15˚C per min, and kept constant at 280˚C for 5 min. Injector and interface temperatures were 220˚C and 260˚C, respectively. The flow rate of helium as carrier gas was 1 ml/min C.F. The percentages were calculated by electronic integration of FID peak areas without the use of response factors correction. Linear retention indices for all components were determined by coinjection of the samples with a solution containing homologous series of C8–C24 n-alkanes.
GC/MS analysis: GC/MS analysis was performed using a Thermoquest-Finnigan gas chromatograph equipped with fused silica capillary DB-5 column (30 m×0.25 mm, film thickness 0.25 mm) coupled with a TRACE mass (Manchester, UK). Helium was used as carrier gas with ionization voltage of 70 eV. Ion source and interface temperatures were 220˚C and 280˚C, respectively. Mass range was from 30 to 450 unit. Oven temperature program was the same as given above for the GC.
Identification of the essential oil components
The components of the essential oil were identified by comparison of their relative retention time and mass spectra with those of standards Wiley 2001 library data of the GC-MS system or with those reported in the literature data [12].
Drug dilutions
For the preparation of dilutions of the essential oil of M. communis, 0.1 ml of the essential oil was dissolved in 9.7 ml of normal saline. In addition, to enhance the dispersal of the essential oil in normal saline, 0.3 ml of Tween 20 (Sigma-Aldrich) was added to the test tube. The resulting solution was mixed adequately by a magnetic stirrer. Serial dilution was then made to obtain the essential oil at 3.125, 6.25, 12.5, 25, 50, and 100 μg/ml concentrations. To prepare the dilutions of methanolic extract, 10 mg of methanolic extract was dissolved in 10 ml normal saline. Then, serial dilution was made to obtain the essential oil at 3.125, 6.25, 12.5, 25, 50, and 100 μg/ml concentrations. The selection of dilutions of the essential oil was based on initial experiments, which also revealed that normal saline plus Tween 20 (3%) had no inhibitory effect on the growth of the parasite.
Anti-promastigote assay
To evaluate antipromastigote effects of essential oil and methanolic extract on promastigotes of L. tropica, colorimetric cell viability MTT assay was used as described elsewhere [13]. Briefly, 100 μl of promastigotes (106 cells/ml) harvested from logarithmic growth phase were added to a 96-well tissue culture plate. Then, 100 μl of various concentrations of essential oil (3.125-50 μg/ml) and methanolic extract (0-100 μg/ml) was added to each well and incubated at 25˚C±1˚C for 48 hr. After incubation, 10 μl of MTT solution (5 mg/ml) was added to each well and incubated at 25˚C for 4 hr. Then, cold isopropanol was added as a solvent for formazan crystals to produce purple color. The absorbance was measured for each well at 490 nm using an ELISA reader (BioTek-ELX800, Winooski, Vermont, USA). Promastigotes were cultured in complete medium with no drug used as positive control and complete medium with no promastigotes and drugs as blank. The absorbance was measured for each well at 490 nm using the ELISA reader. Fifty % inhibitory concentrations (IC50 values) were measured for all the tested extracts by Probit test in SPSS software.
Anti-amastigote assay
This was carried out according to the method described by Mahmoudvand et al. [14], Initially, before adding the macrophages to the plates, 1 cm2 cover slips were placed in the wells of 6-chamber slides (Lab-Tek, Nalge Nunc International, New York, USA). In the next step, 200 μl of macrophage cells (105 cells/ml) were incubated at 37˚C in 5% CO2 for 2 hr. Then, 200 μl (106 cells/ml) promastigotes in stationary phase were added to murine macrophages, so that the proportion of Leishmania : macrophage was 10:1 and incubated again in a similar condition for 24 hr. Free parasites were removed by washing with RPMI 1640 medium and the infected macrophages were treated with 50 μl of various concentrations of (0-50 μg/ml) essential oil and methanolic extract at 37˚C in 5% CO2 for 48 hr. At the end, the dried slides were fixed with methanol, stained by Giemsa, and studied under a light microscope. Also, the macrophages containing amastigotes without extract and those with no parasite and extract were considered positive and negative controls, respectively. Activity of anti-intramacrophage amastigotes of the extracts was evaluated by counting the number of amastigotes in each macrophage by examining 100 macrophages (% amastigotes viability) in comparison with those obtained with positive control. In addition, 50% inhibitory concentrations (IC50 values) were measured for all the tested extracts by Probit test in SPSS software.
Cytotoxic effects
In this investigation, cytotoxic effects of M. communis against J774-A1 cells were assessed by cultivating macrophages (5×105) with various concentrations of essential oil (0 to 100 μg/ml) and methanolic extract (0 to 500 μg/ml) in 96-well tissue culture plates at 37˚C in 5% CO2 for 48 hr. Cell viability was determined by colorimetric MTT assay, and the results were displayed as percentage of dead cells compared to macrophages treated with MA and non-treated macrophages (100% of viability). Moreover, CC50 (cytotoxic concentration for 50% of cells) was calculated by Probit test in SPSS software [3].
Statistical analysis
All the tests in this study were carried out in triplicate. Selectivity index (SI), calculated based on the equation of CC50 for murine macrophage cells/IC50 for amastigote forms of L. tropica, was used to compare toxicity and activity of essential oil and methanolic extract of M. communis as described by Weninger et al. [15]. Data analysis was carried out by using SPSS statistical package version 17.0 (SPSS Inc., Chicago, Illinois, USA). Differences between test and control groups were analyzed by t-test. In addition, P<0.05 was considered statistically significant.
RESULTS
GC/MS analysis of essential oil
Table 1 indicates the results obtained by GC/MS analysis of M. communis essential oil (as the most effective extract). Twenty-five compounds were identified, representing 93.0% of the total oil. The main components were α-pinene (24.7%), 1,8-cineole (19.6%), and linalool (12.6%).
Phytochemical analysis of the methanolic extract
Evaluation of the primary phytochemical screening of the M. communis methanolic extract revealed the presence of high amount of terpenoid, flavonoids, tannins, phenols, and glycosides and lacking the alkaloids in this plant.
Antileishmanial effects
Anti-promastigote assay: In this study, to investigate the anti-promastigote activity, promastigotes of both species were incubated in the presence of various concentrations of essential oil and methanolic extract and cell viability was determined after 72 hr using MTT assay. The findings demonstrated that essential oil and methanolic extract of M. communis had potent antileishmanial activity against the promastigote forms based on a dose-dependent response (P<0.05). These results also revealed that essential oil in comparison with methanolic extract and MA had significantly (P<0.05) higher leishmanicidal effect on the promastigotes of L. tropica once it exhibited lower IC50 value for the tested promastigotes. The IC50 values for the essential oil and methanolic extract were 8.4 μg/ml and 28.9 μg/ml against promastigotes of L. tropica, respectively, whereas this value was 88.3 μg/ml for MA as control drug.
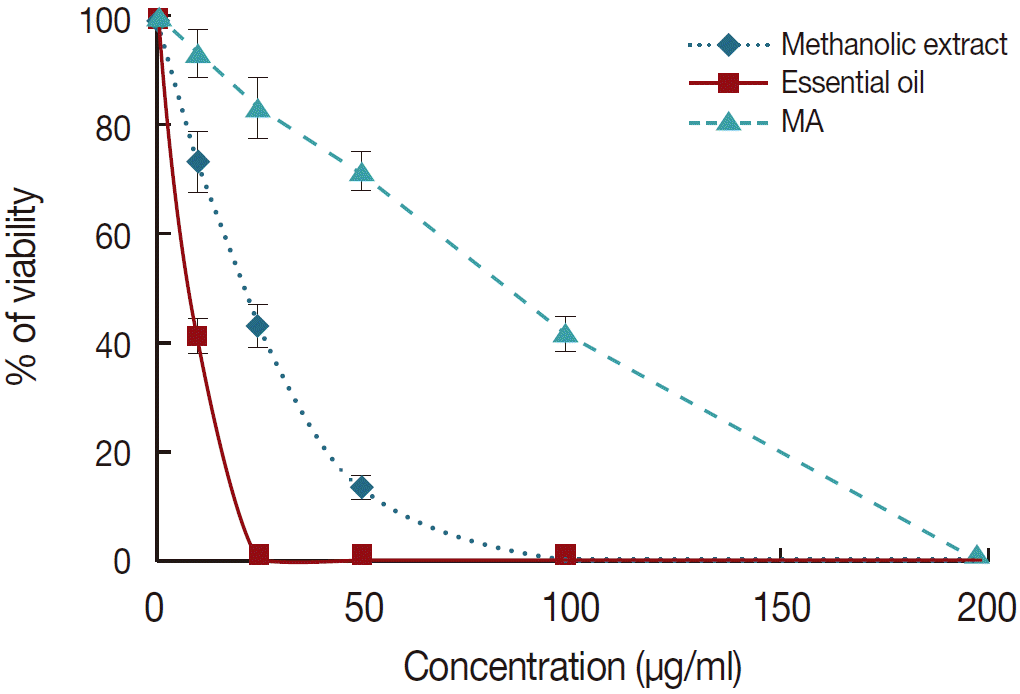
Anti-amastigote assay: The results showed that essential oil and methanolic extract of M. communis significantly (P<0.05) inhibited the growth rate of intramacrophage amastigotes as a dose-dependent manner (Fig. 1). Similar to anti-promastigote assay, the essential oil was more effective on the amastigote forms than the methanolic extract and MA once it exhibited the lower IC50 value for amastigote forms. The IC50 value for the essential oil (11.6 μg/ml) against promastigotes of L. tropica was significantly (P<0.05) lower than those of MA (44.6 μg/ml) and methanolic extract (40.8 μg/ml).
Cytotoxic effects
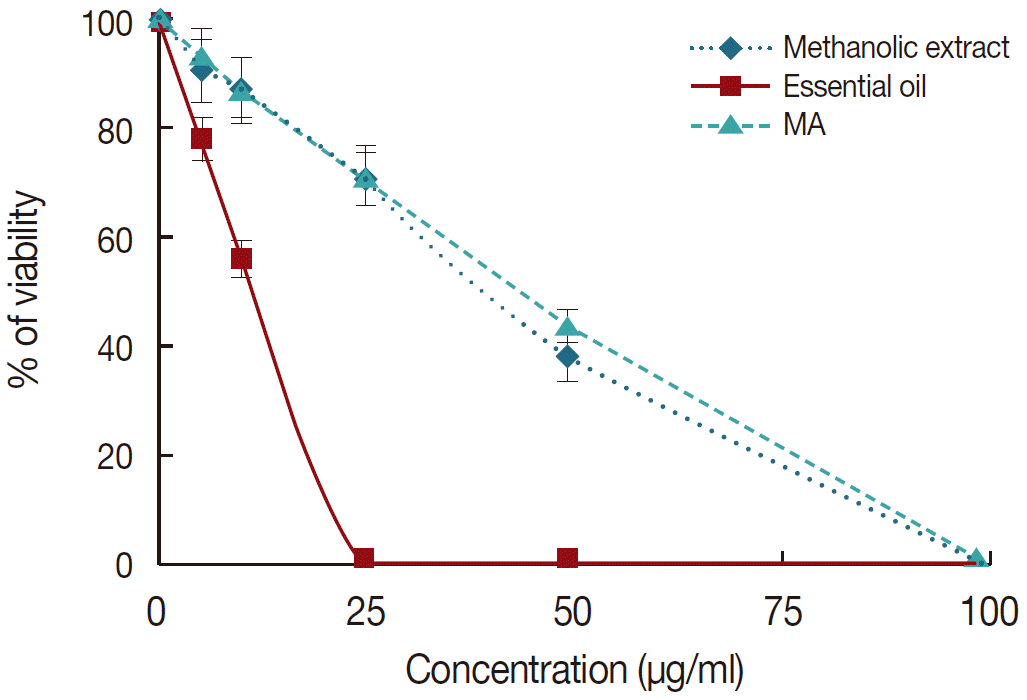
Cytotoxic effects of M. communis were determined in J774-A1 cells using MTT assay. The obtained results indicated no significant cytotoxicity in J774-A1 cells. However, essential oil had a higher cytotoxic effect on cells as compared with the methanolic extract of M. communis (Fig. 2). The CC50 values for essential oil and methanolic extract and also their SI values for amastigote forms of L. tropica were shown in Table 2.
DISCUSSION
Natural products, such as plants extract, either as pure compounds or as standardized extracts, provide unlimited opportunities for new and selective drug discoveries because of the unmatched availability of chemical diversity [16]. According to the World Health Organization (WHO), more than 80% of the world's population relies on traditional medicine for their primary healthcare needs. The use of herbal medicines in Asia represents a long history of human interactions with the environment. Plants used for traditional medicine contain a wide range of substances that can be used to treat various diseases such as infectious diseases [5]. In the present investigation, we evaluated antileishmanial activity of methanolic extract and essential oil of M. communis and their cytotoxic activities against J774-A1 cells on an in vitro model.
Our findings demonstrated that M. communis, particularly its essential oil, significantly (P<0.05) inhibited the growth rate of promastigote and amastigote forms of L. tropica based on a dose-dependent response. These results also displayed that amastigotes were more susceptible to M. communis than promastigotes. This difference in susceptibility might be related to structural, biochemical, and morphological features as previously shown elsewhere [17].
Previously, it has been proven that the activity of plant extracts could be influenced by the nature of the plant material or its origin as well as the climatic conditions in which plant grow, the plant part used, or the solvent used for extraction, because plants have different constituents depending on those factors [18]. The chemical composition of M. communis essential oil has widely been investigated; its composition is quite variable depending on the geographic region of production, the season of harvest, and the length of distillation [8,10]. However, in most regions, terpenoid compounds (1,8-cineole, α-pinene, myrtenyl acetate, limonene, linalool, and α-terpinolene) are the major constituents found in the essential oil obtained from the leaves of M. communis. In the present study, in line with other study, we found that the main components of M. communis essential oil are α-pinene, 1,8-cineole, and linalool. The phytochemical screenings of the M. communis methanolic extract showed the presence of terpenoid, flavonoids, tannins, and phenols [6]. So far, individual activities of these compounds have been demonstrated [19]. Moreover, in several investigations, potent antibacterial, antifungal, and antiparasitic activities of these compounds and their derivatives such as α-pinene, 1,8-cineole, limonen, thymol, and carvacrole against some pathogenic strains have been proven [20-25]. Therefore, phytoconstituents in this plant could be responsible for their antileishmanial activity whereas their exact mechanism of action is unclear. However, in the case of antimicrobial mechanism of some terpenoid compounds such as monoterpens, Sikkema et al. [26] revealed that they diffuse into pathogens and damage cell memberane structures. On the other hand, other reports suggested that the antimicrobial activity is related to ability of terpenes to affect not only permeability but also other functions of cell membranes; these compounds might cross the cell membranes, thus penetrating into the interior of the cell and interacting with critical intracellular sites [27,28]. Our findings also exhibited that SI of greater than 10 for essential oil and methanolic extract of M. communis represent their safety to the mammalian cells and specificity to the parasite. In agreement with these results, no health hazards or side effects are reported as a result of the proper administration of designated therapeutic dosages of M. communis [6].
In conclusion, the results of this study demonstrated potent antileishmanial effects of M. communis that might be a natural source for production of new antileishmanial agents against CL. However, further clinical studies are required to evaluate exact biological activity of M. communis in animal models as well as volunteer human subjects as a new therapeutic agent.