INTRODUCTION
Toxoplasma gondii, the causative pathogen of toxoplasmosis, is an intracellular parasite belonging to the phylum Apicomplexa together with Plasmodium, Neospora, and Theileria. Apicomplexan parasites have 3 distinctive secretory organelles in their cytoplasm, named as the microneme, rhoptry, and dense granule (reviewed by Mercier et al., 2005; Tonkin et al., 2006). Proteins of these organelles are secreted cascadally (Ngo et al., 2000) and/or sometimes concertedly (Ahn et al., 2005) to function in the entry into host cells, in the formation of parasitophorous vacuole (PV), and in the host-parasite interactions across the PV membrane (PVM).
Among these, the rhoptry proteins are major secreted proteins to the plasmodial PVM formed in the erythrocytes by the infection with Plasmodium spp. They interact to host components in the PVM and some are secreted to the cytosol across the PVM to reach the surface membrane via the tubulovesicular membrane network (TVN) in the relatively simple anucleated host cells with few subcellular organelles (Templeton and Deitsch, 2005; Tilley et al., 2007). There is only a little information on the involvement of dense granular proteins in the parasitism of Plasmodium. On the while, T. gondii infects almost all nucleated host cells, in which the parasite encounters a great deal of complex and various interactions with host cell components and subcellular organelles across the toxoplasmal PVM. In the PV and PVM, many dense granular proteins are found to be secreted to decorate the TVN of PV and PVM in addition to rhoptry proteins. These GRA proteins are suggested to be the key proteins in the maintenance of relationship between nucleated host cells and intracellular parasites, such as interactions with the cytoplasmic components and the recruitment of the host endoplasmic reticulum and mitochondria (Magno et al., 2005). In the dense granule, 10 GRA proteins have been identified in T. gondii tachyzoites. Still the function of each GRA protein is not known, and the molecular information is restricted to cDNA and deduced amino acid sequences and the localization within the PV during the growth of the parasite.
In the previous study, the yeast two-hybrid technique using GRA proteins as baits was applied to profile the interaction of host proteins to each GRA protein (Ahn et al., 2006). GRA proteins interacted with a number of host cell proteins, such as enzymes, structural and functional organellar proteins with a certain specific pattern to each GRA protein. Among them, GRA10 showed a peculiar binding pattern to those proteins related with nuclear and nucleolar involvements, such as signal transducer and activator of transcription 6 (STAT6), TATA-binding protein (TBP)-associated factor 1B (TAF1B), and Ran-binding protein 1 (RanBP1), whereas the other GRA proteins interacted approximately with cytoplasmic proteins and cytosolic organellar proteins. Here, we tried expression of GRA10 in host cells directly to confirm the translocalization of the protein into the nucleolus and the specific interaction with a nucleolar protein, TAF1B, which involves in the synthesis of rRNA.
MATERIALS AND METHODS
Parasite and host cells
The RH strain of T. gondii was maintained by peritoneal passages in BALB/c mice. Prior to use, the tachyzoites were purified by centrifugation over 40% Percoll (Amersham Pharmacia Biotech, Uppsala, Sweden) in PBS solution. HeLa (ATCC CCL-2) cells were cultured in MEM supplemented with 10% FBS and used as host cells.
Expression of GFP-GRA proteins in HeLa cells
The GRA cDNAs downstream of signal sequence to terminal stop sequence was amplified by PCR to insert into pEGFP-C2 plasmid (Clontech, Palo Alto, California, USA). For the GRA3, 5'-gcg gca agc ttg cct gaa aat cat ca-3' and 5'-cca gga tcc gtc aac gaa tgt ttc ag-3' were used for HindIII/BamHI insertion, for the GRA5, 5'-cgt gaa gct tca aaa tgg cgt ctg-3' and 5'-cga gga tcc cag tgc ccc ttg ct-3' for HindIII/BamHI insertion, and for the GRA10, 5'-gca gaa ttc att gag gcc gct gtg gag-3' and 5'-ctg ggt acc tca gac agg cgt ttc-3' were used for EcoRI/KpnI insertion.
Transient transfection of HeLa cells was achieved using the calcium phosphate co-precipitation method (Hoeck and Woisetschlager, 2001). The day before transfection, 5 x 104 cells were seeded into 24-well culture plates in fresh medium. The plasmid DNA (1-2 µg) was diluted in 42 µl of H2O, mixed with 7 µl of 2 M CaCl2 and added by drops to 50 µl of 2 x HeBS (280 mM NaCl, 1.5 mM Na2HPO4, and 50 mM HEPES, pH 7.05). After incubation for 20 min at room temperature, the mixture was added to the cells. The cells were incubated further for 24 hr and fixed either with cold absolute methanol for 5 min or with 3% formaldehyde for 10 min and then permeabilized by 0.05% (v/v) Triton X-100 for 5 min. Mouse anti-GRA10 antibody (mAb Tg378), mouse anti-nucleophosmin (B23) antibody (Chemicon, Temacula, California, USA), and mouse anti-nucleolin (C23) antibody (Santa Cruz Biotechnology, Santa Cruz, California, USA) were diluted 1: 200 in 3% BSA/PBS, and the FITC- or TRITC-conjugated anti-mouse IgG antibodies (Sigma Chem Co., St. Louis, Missouri, USA) were used at 1:100 dilutions. The fluorescence was observed under a fluorescence microscopy (Axiophot, Carl Zeiss, Oberkochen, Germany).
Construction of GRA10 fragments
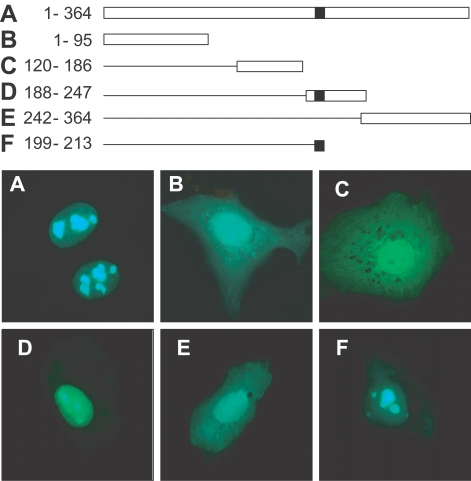
GRA10 was fragmented according to the scheme in Fig. 3. A: full GRA10 from 1 to 364 amino acids cloned with cDNA amplified by PCR with 5'-gca gaa ttc att gag gcc gct gtg gag-3' and 5'-ctg ggt acc tca gac agg cgt ttc-3' primers; B: GRA10 fragment from 1 to 95 amino acids by 5'-gca gaa ttc att gag gcc gct gtg gag-3' and 5'-acc ggt acc tcc tgg caag aga cgc cc-3'; C: fragment from 120 to 186 amino acids by 5'-ctg gaa ttc tct ctc ggt gac gag agg aag-3' and 5'-cat ggt acc ctt ctg cag ccg tcg-3'; D: fragment 188 to 247 amino acids by 5'-ctc tgc gaa ttc aca cag tct cc acct g-3' and 5'-ccc ggt acc ctc tct ctc cgc ttc ctc-3'; E: fragment from 242 to 364 amino acids by 5'-gag gaa ttc gga acg ggg tgc atg agt-3' and 5'-ctg ggt acc tca gac agg cgt ttc-3' for EcoRI/KpnI insertion; and H: fragment from 199 to 213 amino acids by 5'-tcc gct agc gct acc ggt cgc cac-3' and 5'-tga gct cga gct gcg ctt tcc ccg ttt ctt ttt gcc tga acg gcg tct ctt ctt cct ccc ggc ggc ggt cac gaa ctc-3' for NheI/XhoI insertion. Amplified DNA fragments were inserted into pEGFP-C2 plasmid for the transfection.
GST pull-down assay and immunoprecipitation
GST pull-down assay was performed according to the method of Fan et al. (2004). Two µg of GST or GST-fused TAF1B were mixed with 40 µl of 50% suspension of glutathione-Sepharose 4B beads for 2 hr in a binding buffer (25 mM HEPES-NaOH, pH 7.5, 12.5 mM MgCl2, 10% glycerol, 5 mM DTT, 0.1% NP-40, 150 mM KCl, and 20 µM ZnCl2). Herein His-tagged GRA10 lysate was added and followed by incubation for another 2 hr. The pellets were washed extensively and boiled. The bound proteins were resolved in 12% SDS-PAGE and blotted by mAb Tg378.
Immunoprecipitation was followed the method of Saharinen et al. (2000). RH tachyzoites of T. gondii were lysed with the RIPA buffer (10 mM Tris-HCl, pH 8.0, 140 mM NaCl, 0.025% NaN3, 1% Triton X-100, 0.1% SDS, and 1% deoxycholic acid) containing protease inhibitors and phosphatase inhibitors. RH lysates were then sonicated and insoluble materials were removed by 2 centrifugations, the first for 5 min and the second for 15 min at 4℃ at 13,000 g and the supernatants were transferred to new tubes. Two mg extracts were precleared by incubating them with 30 µl of packed protein A agarose beads. Immunoprecipitation was performed by rocking for 4 hr at 4℃ the precleared extracts with His-tagged TAF1B and mAb Tg378, and then by precipitating the immunocomplexes with protein A agarose. The immunoprecipitates were washed once with 1 ml RIPA buffer. Then, the beads were washed twice with 1 ml TBS and centrifuged at 7,500 g for 2 min. The beads were then suspended in SDS sample buffer containing 0.125 M DTT and boiled for 5 min. The samples were analyzed by 10% SDS-PAGE followed by immunoblotting using anti-His antibody.
Co-localization of GRA10 and TAF1B
GRA10 cDNA was amplified with 5'-cgc gaa ttc ttg agg ccg ctg tgg ag-3' and 5'-ggc ggt acc aca ggc gtt tcc cc-3' to insert into pDSRed2-N1 (BD Biosciences Clontech, Mountain View, California, USA) by EcoRI/KpnI digestion for GRA10-Red fusion protein. TAF1B cDNA was amplified with 5'-ggg agc tcg atg gac ctc gag gag-3' and 5'-ttt gga tcc tgt cgt ctc act ttc ttg gat c-3' to insert into pEGFP-N1 by SacI/BamHI digestion for TAF1B-GFP fusion protein. The 2 plasmids were co-transfected into HeLa cells, and the expressed fusion proteins were observed under a fluorescence microscopy. Distribution of the fluorescence was observed after the treatment of low dose (0.04 µg/ml) and high dose (0.5 µg/ml) of actinomycin D (Sigma) as an inhibitor of RNA polymerase I and 1 to 40 µg/ml of α-amanitin as a RNA polymerase II (Dundr et al., 1995).
RESULTS
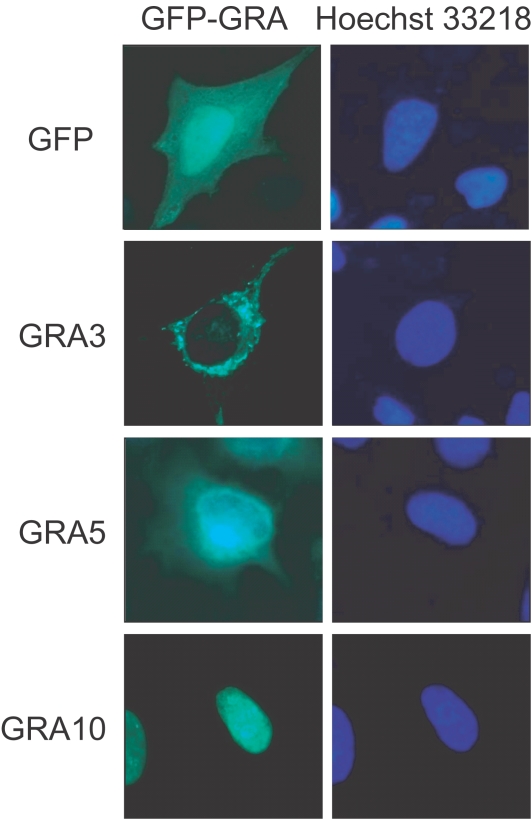
All GFP-GRA fusion proteins were well expressed in HeLa cells to be detectable under a fluorescence microscope 24 hr after transfection. GFP-fused GRA3, GRA5 and GRA10, which have 1 or 2 putative transmembrane domains within the amino acid sequences and are inserted into PVM after secretion from the intracellular T. gondii, showed different fluorescent images compared to that of GFP only as demonstrated in Fig. 1 (other GRA images not shown). Fluorescences of GRA3 and GRA5 were distributed in the cytoplasm of the transfected cells, such that GRA3 was restricted to the shape of cytoplasmic organellar structure and GRA5 was dispersed in the perinuclear region of the cytoplasm. However, GRA10 showed a different image, in which the fluorescence was accumulated clearly in the nucleolus within the nucleus without any smudges in the cytoplasm.
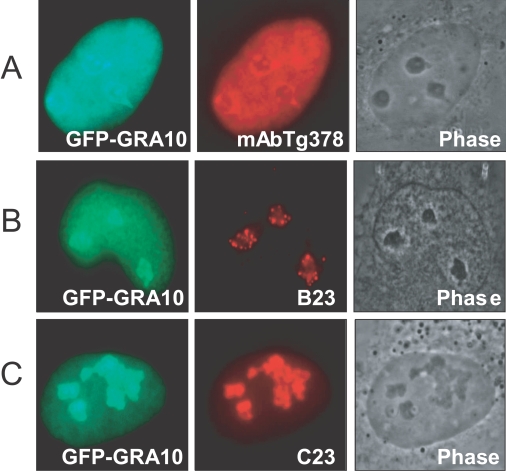
GRA10 was concentrated specifically in the dense fibrillar component (DFC) of the nucleolus morphologically both in the GFP-GRA10 transfection image and in the immunofluorescence image with a monoclonal antibody Tg378 against GRA10 (Fig. 2A). Fluorescence was not detected in the fibrillar center (FC). The localization of GRA10 in the DFC was further confirmed by dual staining with monoclonal antibodies against B23 (nucleophosmin) and C23 (nucleolin), which are already known in the localization of the DFC of nucleolus (Fig. 2B, 2C). The localization of the expressed GFP-GRA10 was overlapped with those by B23 and C23. The translocalization of GRA10 into the nucleolus was caused by the function of a putative nucleolar localizing sequence (NoLS) from 199 to 213 amino acids of random repeat of lysine (K) and arginine (R) as RKKRRRSGKKKRGKR motif in GRA10 (Fig. 3). Expressed GFP from plasmids containing GRA10 DNA fragments including NoLS was translocated into nucleolus (Figs. 3A, 3D, and 3F) whereas the others without NoLS dispersed in the cytoplasm of the cells (Figs. 3B, 3C, and 3E).
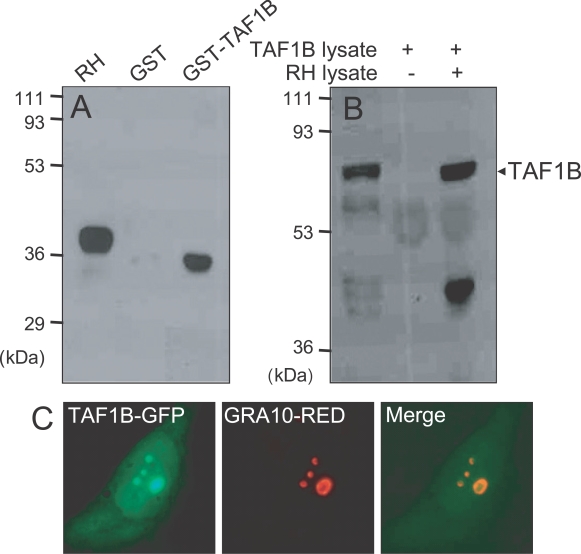
TAF1B was a prey protein to GRA10 in the yeast two-hybrid technique. Specific interaction between TAF1B and GRA10 was confirmed by GST pull-down assay and immunoprecipitation assay (Fig. 4). In the GST pull-down assay (Fig. 4A), GRA10 was bound to glutathione-Sepharose beads to be blotted by mAb Tg378 under the presence of TAF1B-GST fusion protein, while not bound to the beads in the GST only fraction without TAF1B. The size of GRA10 expressed and purified from bacteria was a little smaller than that blotted in RH tachyzoites. By the immunoprecipitation assay (Fig. 4B), His-tagged TAF1B was precipitated with mAb Tg378 to protein A beads under the presence of RH lysate, which containing GRA10, but not precipitated in the fraction without RH lysate. GRA10 and TAF1B were co-localized in the nucleolus after co-transfection (Fig. 4C). Green fluorescence of TAF1B-GFP overlapped with red fluorescence from GRA10-Red as revealed by merge image.
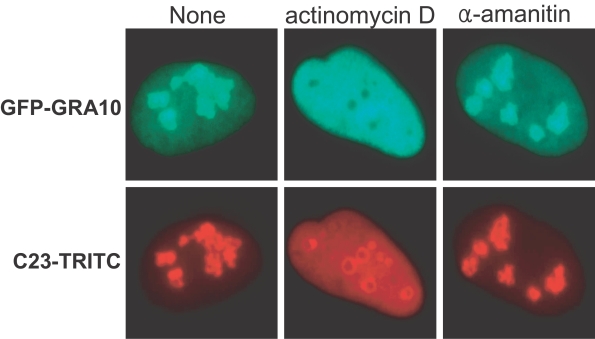
The nucleolar condensation of GRA10 was affected by actinomycin D treatment but not by α-amanitin after transfection (Fig. 5). Expressed GFP-GRA10 was escaped from the nucleoli to disperse evenly over the nucleoplasm and the primary nucleolar locations remained as hollows in the nucleoplasm under a low dose of actinomycin D. Immunofluorescence of C23 was also affected the localization of the DFC of nucleoli by actinomycin D to be dispersed to the nucleoplasm, but not perfect as in the case of GFP-GRA10. The localities of GFP-GRA10 and C23 were not affected by α-amanitin treatment even in high doses.
DISCUSSION
GFP-GRA10 fusion protein expressed in HeLa cells was translocalized clearly to the nucleoli within the nucleus without any traces in the cytoplasm, whereas the other GFP fused GRA proteins were detected in the cytoplasm of the transfected cells. Furthermore, GRA10 was concentrated specifically in the DFC of the nucleolus morphologically. The nucleolar translocalization of GRA10 was caused by an innate NoLS of GRA10. GRA10 interacted with TAF1B in the yeast two-hybrid technique, of which the specific binding was proved by the GST pull-down assay and the immunoprecipitation assay. They were co-localized in the nucleolus after co-transfection. The nucleolar condensation of GRA10 was affected by actinomycin D, such that GFP-GRA10 was escaped from the nucleolus to distribute evenly over the nucleoplasm and the nucleolar locations remained as hollows in the nucleoplasm.
During the infection with T. gondii, proteins from 3 secretory organelles are used for the favorite environment to enter into host cells successfully and for the decoration of PV and PVM to get along with the host cell components. Therefore, the localities of secretory proteins have been restricted to the PV and PVM of the infected cells until now. In recent studies, T. gondii proteins could escape from the PV into the host cell cytoplasm and be processed to present antigens by the endogenous MHC class I pathway (Gubbels et al., 2005) and protein phosphatase 2C of T. gondii was secreted from the rhoptry to be delivered into the host cell and directed to the host nucleus (Gilbert et al., 2007). Prior to interact with host cell nuclear proteins, GRA10 should be secreted across the PVM to the host cell cytoplasm (Fig. 1). Whether this occurs for GRA10 or not is currently unknown as in other dense granular proteins. PVM itself is not a rigid mold, is rather a biomembrane, where the endocytosis/exocytosis occurs continuously between the parasite and the host cell cytoplasm to maintain both the nutritional supply and protection from intracellular attacks. During the exocytosis, PVM inserted GRA10 can be pinched in the protruding exocytosomes. Another presumptive way of GRA10 to cross the PVM barrier resides in the 2 putative transmembrane domains (TMD) of GRA10, which leads to an additional PVM membrane trafficking to nearby endoplasmic reticulum or Golgi apparatus of the host cell cytoplasm.
NoLS located in the downstream of TMDs enables GRA10 to be delivered to the nucleus of the transfected cells. Regardless of the length of protein expressed as GFP-GRA10 fragments, fragments containing NoLS were translocated into nucleolus, whereas the others without NoLS dispersed in the cytoplasm of the cells even the fragments have TMDs. NoLS seemed to be the unique critical motif for nucleolar translocalization of GRA10 (Fig. 3). In the T. gondii, de novo synthesized GRA10 is condensed in dense granules not in nuclei, which suggests the priority of the dense granular targeting to nuclear targeting within T. gondii. Several NLS or NoLS from viruses to eukaryotic cells are summarized in Table 1. The repeating array of lysine and arginine of GRA10 NoLS was similar to various kinds of viral NoLS rather than those of eukaryotic cells. During the infection of viruses, interaction with host nucleolus is a common phenomena and data is accumulating to suggest that viruses use the nucleolus or its antigens to enhance viral replication, either by interacting directly with proteins such as nucleolin (C23), or altering host cell transcription, translation and the cell cycle indirectly (Hiscox, 2002). In case of T. gondii infection, growing parasites within PVM have no direct connection to host nucleolus but secrete a signal to control host cell transcription machinery. Secreted and nuclear translocalized GRA10 is a candidate molecule to contribute to the control.
Nucleolar translocalized GRA10 positioned in DFC mainly among the 3 subcompartment of the nucleolus, those are fibrillar center (FC), DFC, and granular component (GC) of transfected HeLa cell nucleus (Fig. 2). This architecture is thought to largely reflect the vectoral maturation of the pre-ribosomes, with the transcription of the rDNA likely occurring at the interface between the FC and DFC, nascent transcripts reaching out into the body of the DFC and nascent pre-ribosomes progressively migrating from the DFC to the GC, as pre-rRNA processing, pre-rRNA modification and ribosome assembly occur (Thiry and Lafontaine, 2005; Hernandez-Verdun, 2006). Besides, their respective morphological properties, each nucleolar subcompartment is characterized by a distinct biochemical composition. FCs contain DNA, including rDNA in a transcription competent structure, some nascent pre-RNAs and most importantly transcription factors, such as the RNA polymerase I, the upstream binding factors (UBF) and the DNA topoisomerase I. The DFC, largely acknowledged as the site of early pre-rRNA processing and modification reactions, contains nascent pre-rRNAs and antigens such as the fibrillarin. Nucleolar proteins rapidly associate and dissociate with nucleolar components in continuous exchanges with the nucleoplasm (Phair and Misteli, 2000). The nucleolar proteins engaged in rRNA transcription and processing such as UBF, B23, Nop52, nucleolin and Rpp29 also move with rapid recovery rates in the nucleolus as does fibrillarin (Louvet et al., 2005).
Nucleolus controls many processes inside the cell including ribosome biogenesis, RNA processing, cell senescence, telomerase activity and the cell cycle. RNA polymerase I (Pol I) functions in the transcription of genes encoding the large rRNAs in most eukaryotes (reviewed by Grummt, 2003). It requires auxiliary factors that mediate promoter recognition, promote transcription elongation, and facilitate transcription termination. Initiation of rDNA transcription is mediated by a specific multimeric complex containing RNA Pol I and at least four basal transcription initiation factors, such as TATA box binding protein (TBP) and 3 TBP-associated factors (TAFs) (Friedrich et al., 2005). Among them, the TAF1B was a prey of GRA10 by the yeast two-hybrid assay. The binding specificity of the 2 proteins was verified in the GST pull-down assay and immunoprecipitation method (Fig. 4). The rDNA transcription oscillates during the cell cycle progression. Transcription is maximal in the S and G2 phases, shuts down in mitosis, and slowly recovers in G1. Reversible phosphorylation of the TAF1B/SL1 and UBF is used as a molecular switch to shut down rDNA transcription during mitosis (Grummt and Pikaard, 2003). It is suggested that nucleolar localization and binding to TAF1B of GRA10 exerts on the continuous rDNA transcription independent of host cell cycle to supply regardless of the different cell cycles between host cell and growing T. gondii within the PVM.
The nucleolar condensation of GRA10 was affected by actinomycin D, such that GFP-GRA10 was escaped from the nucleolus to distribute evenly over the nucleoplasm and the nucleolar locations remained as hollows in the nucleoplasm (Fig. 5). Nucleolar disintegration is related to the rDNA transcriptional arrest under certain physiological conditions or induction by the treatment of low dose of actinomycin D (Hernandez-Verdun, 2006). Actinomycin D inhibits RNA synthesis via binding to rDNA and blocking the movement of RNA Pol I. Inhibition of Pol I transcription by actinomycin D does not prevent the movement of nucleolar proteins, but the traffic in segregated nucleoli changes differentially for different nucleolar components (Chen and Huang, 2001). GRA10 was diffused rapidly into the nucleoplasm by RNA Pol I inhibition, which supported the role of GRA10 in the rDNA transcriptional machinery.