Shimazu and Kino: Metagonimus yokogawai (Trematoda: Heterophyidae): From Discovery to Designation of a Neotype
Abstract
Metagonimus yokogawai (Katsurada, 1912) Katsurada, 1912 (Trematoda: Heterophyidae) is parasitic in the small intestine of mammals including man and birds in Far Eastern Russia, Korea, Japan, China, and Taiwan. In the present study, the metacercariae and adults of M. yokogawai were redescribed to designate a neotype of this fluke together with reviews of previous studies including study histories from the first discovery to now. We particularly, attempted to review the study histories and morphological descriptions of M. yokogawai for the species validity, and compared with the morphological characteristics and life cycles of the closely related species, Metagonimus takahashii and Metagonimus miyatai. Finally, we proposed a differential key for the 8 known Metagonimus species distributed in East Asia. Metacercariae were obtained from the body muscles of sweetfish (Plecoglossus altivelis) collected in the Asahi River at Takebe-cho, Kita-ku, Okayama City, Okayama Prefecture, Japan. Adults were recovered from the small intestine of Syrian golden hamsters, to which the metacercariae had been fed 14 days before. A neotype was selected out of the present adult specimens. The Asahi River at Takebo-cho became the type locality of M. yokogawai. In conclusion, the present review shows that M. yokogawai, M. takahashii, and M. miyatai are valid and discriminated by means of morphological characteristics.
Key words: Metagonimus yokogawai, metacercaria, adult, morphology, review, neotype
INTRODUCTION
Katsurada [ 1] proposed a new species, Heterophyes yokogawai, on the basis of 5 egg samples from Taiwan and Japan. Soon after that, Katsurada [ 2] established a new genus, Metagonimus (Trematoda: Heterophyidae), to accommodate H. yokogawai, or now Metagonimus yokogawai (Katsurada, 1912) Katsurada, 1912, as the type species. This fluke occurs in Far Eastern Russia, Korea, Japan, China, and Taiwan. Metacercariae of the fluke are found encysted mainly in sweetfish Plecoglossus altivelis (Plecoglossidae). The fluke is one of the foodborne intestinal parasites of people there, where they willingly eat sweetfish and some other freshwater fishes raw or undercooked [ 3, 4]. Many papers including 5 reviews [ 3- 7] have been published on M. yokogawai since Katsurada’s [ 1] description of H. yokogawai in various points of view in endemic areas especially in Japan and Korea. Most of them, especially those of taxonomic importance in an earlier stage of the study, were written in Japanese. Our preliminary review from them has shown the following. Katsurada’s [ 1] proposal of the new species H. yokogawai only on the shape and size of eggs caused much confusion in subsequent studies of the species. The adult morphology of M. yokogawai came to be clarified [ 2, 8- 23]; however, still it needs to be described in more detail. Two closely related species, Metagonimus takahashii Suzuki, 1930 [ 24] and Metagonimus miyatai Saito, Chai, Kim, Lee, and Rim, 1997 [ 23], were added to the genus in Japan and Korea, but these 2 species seem to have previously been often confused with M. yokogawai. We consider that a review should be made here of the history of studies, morphology, life cycle, type specimens, and synonyms of M. yokogawai in order to supplement the previous reviews and correct some mistakes in them. Moreover, it is thought that all the original specimens of H. yokogawai [ 1] and its synonyms Loxotrema ovatum Kobayashi, 1912 [ 9] and Yokogawa yokogawai Leiper, 1913 [ 17] were lost, so that a neotype should be designated for H. yokogawai, or now M. yokogawai, to prevent further confusion between Metagonimus and other closely related genera and between M. yokogawai and other congeneric species. Our purposes in this paper are to describe the morphology of metacercarial and adult forms of M. yokogawai on the basis of our own specimens, review the history of studies, morphology, life cycle, type specimens, and synonyms of M. yokogawai, designate a neotype for the taxon, and finally present a key to 8 known species of Metagonimus in East Asia.
MATERIALS AND METHODS
Collection of metacercariae
Metacercariae were obtained from 15 sweetfish fished by a local fisherman in the Asahi River at Takebe-cho, Kita-ku, Okayama City, Okayama Prefecture, Japan, on 2 and 21 September 2013. Their trunk skeletal muscles (body muscles in this paper) were minced and digested in artificial gastric juice (0.1% pepsin 1:10,000 in 0.7% HCl solution) at 37˚C for 6 hr. The digest was filtrated with a nylon screen with an 80-µm mesh (Nylon screen F-3100-120, Mesh #203, Flon Industry, Tokyo, Japan) to remove the digestive fluid according to the method of Kino [ 25]. The residue on the screen was transferred into normal saline (0.9% NaCl solution), and metacercariae were collected under a binocular stereomicroscope. The metacercariae collected were kept in normal saline in a refrigerator until subsequent uses.
Experimental infection to obtain adult worms
The Syrian golden hamster, Mesocricetus auratus, was used as an experimental final host. Metacercariae were fed to 12 female golden hamsters of 5 weeks (Slc: Syrian, Japan SLC, Inc., Shizuoka, Japan) with a stomach tube. Adults were recovered from the small intestine of them at 14 days after feeding. A part of the adults recovered was used for the present study. The golden hamsters were handled and maintained according to the guidelines provided in the Guide for the Care and Use of Laboratory Animals of Hamamatsu University School of Medicine, Shizuoka, Japan.
As described below, metacercarial cysts were either elliptical or spherical. In one of the feeding experiments, metacercariae with elliptical and spherical cysts were fed to respective golden hamsters to confirm whether both metacercariae belonged to a single species.
Observations on morphological characteristics of metacercariae and adults
Live metacercarial cysts (2 September 2013) were used for taking measurements and photomicrographs. Metacercariae were excysted with slight pressure of a cover slip. Some excysted living worms were used for observation of the excretory system. Others were killed in hot normal saline, fixed in AFA (20 parts 70% alcohol, 1 part commercial formalin, and 1 part glacial acetic acid), stained with Heidenhain’s iron hematoxylin, dehydrated in a graded ethanol series, cleared in xylol, and mounted in Canada balsam (NSMT-Pl 5898, 2 September 2013).
Adult worms were treated as follows: (1) worms were slightly flattened under a cover slip, fixed in 10% neutral buffered formalin, and stained with Heidenhain’s iron hematoxylin (metacercariae, 2 September 2013; adults, NSMT-Pl 5899, 24-26 September 2013); (2) worms originated from the spherical and elliptical metacercarial cysts were fixed in hot 10% neutral buffered formalin and re-fixed in AFA and stained with Heidenhain’s iron hematoxylin (metacercariae, 2 and 21 September 2013; adults, NSMT-Pl 5900 and 5901, respectively, 17 October 2013); and (3) worms were fixed in hot 70% ethanol and stained with Grenacher’s alum carmine (metacercariae, 2 and 21 September 2013; adults, NSMT-Pl 5902, 3 December 2013). These stained worms were dehydrated, cleared, and mounted in Canada balsam.
Measurements (length by width) are given in micrometers. Drawings were made with the aid of a camera lucida and supplemented with free-hand details from living worms. Representatives of the specimens studied have been deposited in the National Museum of Nature and Science (NMNS) (collection name code NSMT-Pl), Tsukuba, Ibaraki Prefecture, Japan.
RESULTS
Class Trematoda Rudolphi, 1808
Family Heterophyidae Leiper, 1909
Genus Metagonimus Katsurada, 1912
Metagonimus yokogawai (Katsurada, 1912) Katsurada, 1912 ( Figs. 1- 9)
[Syn. Heterophyes yokogawai Katsurada, 1912; Heterophyes yokogawai Yokogawa, 1912; Loxotrema ovatum Kobayashi, 1912; Yokogawa yokogawai (Katsurada, 1912) Leiper, 1913; Loxotrema yokogawai (Katsurada, 1912) Kobayashi, 1925]
Metacercariae
Description: Based on living metacercariae obtained on 2 September 2013. Cysts elliptical ( Fig. 1A), 162-217 by 101-134 (17 measured) or spherical ( Fig. 1B), 139-160 by 130-145 (18 measured). Cyst wall transparent, thin, consisting of 2 layers: outer layer thicker, 2-3 µm thick; inner layer much thinner. Based on whole-mounted excysted metacercarial worms (NSMT-Pl 5898); 10 measured ( Fig. 2). Body ovate, slightly tapering posteriorly, 282-336 by 132-151; forebody 141-188 long, occupying 49-57% of total body length. Tegument spinose. Eyespot pigment dispersed in forebody. Brown body pigmentation light around intestinal ceca. Transverse nerve commissure slightly posterior to pharynx, dorsal to esophagus. Oral sucker subventral, 47-85 by 78-100. Large gland cells present, 3 on either side of pharynx, opening into lumen of oral sucker through dorsal wall of oral sucker (not illustrated). Prepharynx present, short. Pharynx elliptical to globular, 56-69 by 41-63. Esophagus 63-110 long, bifurcating at about junction of anterior and middle thirds of body. Intestinal ceca ending blindly near posterior extremity of body, separate there, including many pale disks about 8 in diameter by about 3 thick (not illustrated). Ventrogenital complex consisting of ventrogenital sac and ventral sucker, immediately medial to right intestinal cecum, slightly posterior to midlevel of body. Ventrogenital sac weakly differentiated, not clearly observed. Ventral sucker 47-60 by 44-53; sucker width ratio 1: 0.47-0.57, sucker length ratio 1: 0.48-0.57. Testes 2, slightly diagonal, slightly anterior to cecal ends, dorsal to intestinal ceca and excretory vesicle; anterior or left testis 37-53 by 28-41; posterior or right testis 44-60 by 25-47. Ovary almost globular, 31-53 by 31-50, median, pretesticular, located at about junction of middle and posterior thirds of hindbody, held by 2 arms of excretory vesicle. Uterus pretesticular. Excretory vesicle Y-shaped, extending anteriorly to ovary, filled with fine granules; main collecting canals connected subterminally to respective arms of excretory vesicle, ciliated in posteriormost parts (not illustrated); flame cell formula 2[(3+3+3)+(3+3+3)]=36; excretory pore posteroterminal.
Adults
Most of the adults recovered were fully mature, but a few were small and still immature (about 100 long). The fully mature adults (NSMT-Pl 5900-5901) were very similar in morphology and measurements regardless of the 2 different shapes of metacercarial cysts. They resembled the metacercariae described above in general morphology.
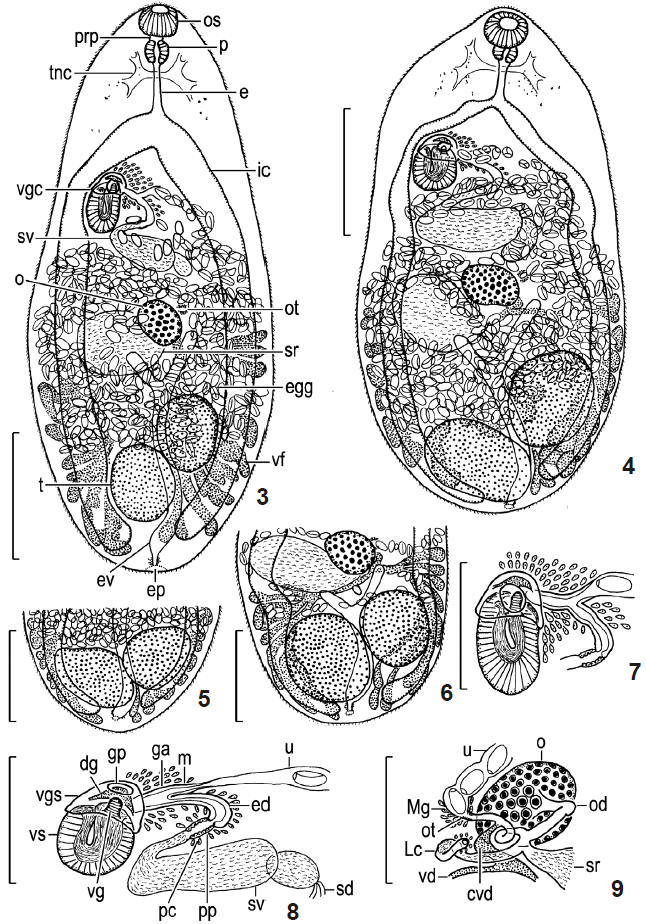
Description: 1) Based on hot formalin-fixed specimens (NSMT-Pl 5900-5901); 10 measured ( Figs. 3 and 7). Body ovate, with tapering anterior extremity and round posterior extremity, very small, 753-1,074 by 314-424; forebody 220-345 long, occupying 28-39% of body length. Tegumental spines ctenoidscaliform in forebody and spinous in hindbody. Oral sucker elliptical, subventral, 48-59 by 59-65. Pharynx elliptical, 35-48 by 36-59. Esophagus 63-119 long. Intestinal ceca ventral, ending blindly at midlevel of post-testicular region of body. Ventrogenital complex lying obliquely (axis inclined anterosinistrally), immediately medial to right intestinal cecum, slightly posterior to bifurcal level, at about junction of anterior and middle thirds of body. Ventral sucker elliptical, large, 71-92 by 48-57, including nuclei and muscle fibers, lined with thick layer of longitudinal muscle, embedded slightly obliquely in parenchyma, protruding into ventrogenial sac, opening at anterior tip; sucker width ratio 1: 0.77-0.95, sucker length ratio 1:1.36-1.93. Ventrogenital sac thick-walled, dome- or cup-like, large, anterosinistral to ventral sucker. Gonotyls 2, on internal wall of ventrogenital sac; ventral gonotyl cylindrical, tall, muscular, 11-20 by 9-17, projecting from ventrosinistral base of ventrogenital sac between ventral sucker and ventrogenital sac, sometimes closing genital pore; dorsal gonotyl crescent, short but wide, fleshy, 16-27 by 8-20, lying transversely at about midlevel of ventrogenital sac. Genital pore opening on ventral wall of ventrogenital sac. Testes globular or elliptical, diagonal, some distance anterior to posterior extremity of body; anterior or left testis 111-151 by 68-135, posterior or right testis 95-175 by 55-127. Sperm duct long; common sperm duct absent. Seminal vesicle bipartite, large, 111-175 by 35-71, posterior to level of ventral sucker; anterior (or distal) portion retortshaped, much larger than elliptical posterior (or proximal) portion. Cirrus and cirrus pouch absent. Pars prostatica small, following neck of anterior portion of seminal vesicle; prostatic cells poorly developed. Ejaculatory duct short, joining to metraterm to form genital atrium. Genital atrium tubular, short, like hermaphroditic duct, opening into ventrogenital sac through its left wall between dorsal gonotyl and ventral sucker. Small gland cells present around ejaculatory duct, metraterm and genital atrium and anteriorly to ventrogenital sac. Ovary usually globular, rarely slightly indented irregularly, small, 68-103 by 48-79, median, between seminal vesicle and testes. Oviduct short, dorsal to ovary, often including sperm. Seminal receptacle retort-shaped, voluminous, 111-175 by 95-159, dorsal, posterodextral to ovary, pretesticular. Laurer’s canal canalicular, short, opening dorsally; proximal half part thicker than distal half part, including sperm. Ootype small, usually sinistorolateral to ovary; Mehlis’ gland weakly developed. Uterus occupying all available space between intestinal bifurcation and testes, extending posteriorly usually to anterior border of posterior or right testis, overlapping anterior or left testis dorsally, sometimes to anterior border of both testes, never embracing anterior testis; metraterm short. Eggs numerous, almost elliptical, only slightly tapering at operculate pole, brawn, 25-28 by 14-16, operculate, with small knob on anoperculate pole, fully embryonated in distal part of uterus. Vitellaria follicular, dorsal; follicles clavate, sometimes branched, distributed between level of ovary and post-testicular region of body, separate posteriorly. Excretory vesicle ventral, Y-shaped, left arm reaching to level of ovary, right arm reaching to seminal receptacle; excretory pore usually posterodorsal. 2) Based on slightly flattened specimens (NSMT-Pl 5899); 10 measured ( Figs. 4- 6, 8, 9). Body oval to elliptical, 684-958 by 330-471; forebody 188-314 long, occupying 27-35% of body length. Oral sucker 36-66 by 57-76. Pharynx 33-48 by 35-44. Esophagus 40-73 long. Intestinal ceca extending into post-testicular region of body. Ventral sucker 60-95 by 55-75; sucker width ratio 1: 0.92-1.11, sucker length ratio 1:1.22-2.00. Ventral gonotyl 11-20 by 9-17, dorsal gonotyl 20-23 by 9-16. Testes shifted posteriorly, leaving short post-testicular region of body; anterior testis 127-182 by 103-143, posterior testis 135-222 by 95-159. Seminal vesicle 175-206 by 64-113. Ovary 75-148 by 60-92. Seminal receptacle 127-206 by 87-143. Uterus usually overlapping anterior testis dorsally or rarely pretesticular, never embracing anterior testis. Eggs 25-28 by 14-16. Vitelline follicles extending usually into short post-testicular region of body ( Figs. 5, 6), sometimes not ( Fig. 4), separate there.
Remarks
We used metacercariae obtained from the body muscles of sweetfish collected in the Asahi River, because (1) the egg samples nos. 3-5 [ 1] (original specimens of Heterophyes yokogawai) were obtained from the stools of 3 men, who perhaps had eaten sweetfish from the nearby river, one of the most famous fishing spots of sweetfish then, (2) metacercariae of M. yokogawai and Loxotrema ovatum were found in sweetfish from the river [ 2, 9- 16, 26], and (3) metacercariae of M. yokogawai are found encysted mainly in the body muscles of sweetfish [ 23, 27]. In the present metacercarial and adult specimens, the ventrogenital complex was present, located immediately medial to the right intestinal cecum at about the junction of the anterior and middle thirds of the body ( Figs. 2- 4). The oral sucker was larger than the ventral sucker in the metacercariae ( Fig. 2) but smaller in adults ( Figs. 3, 4). The uterus extended posterior to the anterior border of the 2 testes in the metacercariae ( Fig. 2). In the adults, it extended to the anterior border of the posterior testis and usually overlapped the anterior testis dorsally ( Figs. 3, 4) or rarely extended to the anterior border of the anterior testis ( Figs. 5, 6) but never embraced the anterior testis. Uterine eggs were fully embryonated and 25-28 by 14-16 µm. The flame cell formula was 2[(3+3+3)+(3+3+3)]=36 in the metacercariae ( Fig. 2). The intestinal ceca and vitelline follicles entered the short post-testicular region of the body and were separate there in the adults ( Figs. 3- 6). The uterus reached posteriorly to the anterior border of the 2 testes [ 23]. However, it overlapped the anterior testis dorsally in adult specimens, the metacercariae of which were found in sweetfish from the Tenryu River in 2008 and the Oochise River in 2009 both in Shizuoka Prefecture, Japan [Kino, unpublished data]. The eggs were 25.0-30.5 by 14.1-18.2 µm [ 23]. The flame cell formula in the cercaria-metacercaria-adult generation was 2[(3+3+3)+(3+3+3)]=36 [ 22]. The intestinal ceca and vitelline follicles entered the short post-testicular region of the body and were separate there in adults [ 16, 17, 23]. Consequently, we identified the present metacercarial and adult specimens as M. yokogawai according to the characteristics of this species as discussed above and below. The present adult specimens were very similar in morphology and measurements regardless of the 2 different shapes of metacercarial cysts, which indicates that all of them belong to M. yokogawai. Of 152 whole-mounted adults (NSMT-Pl 5899-5902), 150 and 2 were identified as M. yokogawai and M. miyatai, respectively.
The uterus usually overlapped the anterior testis dorsally in the present adult specimens. This feature was seen in both slightly flattened and hot formalin and ethanol-fixed specimens. The dorsal overlap was also observed in the above-mentioned Kino’s adult specimens. Although nobody has previously mentioned the dorsal overlap, we consider that it is also a normal state in fully mature adult specimens, not an artifact due to flattening of specimens before fixation.
DISCUSSION
With regard to the history of studies, Yokogawa [ 28] at Taiwan Sotoku-fu Igakko in Taihoku (Taipei), Taiwan, first found elliptical (see [ 29]) metacercariae encysted in the gills of sweetfish in Taiwan (locality not specified) on 1 December 1911. He fed them to a dog and detected eggs in the stool of the dog on 10 January 1912. He then recovered adults from the upper jejunum of the dog about 40 days after feeding (see [ 1]). He briefly described measurements of 2 adult specimens and eggs in the stool of the dog (?), mentioning that Katsurada had named this fluke Heterophyes yokogawai [sic, should be yokogawai [ 1]]. He also showed that 2 men, who used to eat sweetfish raw or undercooked, were infected with the fluke in Taiwan, detecting eggs of the fluke in their stools. Katsurada [ 1] at Okayama Igaku Senmon Gakko (Okayama Medical Collage), Okayama, Japan, proposed a new species, Heterophyes yokogawaii, on the basis of the shape and size of 5 egg samples (nos. 1-5 in the text table on p. 378). The papers by Yokogawa [ 28] and Katsurada [ 1] were published on the same day (31 May 1912). Since Katsurada named the parasite with the description of the eggs, we also cite the author and date of the taxon as Katsurada, 1912 as usual. Ito [ 3] neither cited Katsurada’s [ 1] original description nor mentioned his controversial proposition of the new species only on the shape and size of eggs. Yokogawa [ 29] suggested that the adult of H. yokogawai was distinct from those of then known species of Heterophyes Cobbold, 1866 in the sizes and relative positions of the oral and ventral suckers and the genital pore. He also found spherical cysts of metacercariae of H. yokogawai in the body muscles of sweetfish in Taiwan. Katsurada [ 26] also found metacercariae of H. yokogawai in the scales, skin, subcutaneous tissue, body muscles, and caudal fin of sweetfish from the Asahi River in Okayama Prefecture, Japan. He fed 5 sweetfish from this river to a dog and subsequently recovered adults from the small intestine of the dog 8 days after feeding. According to Yokogawa’s suggestion [ 29] and his own observations, Katsurada [ 2] established a new genus, Metagonimus, with H. yokogawai, or now M. yokogawai (Katsurada, 1912) Katsurada, 1912, as the type species, distinguishing it from Heterophyes by that the “Bauchsaugnapf” (ventral sucker in this paper) and “Genitalnapf” (ventrogenital sac in this paper) were combined to form the “Genitalnapf-Bauchsaugnapfapparat” (ventrogenital complex in this paper), which was located immediately medial to the right intestinal cecum, instead of median, at the junction of the anterior and middle thirds of the body or slightly posterior to it (see also Katsurada [ 8]). He described some of the 8-day-old adult specimens (figs. 1, 2). In addition, he included an adult (fig. 3) found in a woman in Okayama (the egg samples nos. 9-10 [ 1]) in M. yokogawai. Furthermore, he stated that metacercariae of M. yokogawai were found not only in sweetfish but also in freshwater cyprinid fishes of several species. On the other hand, Kobayashi [ 9] found metacercariae which were similar to those of H. yokogawai under the scales and in the body muscles, gills, and fins of sweetfish from the Asahi River (see [ 24]), not Lake Biwa as mentioned by Ito [ 3]. He fed them to a dog and subsequently recovered adults from the small intestine of the dog 49 days after feeding. He described the metacercarial and adult specimens and erected a new genus and species, Loxotrema ovatum, on the basis of the adult specimens (see also Kobayashi [ 10]). Yokogawa [ 11- 16] also fed metacercariae of M. yokogawai found in the scales, fins, body muscles, and gills of sweetfish from the Asahi River (see [ 24]) to mice to study the development of worms in them during his stay in Okayama. He described the metacercariae from the Asahi River and the adult specimens of about 40 days [ 28] (?) in the dog in Taiwan and the mice in Okayama. Leiper [ 17] at the London School of Tropical Medicine, London, UK, created a new genus, Yokogawa, with H. yokogawai, or now Yokogawa yokogawai (Katsurada, 1912) Leiper, 1913, as the type species on the basis of adult specimens of about 40 days in the dog [ 28] (?) of H. yokogawa [sic, should be yokogawai [ 1]] received from Yokogawa in Taiwan. Because both Metagonimus and Yokogawa have H. yokogawai as the type species, these 2 generic names are objective synonyms in accordance with Article 61.3.3 of the International Code of Zoological Nomenclature, ICZN [ 30]. Since Metagonimus is a senior synonym, it is valid (Article 23.3, ICZN).
Loxotrema ovatum [ 9, 10] is very similar to M. yokogawai [ 2] in adult morphology, shape and size of eggs, and species and geographical distribution of the second intermediate host (sweetfish from the Asahi River). Agreeing with Katsurada [ 8] and Leiper [ 31] that L. ovatum and M. yokogawai were identical, Kobayashi [ 32] changed the species name of the taxon to Loxotrema yokogawai (Katsurada, 1912) Kobayashi, 1925, because the establishment of Loxotrema on 10 October 1912 [ 9] antedated that of Metagonimus on 31 October 1912 [ 2]. However, Loxotrema Kobayashi, 1912 was preoccupied by Loxotrema Gabb, 1868 (a molluscan genus) [ 33, 34]. The rejected junior homonym Loxotrema Kobayashi, 1912 has 2 junior synonyms, Metagonimus Katsurada, 1912 and Yokogawa Leiper, 1913. Since the former is a senior synonym, Metagonimus is valid as mentioned above.
Takahashi [ 35] studied the life cycles of a new species of Metagonimus (large egg type of Metagonimus) and M. yokogawai (see below) in the field (Okayama Prefecture, Japan) and laboratory and described developmental stages of these 2 species. He first used the species name Metagonimus takahashii given by Suzuki for the former species. Since Suzuki merely named the former species M. takahashii, he was not responsible at all for the work by Takahashi [ 35]. Therefore, we do not consider that Suzuki is the author of the species name. Metagonimus takahashii Takahashi, 1929 is available in this case (Article 50.1, ICZN). Suzuki [ 24] cited Takahashi’s description for the former species and named it “ Metagonimus takahashii nov. spec.” This species name is also available (Article 12.1, ICZN). Although M. takahashii Takahashi, 1929 has the priority over M. takahashii Suzuki, 1930 (Article 23, ICZN), we here follow Saito et al. [ 23] who used the latter. Saito [ 21, 22] compared M. yokogawai with M. takahashii in morphology of the cercariae of natural infection and metacercariae and adults of experimental infection and in infectivity of their cercariae to second intermediate hosts, sweetfish and goldfish ( Carassius carassius auratus) (Cyprinidae), in Hiroshima, Japan. Saito [ 22] concluded that the 2 species were valid and sibling species of each other. Saito’s [ 21] cercaria (fig. 1A, photo. 1) and adult (photo. 9) refer to M. yokogawai. Saito [ 22] recovered metacercariae from sweetfish exposed to the cercariae identified as M. yokogawai: 48.1% of them from the body muscles and 39.3% from the scales. In this exposure experiment, cercariae of M. miyatai today [ 23] (see below) should have been mixed in the cercariae of M. yokogawai used, because metacercariae of M. yokogawai and those of M. miyatai are found encysted mainly in the body muscles and under (or in) the scales, respectively [ 23, 27]. Neither Saito [ 22] nor Saito et al. [ 23] paid attention to this unexpected, taxonomically important finding that the metacercariae of M. miyatai today were also experimentally recovered from the scales of sweetfish. Miyata [ 18, 19] pointed out that some specimens identified as M. yokogawai had the uterus extended posteriorly as in M. takahashii: to the anterior border of the posterior testis and posteriorly to the anterior testis to embrace it. Saito [ 20] referred to them as “Miyata type.” Later, Saito et al. [ 23] described “Miyata type” as a new species, Metagonimus miyatai, on the basis of adult specimens. They recovered them from the small intestine of dogs, to which they had fed metacercariae found under the scales of sweetfish, dace ( Tribolodon hakonensis and T. taczanowskii), pale chub ( Zacco platypus), dark chub ( Z. temminckii), and common fat-minnow ( Moroco steindachneri) (Cyprinidae) from Japan and Korea 14-17 days before. Saito et al. [ 23] and Chai et al. [ 4] summarized the history of studies of M. miyatai in Korea and Japan. Shimazu [ 36] studied the life cycle of M. miyatai in the field (Nagano Prefecture, Japan) and laboratory and demonstrated that Takahashi’s [ 35] M. yokogawai was actually M. miyatai. Katsurada [ 2, 26], Kobayashi [ 9, 10], and Yokogawa [ 11- 16] found metacercariae in various sites of sweetfish from the Asahi River. It is possible that they should have described the metacercariae and adults of M. yokogawai on the basis of metacercariae and adults of M. miyatai as well as M. yokogawai. A barely mature adult specimen of 5 days in a mouse in Yokogawa (fig. 8 [ 12], fig. 8 [ 13], fig. 8 [ 14], fig. 8 [ 15]) is, in fact, identified as M. miyatai because of the uterus embracing the anterior testis. Katsurada’s [ 2] adult specimen of M. yokogawai (fig. 3) found in the small intestine of the Japanese woman (the egg samples nos. 9, 10) is regarded as M. miyatai [ 23]. With regard to the morphology, Metagonimus in the Heterophyidae [ 37] is characterized by the presence of the ventrogenital complex, or a combination of the ventrogenital sac and ventral sucker, which is located immediately medial to the right intestinal cecum at about the junction of the anterior and middle thirds of the body, in the adult stage [2,37, this paper]. The ventral sucker is weakly formed in a median position in the cercarial stage. As metacercariae develop, the ventral sucker moves to the strongly dextral position and then develops fully there [ 35, 36, 38, 39]. The anatomy of the ventrogenital complex has been described [12-16,36,38-40, this paper]. Pearson [ 37] was incorrect in his description (fig. 5.18) that the ventral gonotyl is located at the center of the ventral wall of the ventrogenital sac and the dorsal gonotyl is orbicular and large in M. takahashii. Adults of M. yokogawai have been described by Yokogawa [ 28, 29] in Taiwan, Leiper [ 17], Saito et al. [ 23] in Japan and Korea, and in the present paper. The figures by Yokogawa (fig. 9 [ 12], fig. 9 [ 13], fig. 9 [ 15], fig. 1 [ 16]) and Leiper (fig. 31 [ 17]) clearly show gross morphology of 2 fully matured adult specimens probably of about 40 days in the dog [ 28] from Taiwan. The uterus extends posteriorly to the anterior border of the 2 testes in these adult specimens. Katsurada (fig. 2 [ 2]), Yamaguti (fig. 7 [ 41]), Saito (fig. 9 [ 21]), and Saito et al. (fig. 4 [ 23]) described similar adult specimens from Japan. In the present adult specimens, the uterus usually overlapped the anterior testis dorsally but never embraced it ( Figs. 3, 4). In M. takahashii and M. miyatai, on the other hand, the uterus extends posteriorly to the anterior border of the posterior testis (sometimes farther on the lateral side of it) and always posteriorly to the anterior testis to embrace it [ 18, 19, 23, 35, 36] as seen in the metacercariae of M. miyatai (fig. 9 [ 23]). Katsurada [ 1] included 5 egg samples in H. yokogawai. It is certain that the egg sample no. 1 (28.0 by 16.2 µm (mean), found in the stool of the dog [ 28]), and no. 2 (28.9 by 16.2 µm (mean), dissected out of an adult [adults (?)] recovered from the same dog [ 28]) belong to H. yokogawai. On the other hand, it is uncertain that the egg sample no. 3 (av. 28.8 by 16.9 µm, fig. 1), no. 4 (av. 28.4 by 16.5 µm), and no. 5 (av. 29.1 by 16.3 µm) are really assigned to H. yokogawai. They were found in the stools of 3 men in Okayama, Japan. The 3 men used to eat sweetfish raw or undercooked [ 1]. Sweetfish is usually infected with metacercariae of M. yokogawai (mainly in the body muscles) and M. miyatai (mainly under or in the scales) in at least Japan and Korea [ 23, 27]. M. miyatai is also infective to man [ 23]. People usually eat the whole body of sweetfish including the scales. The 3 men may have been infected with both M. yokogawai and M. miyatai at that time, and these 2 species can hardly be separated by egg size alone [ 23]. It is unknown at present which species the egg figured by Katsurada (fig. 1 egg sample no. 3 [ 1] and fig. 4 [ 2]) belongs to H. yokogawai or M. miyatai. Katsurada’s [ 1] egg samples nos. 6-8 (larger, 30.6-35.3 by 18.5-23.1 µm (mean), found in the stools of 3 other Japanese) are identified as M. takahashii [ 35]. The egg samples nos. 9-10 have remained unidentified. Eggs of M. yokogawai are 28 by 15.8 µm [ 28], 28.0 by 16.2 µm (mean) and 28.9 by 16.2 µm (mean) [ 1], 25.0-30.5 by 14.1-18.2 µm [ 23], and 25-28 by 14-16 µm in the present adult specimens. Eggs of M. miyatai are 26.3-32.3 by 14.1-19.2 µm [ 23] and 28-32 by 16-18 µm [ 36], largely overlapping those of M. yokogawai in size [ 23]. Eggs of M. takahashii are 30-36 by 18-23 µm [ 35], 30-35 by 18-23 µm [ 41], and 28.8-36.1 by 17.8-22.3 µm [ 23], evidently larger than those of M. yokogawai. With regard to the life cycle, the entire life cycle of M. yokogawai is known. Natural first intermediate hosts are snails of Semisulcospira (Pleuroceridae): S. libertina, S. coreana, and S. reiniana [ 4, 22, 39]. Cercariae of the pleurolophocercous type are produced in daughter rediae. It has been said that Muto [ 42] first definitively elucidated the entire life cycle of M. yokogawai [ 3]. His cercariae found in S. libertina in Japan were capable of encysting in goldfish ( Carassius auratus) and carp ( Cyprinus carpio) (Cyprinidae). In his adults recovered from cats, to which metacercariae found in the experimental fish had been fed, eggs were 30.0-33.0 by 19.5-21.0 µm in the stools of the cats (table 10) and 20.0-20.8 by 12.8-13.6 µm in formalin-fixed adults (table 11). We agree with Takahashi [ 35] that the adult specimens with larger eggs belong to M. takahashii (see also [ 19]). We cannot identify the other adult specimens with much smaller eggs at present. It thus remains undetermined who was the first to discover the cercaria of M. yokogawai. Incidentally, the 3 shells in the figure 1 [ 42] from the Seta River refer to Semisulcospira reiniana (left and middle) and Semisulcospira habei yamaguchii (right) [Keiji Matsuoka, 13 April 1979, personal communication]. The oral spines of the first (or posterior) row in the cercaria are 4 in M. yokogawai and M. miyatai [ 22, 36] and 5 in M. takahashii [ 22, 35]. The penetration glands in the cercaria are globular and small in M. yokogawai and M. miyatai [ 22, 36] and square and large in M. takahashii [ 22]. The flame cell formula in the cercaria-metacercaria-adult generation is 2[(3+3+3)+(3+3+ 3)]=36 in M. yokogawai and M. takahashii [ 22, 39] and 2[(2+2+ 2)+(2+2+2)]=24, not 2[(3+3+3)+(3+3+3)]=36 as determined by Saito et al. [ 23], in M. miyatai [ 36]. The cercaria in Ito (fig. 74 [ 5]) is that of M. miyatai after Takahashi [ 35]. Likewise, the daughter redia, cercaria, metacercaria, and adult in Yamaguti (figs. 1162, 1163 [ 43]) are those of M. miyatai after Takahashi [ 35]. Besprozvannykh et al. [ 44] and Besprozvannykh [ 45] studied the life cycle of M. yokogawai in Primorsky Region, Russia. They found cercariae in Juga spp. (Gastropoda: Pachychilidae). Sporocysts were branched and large. The daughter redia has a small intestine. The cercaria has the flame cell formula of 2[(3+3+3)+(3+3+3)]=36. We doubt that the adult specimen (fig. 2A [not 1A]) belongs to M. yokogawai [ 44] because the uterus embraces the anterior testis and the excretory vesicle is nearly V-shaped and small. Metacercariae of M. yokogawai have been recorded from freshwater fish: sweetfish (gills and body muscles), dace ( Tribolodon hakonensis and T. ezoe) (Cyprinidae) (body muscles, epidermis, and scales), and perch ( Lateolabrax japonicus) (Lateolabracidae) [4,23,27-29, this paper]. Adults of M. yokogawai have been reported from the small intestine of final hosts: mammals (man, dog, cat, and rat), in addition to experimental animals, and a bird (kite) [ 4, 36]. With regard to the synonyms, besides Loxotrema [ 6] and Yokogawa [ 17], Loossia Ciurea, 1915, Dexiogonimus Witenberg, 1929, Metagonimoides Price, 1931 and Loxotremuna Strand, 1942 are considered to be synonymous with Metagonimus [ 37]. As discussed above, H. yokogawai, L. ovatum, L. yokogawai, and Y. yokogawai are synonyms of M. yokogawai. Metagonimus ovatus Yokogawa, 1913 from Taiwan [ 46, 47] and Loossia romanica Ciurea, 1915, Loossia parva Ciurea, 1915, and Loossia dobrogiensis Ciurea, 1915 from Rumania [ 48] have been synonymized with M. yokogawai [ 49]. Yokogawa [ 46, 47] found metacercariae from carp ( Cyprinus carpio), Funa ( Carassius auratus (?)), and Haya ( Zacco platypus (?)) (sites of infection not indicated) in Taiwan and obtained adults of M. ovatus by feeding experiment (experimental final host not indicated). M. ovatus [ 46, 47] differs from M. yokogawai in having shorter intestinal ceca ending in the anterior half of the testicular zone, instead of entering the post-testicular region. Later, Ciurea [ 50, 51] synonymized Loossia with Metagonimus. However, we consider that Loossia differs from Metagonimus in possessing a large ligulate ventral gonotyl (Appendix [ 48]) at the edge of the genital pore. No more detailed description of the anatomy of the ventrogenital sac in Loossia is available to us. Witenberg [ 52] recognized M. yokogawai and M. romanicus (syn. M. dobrogiensis) as valid. With regard to the type specimens, we looked for the original specimens of H. yokogawai of Katsurada [ 1] and the adults, metacercariae, and cercariae of M. yokogawai of Yokogawa [ 11- 16, 28, 29], Katsurada [ 2, 8, 26], and Takahashi [ 35]. None of them were found in the collections of the Faculty of Medicine (formerly Okayama Igaku Senmon Gakko), Okayama University, Okayama (Toshiki Aji, 14 November 2012, personal communication); the Department of Parasitology, Faculty of Medicine (formerly Taiwan Sotoku-fu Igakko), Taiwan National University, Taipei, Taiwan (Chin-Hung Sun, 27 February 2013, personal communication); and the Department of Parasitology, Faculty of Medicine (for which Muneo Yokogawa, the son of Yokogawa, used to work), Chiba University, Chiba (our search, 28 February 2013). The original specimens of Loxotrema ovatum of Kobayashi [ 6] could not be traced at all. None of them were deposited in the Department of Zoology, The University Museum, The University of Tokyo, Tokyo, Japan [ 53]. The parasite collection of the London School of Tropical Medicine, London, UK, was transferred to the Commonwealth Institute of Parasitology. When this institute was closed in the 1990s, all the type specimens in the collection were transferred to The Natural History Museum, London, UK; and the rest, to the Royal Veterinary College, London (David I. Gibson, 14 March 2013, personal communication). None of the original specimens of Yokogawa yokogawai of Leiper [ 17] were found in the collection of either the Royal Veterinary College (Mark Fox, 15 April 2013, personal communication) or The Natural History Museum (Eileen Harris, Senior Curator, 8 March 2013, personal communication). Consequently, we consider that all the original specimens of H. yokogawai, L. ovatum, and Y. yokogawai and the closely related specimens studied by Yokogawa, Katsurada, and Takahashi were lost. Katsurada’s [ 1] egg samples nos. 1-2 of H. yokogawai were from Taiwan and nos. 3-5 were from Okayama. We obtained good whole-mounted adult specimens (NSMT-Pl 5899-5902) of M. yokogawai that had been originated from metacercariae found in the body muscles of sweetfish from the Asahi River in Okayama Prefecture. M. yokogawai is the type species of Metagonimus and one of the parasites of medical importance. We designate one of our adult specimens as a neotype for the nominal species M. yokogawai as follows to indicate adult morphology of M. yokogawai more clearly and prevent further confusion between Metagonimus and other closely related genera and between M. yokogawai and other congeneric species. Since the metacercaria of the neotype was found in sweetfish from the Asahi River, this river becomes the type locality (Articles 76.1.1 and 76.3, ICZN). Neotype: H. yokogawai Katsurada, 1912, or now M. yokogawai (Katsurada, 1912) Katsurada, 1912. Adult (NSMT-Pl 5900, Fig. 3A, 785 µm long by 345 µm wide), hot formalin-fixed, whole-mounted, ex small intestine of Syrian golden hamster, 14 days after infection, 17 October 2013. Type host: Syrian golden hamster (experimental).
Type locality: Asahi River at Takebe-cho, Kita-ku, Okayama City, Okayama Prefecture, Japan.
In conclusion, the present review shows that M. yokogawai, M. takahashii, and M. miyatai are valid and that M. yokogawai differs from M. takahashii mainly in the posterior extent of the uterus and egg size in the adult, in number of the oral spines and shape and size of the penetration glands in the cercaria and in encysting as metacercariae in the body muscles of sweetfish instead of in the scales of crucian carp; and from M. miyatai mainly in posterior extent of the uterus in the adult, in the flame cell formula in the cercaria-metacercaria-adult generation and in encysting as metacercariae in the body muscles of sweetfish instead of the scales of dace, pale and dark chubs, common fat-minnow, etc. and that the designation of the neotype of M. yokogawai, the type species of Metagonimus, is necessary to define adult morphology of M. yokogawai. It is desired that adult specimens from the Asahi River and Taiwan be morphologically and molecularly compared in the near future.
In Metagonimus in East Asia, 8 species have hitherto been described: M. yokogawai [this paper], M. ovatus [ 46, 47], M. takahashii [ 18- 24, 35], M. minutus Katsuta, 1932 [ 54], M. katsuradai Izumi, 1935 [ 55- 57], M. otsurui Saito and Shimizu [ 39, 58], M. miyatai [ 23, 36], and M. hakubaensis Shimazu, 1999 [ 38, 59]. The following is a key to the species of Metagonimus in East Asia.
A key to the species of Metagonimus in East Asia
1.1. Oral sucker smaller than ventral sucker …………....… 2 1.2. Oral sucker larger than ventral sucker …………....…..... 3 1.3. Oral sucker smaller or larger than or as large as ventral sucker; uterus pretesticular or rarely embracing anterior testis; intestinal ceca ending in testicular zoon; post-testicular region almost absent; eggs 28-34 by 14-20 µm …. ......................................................................... M. hakubaensis 3.2. Uterus extending posteriorly between 2 testes, not embracing anterior testis; intestinal ceca extending into post-testicular region; post-testicular region short; eggs 26-30 by 14-18 µm .......……............……….…. M. otsurui 2.2. Uterus embracing anterior testis …………………...….. 5 3.1. Uterus embracing anterior testis; intestinal ceca extending into post-testicular region; post-testicular region long; eggs 26-32 by 14-16 µm ….................. M. katsuradai 4.1. Uterus pretesticular; intestinal ceca extending into post-testicular region; post-testicular region very short; eggs 21-24 by 12-15 µm …........................................ M. minutus 4.2. Uterus pretesticular or overlapping anterior testis dorsally; intestinal ceca extending into post-testicular region; post-testicular region short; eggs 25-28 by 14-16 µm........ ............................................................................ M. yokogawai 5.1. Intestinal ceca extending into post-testicular region; post-testicular region short; eggs 30-36 by 18-23 µm ............................................................................................ M. takahashii 5.2. Intestinal ceca ending in posterior half of testicular zoon; post-testicular region almost absent; eggs 28-32 by 16-18 µm …...….…..............................................…. M. miyatai 5.3. Intestinal ceca ending in anterior half of testicular zoon; post-testicular region almost absent; eggs 26.4-29.7 by 14.4-16.8 µm ……………..........................…...... M. ovatus
ACKNOWLEDGEMENTS
We are grateful to Mr. Makoto Mizumoto (Okayama) for collecting sweetfish for the present study; Dr. Toshiki Aji (formerly Department of Immunology, Okayama University Graduate School of Medicine, Okayama), Prof. Chin-Hung Sun (Department of Parasitology, Faculty of Medicine, Taiwan National University, Taipei, Taiwan), Dr. Mark Fox (Royal Veterinary College, London), Ms. Eileen Harris and Dr. David I. Gibson (Department of Life Sciences, The Natural History Museum, London) for informing us about the specimens in the collections; Dr. Fumie Aosai (Department of Infection and Defense, Chiba University Graduate School of Medicine, Chiba then) for permitting us to looking for the specimens in the collection; Dr. Takashi Iwaki (Meguro Parasitological Museum, Tokyo) for sending copies of some of the references cited; Dr. Keiji Matsuoka (formerly Earth Science Department, School of Science, Nagoya University, Nagoya) for identifying Muto’s shells; and Prof. Jong-Yil Chai (Department of Parasitology and Tropical Medicine, Seoul National University College of Medicine, Seoul, Korea) and Dr. Tomáš Scholz (Institute of Parasitology, Biology Centre, Academy of Sciences of the Czech Republich, Branišovská, České Budějovice, Czech Republic) for reviewing the draft of this manuscript.
Conflict of interest
We have no conflict of interest related to this work.
REFERENCES
1. Katsurada F. Heterophyes in Japan. Okayama Igakkai Zasshi 1912;(268):373-380 (in Japanese).
2. Katsurada F. Heterophyes in Japan (Supplementary note 2). No. 1, Erection of a new genus allied to Heterophyes. No. 2, Significance of “Carassius” as the intermediate host for trematodes. Okayama Igakkai Zasshi 1912;(273):768-778 (in Japanese).
3. Ito J. Metagonimus and other human heterophyid trematodes. In Morishita K, Komiya Y, Matsubayashi H eds, Progress of Medical Parasitology in Japan. Meguro Parasitological Museum. Tokyo. 1964, 1:pp 315-393.
4. Chai JY, Shin EH, Lee SH, Rim HJ. Foodborne intestinal flukes in Southeast Asia. Korean J Parasitol 2009;47(suppl):S69-S102.  5. Ito J. A monograph of cercariae in Japan and adjacent territories. In K, Komiya Y, Matsubayashi H eds, Progress of Medical Parasitology in Japan. In Morishita K, Komiya Y, Matsubayashi H eds, Progress of Medical Parasitology in Japan. Meguro Parasitological Museum. Tokyo. 1964, 1:pp 395-550.
6. Komiya Y. Metacercarie in Japan and adjacent territories. In Morishita K, Komiya Y, Matsubayashi H eds, Progress of Medical Parasitology in Japan. Meguro Parasitological Museum. Tokyo. 1965, 2:pp 1-328.
7. Saito S. Metagonimus–Research done after 1960–. In Otsuru M, Kamegai S, Hayashi S eds, Progress of Medical Parasitology in Japan. Meguro Parasitological Museum. Tokyo. 2003, 8:pp 219-231.
8. Katsurada F. On a new trematode, Metagonimus. Tokyo Iji Shinshi 1912;(1796):3483-3489 (in Japanese).
9. Kobayashi H. On a new genus of trematodes (a preliminary report). Saikingaku Zasshi 1912;(204):780-786 (in Japanese).
10. Kobayashi H. On a new genus of trematodes (a preliminary report). Dobutsugaku Zasshi 1912;24:603-608 (in Japanese).
11. Yokogawa S. A new parasite using Plecoglossus altivelis as the intermediate host and erection of a new genus for it. Taiwan Igakkai Zasshi 1912;(122):1055-1068 (in Japanese).
12. Yokogawa S. A new parasite using Plecoglossus altivelis as the intermediate host and erection of a new genus for it (continued). Taiwan Igakkai Zasshi 1913;(123):1-26 (in Japanese).
13. Yokogawa S. A new parasite using Plecoglossus altivelis as the intermediate host and erection of a new genus for it. Okayama Igakkai Zasshi 1913;(279):255-266 (280): 337-358 (in Japanese).
14. Yokogawa S. A new trematode using Plecoglossus altivelis as the intermediate host, Metagonimus yokogawai. Tokyo Igakkai Zasshi 1913;7:685-709 1 fig. pl. (in Japanese).
15. Yokogawa S. Ueber einen neuen Parasiten Metagonimus Yokogawai [sic], der die Forellenart Plecoglossus altivelis (Temminck) [sic] zum Zwischenwirt hat. Bildung einer neuen Gattung. Centralb Bakteriol, 1. Abt, Orig 1913;72:158-179 fig. pl. 1-3.
16. Yokogawa S. Metagonimiasis yokogawai. Nisshin Igaku, Periodical extra issue 1922. 85-121 fig. pl. 1-2 (in Japanese).
17. Leiper RT. Observations on certain helminths of man. Trans Soc Trop Med Hyg (1912-13) 1913;6:265-297.  18. Miyata I. Supplements to the taxonomic knowledge of trematodes of the genus Metagonimus. Shokubutsu Oyobi Dobutsu 1941;9:533-534 (in Japanese).
19. Miyata I. A taxonomic study of trematodes of the genus Metagonimus. Dobutsugaku Zasshi 1944;56:16-19 (in Japanese).
20. Saito S. Taxonomic consideration on the flukes of the genus Metagonimus. Kiseichu Bunrui Keitai Danwakai Kaiho 1984;(2):1-4 (in Japanese).
21. Saito S. On the differences between Metagonimus yokogawai and Metagonimus takahashii. I. The morphological comparisons. Jpn J Parasitol 1972;21:449-458 (in Japanese with English abstract).
22. Saito S. On the differences between Metagonimus yokogawai and Metagonimus takahashii. II. The experimental infections to the second intermediate hosts. Jpn J Parasitol 1973;22:39-44 (in Japanese with English abstract).
23. Saito S, Chai JY, Kim KH, Lee SH, Rim HJ. Metagonimus miyatai sp. nov. (Digenea: Heterophyidae), a new intestinal trematode transmitted by freshwater fishes in Japan and Korea. Korean J Parasitol 1997;35:223-232.   24. Suzuki M. Metagonimus yokogawai. A new species of Metagonimus. Geographical distribution of two species of Metagonimus. History of studies of two species of Metagonimus. Bibliography on Metagonimus. In Okayama Prefecture ed, Peculiar Animals from Okayama Prefecture and a Bibliography of Researches on Them. Okayama. 1930, pp 146-168 (in Japanese).
25. Kino H. Filtration technique using nylon mesh in the digestion method with artificial gastric juice to collect parasite larvae in food materials. In Asakawa M ed, Materials and Methods in Parasitology 2013. Sankeisha, Nagoya, Japan. 2013, pp 139-141 (in Japanese with English abstract).
26. Katsurada F. Heterophyes in Japan (Supplementary note 1). Okayama Igakkai Zasshi 1912;(269):449-451 (in Japanese).
27. Kino H, Suzuki T, Oishi H, Suzuki S, Yamagiwa S, Ishiguro M. Geographical distribution of Metagonimus yokogawai and M. miyatai in Shizuoka Prefecture, Japan, and their site preferences in the sweetfish, Plecoglassus altivelis, and hamsters. Parasitol Int 2006;55:201-206.   28. Yokogawa S. Reports on visceral parasites on Taiwan. II. Taiwan Igakkai Zasshi 1912;(114):366-370 (in Japanese).
29. Yokogawa S. Supplementary note to a new species of Heterophyes using Plocoglossus altivelis as the intermediate host (a preliminary report). Tokyo Iji Shinshi 1912;(1776):1527-1529 (in Japanese).
30. International Commission on Zoological Nomenclature. International Code of Zoological Nomenclature. 4th ed. International Trust for Zoological Nomenclature. London, UK. 1999, p 306 306.
31. Leiper RT. Medical helminthology: a review. Part I. Trop Dis Bull 1922;19:361-365.
32. Kobayashi H. On the scientific name of Metagonimus yokogawai. Chosen Igakkai Zasshi 1925;(55):404 (in Japanese).
33. Faust EC, Nishigori M. The life cycles of two new species of Heterophyidae, parasitic in mammals and birds. J Parasitol 1926;13:91-128 fig. pl. 3-6.  34. Strand E. Miscellanea nomenclatoria zoologica et palaeontologica. X. Folia Zool Hydrobiol 1942;11:386-402.
35. Takahashi S. On the life-history of Metagonimus yokogawai, a new species of Metagonimus and Exorchis major. Okayama Igakkai Zasshi 1929;(479):2687-2755 (in Japanese).
36. Shimazu T. Life cycle and morphology of Metagonimus miyatai (Digenea: Heterophyidae) from Nagano, Japan. Parasitol Int 2002;51:271-280.   37. Pearson J. Family Heterophyidae Leiper, 1909. In Bray RA, Gibson DI, Jones A eds, Keys to the Trematoda. CAB International and The Natural History Museum. Wallingford. 2008, 3:pp 113-141.
38. Shimazu T. Metagonimus hakubaensis sp. n. (Digenea, Heterophyidae) from Nagano, Japan: morphology and life cycle. Bull Natl Sci Mus, Tokyo, Ser A (Zool) 1999;25:87-99.
39. Shimazu T, Urabe M. Morphology and life cycle of Metagonimus otsurui (Digenea, Heterophyidae) from Nara, Japan. Bull Natl Sci Mus, Tokyo, Ser A (Zool) 2002;28:21-28.
40. Suzuki M. On the ventrogenital complex in Metagonimus. Proceedings of the 5th General Meeting and Abstracts of the Japanese Society of Parasitology. 1933, pp. 69-70 (in Japanese).
41. Yamaguti S. Über die Ursache der sog. “schwarzen Winterflecke” der japanischen Süsswasserfische. Z Parasitenk 1938;10:691-693.  42. Muto M. On the first intermediate host of Metagonimus yokogawai. Kyoto Igakkai Zasshi 1917;14:114-133 (in Japanese).
43. Yamaguti S. A Synoptical Review of Life Histories of Digenetic Trematodes of Vertebrates with Special Reference to the Morphology of Their Larval Forms. Keigaku Publishing Co.. Tokyo. 1975, pp 1-590.
44. Besprozvannykh VV, Ermolenko AV, Dvoryadkin VA. On the discovery of Metagonimus katsuradai Isumi [sic], 1935 (Trematoda: Heterophyidae) in northern Primor’e. In Mamaev YuL ed, Helminths and Diseases Caused by Them. Akademiya Nauk SSSR, Dal’nevostochnyy Nauchnyy Tsentr. Biologo-Pochvennyy Institut. Vladivostok, Russia. 1987, pp 47-52 (in Russian).
45. Besprozvannykh VV. Fauna, Biology, and Ecology [of] Part[h]enitae and Cercariae [of] Trematodes in Mollusks of Genus Juga (Pachychilidae) from River [in] Primorye Territory. Dalnauka. Vladivostok, Russia. 2000, pp 1-120 (in Russian).
46. Yokogawa S. On a new species of the genus Metagonimus, Metagonimus ovatus n. sp. Tokyo Igakkai Zasshi 1913;27:1705-1709 (in Japanese).
47. Yokogawa S. Studies of visceral parasites on Taiwan (second report). Taiwan Igakkai Zasshi 1914;(335):1-25 (in Japanese).
48. Ciurea I. Über einige neue Distomen aus dem Darm unserer Haustiere und des Pelikans, für welche die Fische als Infektionsquelle zu betrachten sind. Z Infektionskr Haustiere 1915;16:445-458.
49. Ransom BH. Synopsis of the trematode family Heterophyidae with descriptions of a new genus and five new species. Proc US Natl Mus 1920;57:527-573.  50. Ciurea I. Heterophyidés de la faune parasitaire de Roumanie. Parasitology 1924;15:1-21.  51. Ciurea I. 1933. Les vers parasites de l’homme, des mammifères et des oiseaux provenant des poissons du Danube et de la Mer Noire. Premier Mémoire. Trématodes, famille Heterophyidae Odhner, avec un essai de classification des Trématodes de la superfamille Heterophyoidea Faust. Arch Roum Pathol Exp Microbiol 1933;6:5-134.
52. Witenberg G. Studies on the trematode-family Heterophyidae. Ann Trop Med Parasitol 1929;23:131-239.
53. Shimazu T, Araki J. A list of the helminth parasite specimens deposited in the Department of Zoology, the University Museum, the University of Tokyo. In Ueshima R ed, Catalogue of Invertebrate Collection Deposited in the Department of Zoology, The University Museum, The University of Tokyo. Tokyo, Japan. The University Museum, The University of Tokyo, Material Reports 2006;(62):151-161.
54. Katsuta I. Studies on trematodes whose second intermediate hosts are fishes from the brackish waters of Formosa (II Report). On a new trematode Metagonimus minutus of which the mullet is the second intermediate host. Taiwan Igakkai Zasshi 1932;31:26-39 (in Japanese).
55. Izumi M. Studies of a new species of trematode of the genus Metagonimus and its life cycle. Tokyo Iji Shinshi 1935;(2929):1224-1236 (in Japanese).
56. Izumi M. Studies concerning a new species of Metagonimus and its life cycle. Kitasato Arch Exp Med 1935;12:362-384 (in Japanese).
57. Shimazu T. Morphology of metacercariae and adults of Metagonimus katsuradai Izumi (Digenea, Heterophyidae) from Shiga, Japan. Bull Natl Sci Mus, Tokyo, Ser A (Zool) 2003;29:47-51.
58. Saito S, Shimizu T. A new trematode, Metagonimus otsurui sp. nov. from the fresh-water fishes (Trematoda: Heterophyidae). Jpn J Parasitol 1968;17:167-174.
59. Shimazu T. Redescription of the adult of Metagonimus hakubaensis (Digenea: Heterophyidae) from Nagano, Japan. J Nagano Pref Coll 2001;(56):79-82.
Fig. 1.
Metagonimus yokogawai metacercariae found in the body muscles of sweetfish from the Asahi River, Okayama, Japan. (A) Elliptical cyst. (B) Spherical cyst. Scale bar=100 μm. 
Fig. 2.
Metagonimus yokogawai, excysted metacercarial worm (NSMT-Pl 5898), heat-killed, whole-mounted, entire body, ventral view, and excretory organs on left side of body added from free-hand sketches. Abbreviations as in Figs. 3-9. Scale bar=100 μm. 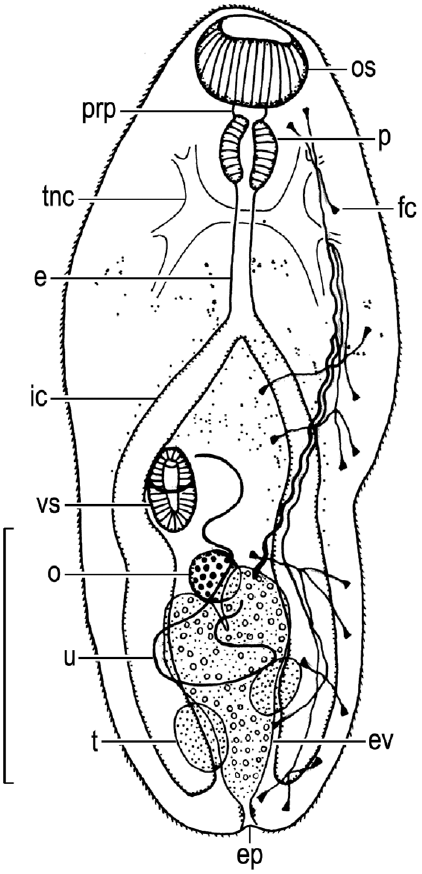
Fig. 3-9.
Metagonimus yokogawai, adults experimentally recovered from the small intestine of Syrian golden hamsters 14 days after infection, whole-mounted. (3) Neotype (NSMT-Pl 5900), fully mature, hot formalin-fixed, entire body, and ventral view. (4) Adult (NSMT-Pl 5899), fully mature, slightly flattened, entire body, and ventral view. (5) Adult (NSMT-Pl 5899), fully mature, slightly flattened, posterior part of body, showing pretesticular uterus, and ventral view. (6) Adult (NSMT-Pl 5899), barely mature, slightly flattened, posterior part of body, showing pretesticular uterus and vitelline follicles, ventral view. (7) Neotype, terminal genitalia, prostatic cells not clearly observed, and ventral view. (8) Adult (NSMT-Pl 5899), fully mature, slightly flattened, terminal genitalia and, ventral view. (9) Adult (NSMT-Pl 5899), fully mature, slightly flattened, ovarian complex, and dorsal view. Scale bars=200 μm in Figs. 3- 6; 100 μm in Figs. 7- 9.
Abbreviations: cvd, common vitelline duct; dg, dorsal gonotyl; e, esophagus; ed, ejaculatory duct; egg, eggs; ep, excretory pore; ev, excretory vesicle; fc, flame cell; ga, genital atrium; gp, genital pore; ic, intestinal cecum; Lc, Laurer’s canal; m, metraterm; Mg, Mehlis’ gland; o, ovary; od, oviduct; os, oral sucker; ot, ootype; p, pharynx; pc, prostatic cells; pp, pars prostatica; prp, prepharynx; sd, sperm duct; sr, seminal receptacle; sv, seminal vesicle; t, testis; tnc, transverse nerve commissure; u, uterus; vd, vitelline duct; vf, vitelline follicles; vg, ventral gonotyl; vgc, ventrogenital complex; vgs, ventrogenital sac; vs, ventral sucker.
|
|









