INTRODUCTION
Trichomonas vaginalis, the causative pathogen for the most prevalent non-viral sexually transmitted disease in humans, triggers vaginitis, cervicitis, and urethritis [1]. The impact of T. vaginalis, as a model organism of host-parasite interaction has been extensively investigated, and multiple interactions between host receptors and protozoan virulence determinants are known to occur [2,3]. A previous study identified several antigenic proteins present in the membrane portion of T. vaginalis, which includes α-actinin 2 [4]. Alpha-actinin is a Ca2+-regulated protein involved in actin cross-linking [5]. Five putative α-actinin genes were discovered in the databases of T. vaginalis. Among them, T. vaginalis α-actinin 2 (Tvα-actinin 2) was investigated, due to its strong reactivity to patients’ sera [6]. When the parasite adheres to the host cells and it transforms into the amoeboid form, this protein is located close to the cell surface showing colocalization with actin.
As the main antigen of T. vaginalis, Tvα-actinin 2 has been used to monitor trichomoniasis in populations [7] and to examine the history of trichomoniasis in plasma samples of male patients [8]. Reactive epitopes of Tvα-actinin 2 have been finely mapped using sera from both male and female patients with trichomoniasis [9]. In this study, Tvα-actinin 2 was found to be secreted from T. vaginalis. We examined a possibility that this secreted protein of T. vaginalis plays a role in the modulation of host immune responses using vaginal and prostate epithelial cells as well as mouse dendritic cells (DCs).
MATERIALS AND METHODS
Bacterial and human cell culture
Escherichia coli BL21 (DE3) were grown at 37°C in Luria-Bertani medium (0.5% yeast extract, 1% tryptone, 1% NaCl, pH 7.5) supplemented with ampicillin (100 μg/ml) in order to maintain the plasmids. Medium components were obtained from BD-Difco (Franklin Lakes, New Jersey, USA), and the chemicals and antibiotics were purchased from Sigma-Aldrich (St. Louis, Missouri, USA).
Human vaginal epithelial cells (VECs, ATCC CRL-2614, American Type Culture Collection, Manassas, Virginia, USA) were grown in Dulbecco’s Modified Eagle’s Medium (Gibco BRL) complemented with 10% heat-inactivated fetal bovine serum (FBS), 100 U/ml penicillin, and 100 μg/ml streptomycin. Human prostatic epithelial cells (RWPE-1, ATCC CRL-11609, ATCC) were grown using Keratinocyte Serum Free Medium (Gibco BRL) containing 0.2 ng/ml epidermal growth factor and 0.05 mg/ml bovine pituitary extract, 5% FBS, 100 U/ml penicillin, and 100 μg/ml streptomycin. These human epithelial cells were cultured to 80–90% confluence at 37°C/5% CO2.
Formation of Tvα-actinin 2-specific antibodies (Abs)
Full-length recombinant Tvα-actinin 2 (rTvα-actinin 2) was prepared as previously described [12]. rTvα-actinin 2 was used as antigen to produce polyclonal Abs via intraperitoneal injection into pathogen-free rats (CrjBgi: CD[SD]IGS, 7-week-old, female). After 3 time injections at 2-week intervals, sera were obtained from immunized rats, and the titer of the resultant Abs was tested.
Preparation of secreted proteins and western blot analysis
Secretomes were prepared from VECs, T. vaginalis trophozoites, and VECs incubated with T. vaginalis at a multiplicity of infection (MOI) of 5. VECs were seeded in 12-well culture plates (1×105 cells/well), and then incubated overnight at 5% CO2 and 37°C. T. vaginalis washed with phosphate buffered saline (PBS: 1.7 mM KCl, 137 mM NaCl, 2 mM KH2PO4, and 10 mM Na2HPO4, pH 7.3) were resuspended in the TYM media without serum. After 6 h-incubation of VEC with and T. vaginalis at a MOI of 5, the cells were removed by 20-min centrifugation at 1,200 g, and the collected secretomes were concentrated using a Centricone (Milipore, Darmstadt, Germany).
Ten micrograms of secreted proteins were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and transferred to a polyvinnylidene fluoride membrane (Millipore). The membrane was incubated with anti-rTvα-actinin 2 Abs (1:1,000 dilution) and subsequently with horseradish peroxidase (HRP)-conjugated mouse anti-rat IgG (1:1,000 dilution). Immunoreactive bands were visualized using an Enhanced Chemiluminescence (ECL) System (GE Healthcare, Chicago, Illinois, USA).
Determination of cytokine concentration
A sandwich ELISA was performed to determine the levels of TNF-α, IL-12, IL-6, and IL-10 in culture supernatants (BD Biosciences, Franklin Lakes, New Jersey, USA). The levels of cytokines were determined by monitoring the absorbance at 450 nm, and the cytokine concentrations were calculated based on the standard curves generated using recombinant cytokines.
To examine whether cytokine production in rTvα-actinin 2-induced VECs was caused by contamination with lipopolysaccharide (LPS) in the prepared rTvα-actinin 2, endotoxin level of rTvα-actinin 2 was measured using the Limulus Amebocyte Lysate LPS Detection Kit (Lonza, Basel, Switzerland). In addition, rTvα-actinin 2 were purified using the Detoxi-GelTM Endotoxin Removing Gel (Pierce, Rockford, Illinois, USA) before it was challenged to the host cells for measurement of cytokine production.
Experimental mice
BALB/c mice (6 week-old females) were purchased from OrientBio (Seongnam, Korea). Mice received care according to our institutional guidelines and the legal requirements of Korea.
Preparation of DCs
Mouse bone-marrow cells isolated from the tibia and femur of mice, were resuspended in red blood cell lysis buffer (10 mM KHCO3, 150 mM NH4Cl, and 1 mM EDTA, pH 7.4). The cells were differentiated into bone marrow-derived dendritic cells (BMDCs) in RPMI1640 medium (Gibco BRL) containing 1 mM HEPES buffer, 20 ng/ml granulocyte-macrophages colony-stimulating factor (Peprotech, Rocky Hill, New Jersey, USA), 100 U/ml penicillin/streptomycin (Gibco BRL), 10% FBS, 50 μM mercaptoethanol, and 0.1 mM nonessential amino acid (Sigma-Aldrich). The medium for these cells were replaced with the fresh one every 2 days, and the cultures were maintained at 37°C/5% CO2 until day 6 or 7.
Flow cytometric analysis of cell surface markers
Immature BMDCs were converted into mature cells by incubation with LPS for 16 hr. Mature DCs were harvested, and then reacted with rTvα-actinin 2 for additional 16 hr. The cells were then stained for 20 min on ice using the Live/dead Fixable Dead Cell Stain Kit (Invitrogen, Carlsbad, California, USA) to monitor their viability. Following 3 washes with PBS, the cells were reacted with one of these chemicals [allophycocyanin (APC)-conjugated anti-CD80, and phycoerythrin (PE)-conjugated anti-CD86, and peridinin chlorophyll (PerCP)-Cy 5.5-conjugated anti-mouse I-A/I-E for MHC class II] along with APC-eFluor780-conjugated anti-CD11c for 20 min. After 3 PBS-washes, the cells were prepared in 500 μl of PBS containing 1% BSA, and 0.1% sodium azide, and their fluorescence was measured by flow cytometry. The data analyses were performed using FlowJo data analysis software (FlowJo LLC, Ashland, Oregon, USA).
Assays of BMDC-dependent Treg differentiation
Naive T cells were obtained from the draining lymph nodes of mice. From the prepared cells in RBC buffer, CD4+CD25+ or CD4+CD25− regulatory T cells were prepared using CD4+ CD25+ Regulatory T cell Isolation Kit (Miltenyi Biotec, Auburn, California, USA) as directed by the manufacturer’s instructions. The isolated CD4+CD25+ or CD4+CD25− regulatory T cells were monitored for the expression of Tregs markers, FOXP3, CD103, CD39, and CD152 (CTLA-4) using specific Abs.
LPS-stimulated DCs (1×104 cells/well) incubated with 0.1 μg/ml of rTvα-actinin 2 for 3 h were co-cultured with CD4+ CD25+ or CD4+CD25− regulatory T cells (1×105) at a DC: T cell ratio of 1:10. On day 7 of co-culture, supernatants were collected and assayed for IL-10, TGF-α, IFN-γ, IL-4, and IL-2 production by ELISA (BD Biosciences). As a control, PBS-treated DCs was also assayed for these cytokines.
Statistical analyses
Data were indicated as the means±SD obtained from 3 independent experiments. Statistical analysis of pairwise comparison was carried out using Student’s t-test (SYSTAT program, SigmaPlot version 9, Systat Software Inc., San Jose, California, USA). Differences were considered statistically meaningful if the P-values were<0.05.
RESULTS
Secretion of Tvα-actinin 2 by T. vaginalis
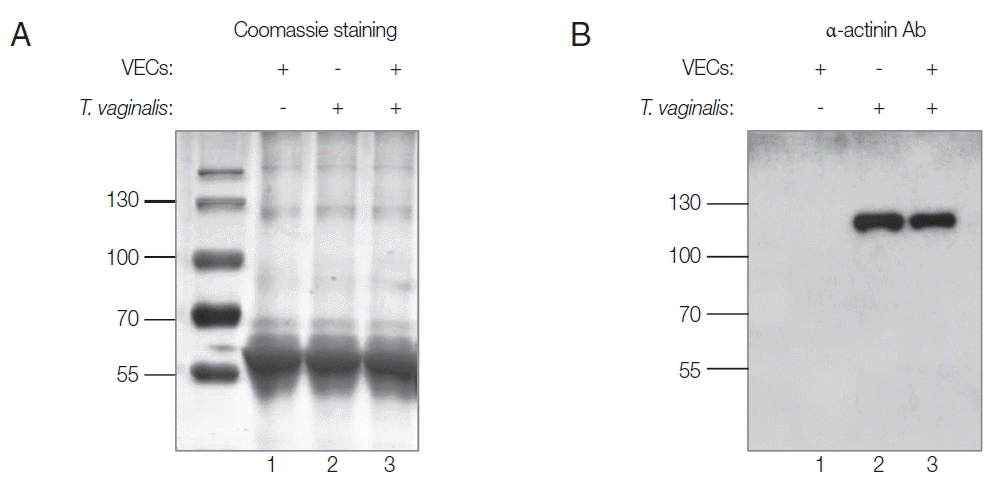
In this study, rTvα-actinin 2 protein was used as an antigen to immunize rats to make anti-Tvα-actinin 2 antibodies. Using Abs specific to rTvα-actinin 2, western blot analysis was performed to examine the presence of Tvα-actinin 2 in secreted proteins by T. vaginalis (Fig. 1B). Secretomes were prepared from VECs (Fig. 1A, B, lane 1) or T. vaginalis (Fig. 1A, B, lane 2). In addition, secreted proteins were also collected from T. vaginalis incubated with VECs (Fig. 1A, B, lane 3). No immunoreactive protein was observed in the western blot analysis of the secretome derived from VECs. In contrast, an immunoreactive protein of 124 kDa was clearly detected in the secretome from T. vaginalis, which is an expected size of Tvα-actinin 2. The same reactive protein was observed in the secretomes of the VECs incubated with T. vaginalis (Fig. 1B). No obvious difference was detected in the amount of this immunoreactive protein between T. vaginalis and T. vaginalis incubated with VECs.
Cytokine production in VEC and RWPE-1 cells incubated with T. vaginalis
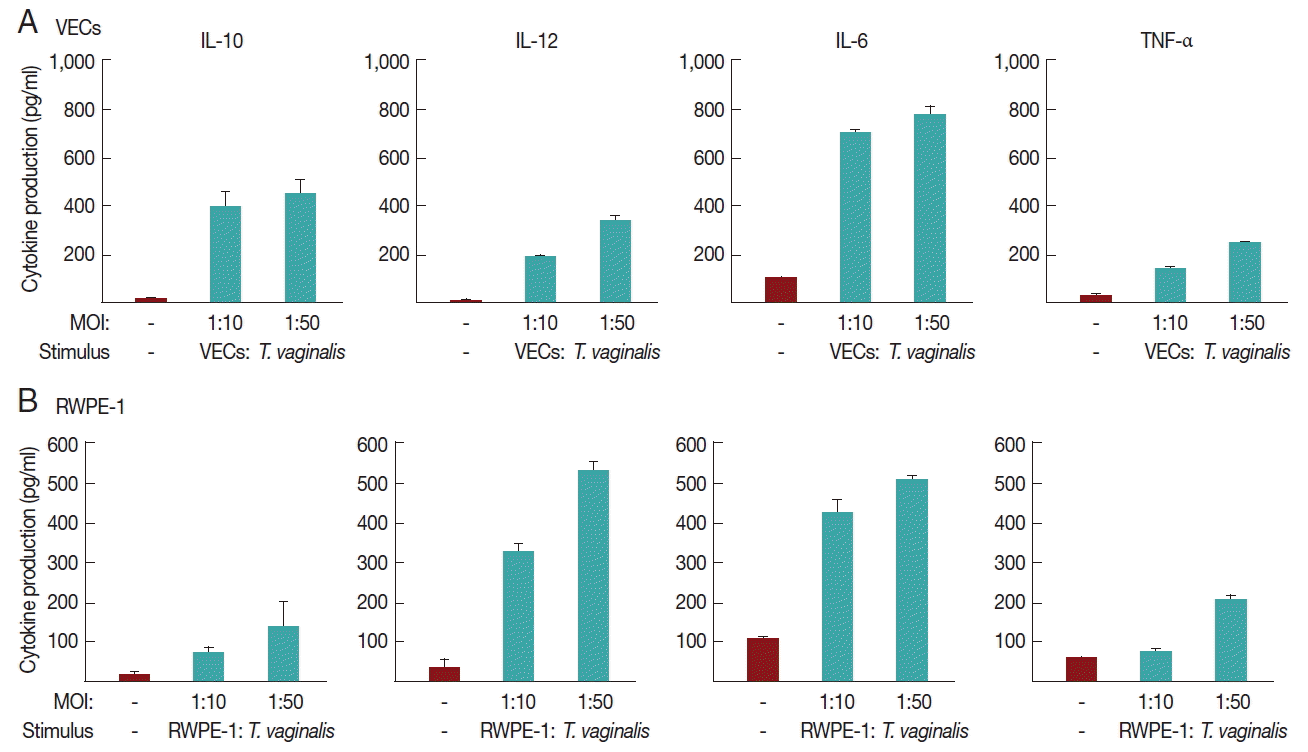
We investigated whether T. vaginalis trophozoites induced cytokine production in VEC and RWPE-1, primary target cells that T. vaginalis may encounter in vivo (Fig. 2).
A 16-hr incubation of VECs with T. vaginalis at MOIs of 10 and 50 triggered the secretion of IL-10, IL-12, and IL-6, and TNF-α. The induction of IL-6 production by T. vaginalis occurred up to 770±39 pg/ml, whereas the amount of TNF-α was 250±2 pg/ml at an MOI of 50 of T. vaginalis. When VECs were stimulated with T. vaginalis at an MOI of 50, the levels of released IL-10 and IL-12 were 450±62 and 330±15 pg/ml, respectively. The levels of TNF-α and IL-12 were proportional to the MOI of T. vaginalis.
RWPE-1 cells were also stimulated with T. vaginalis at MOIs of 10, and 50, and then assayed for cytokine production. Similar levels of TNF-α, IL-10, IL-12, and IL-6 were released from RWPE-1 cells by T. vaginalis at levels of 140–510 pg/ml. The induced levels of these 4 cytokines were increased at a higher MOI of T. vaginalis.
Cytokine production in VEC and RWPE-1 cells stimulated with rTvα-actinin 2
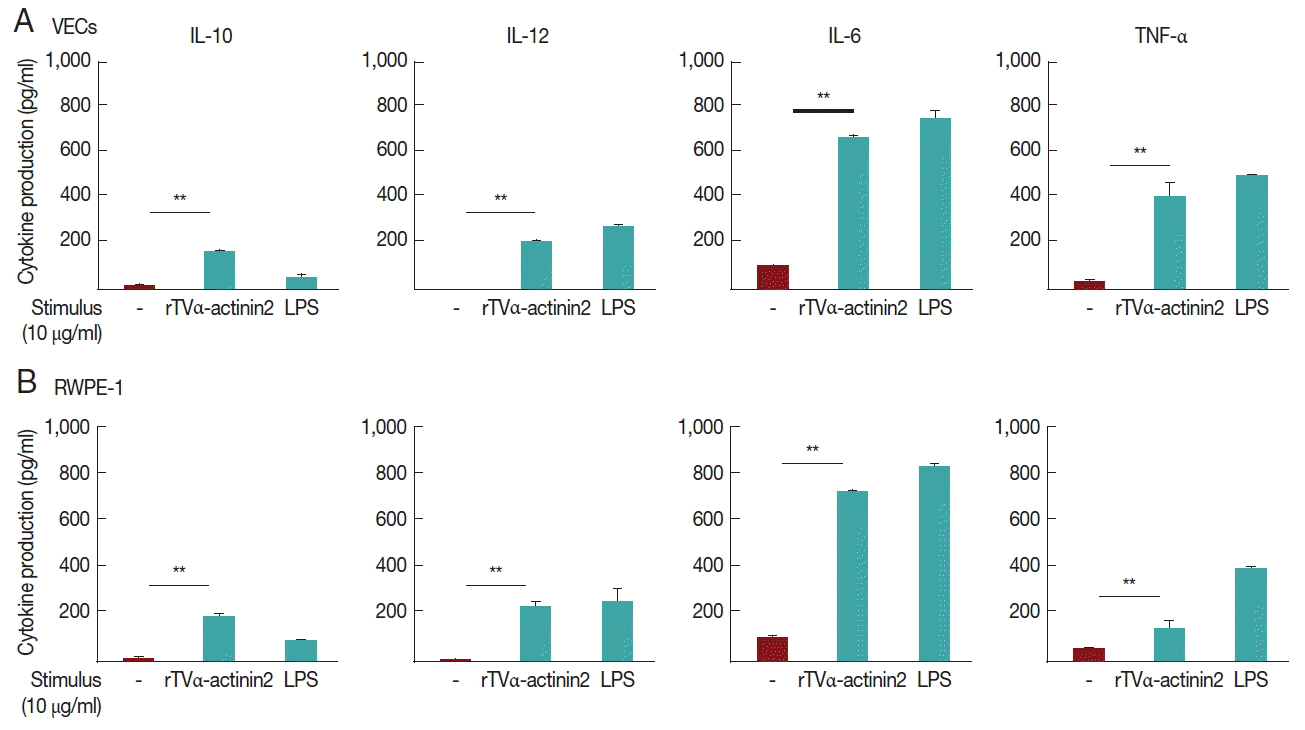
In this experiment, we examined whether rTvα-actinin 2 induces cytokine production in vaginal and prostate epithelial cell lines (Fig. 3A, B, respectively). To exclude a possibility that contaminated LPS in purified rTvα-actinin 2 induced cytokine production, the recombinant protein was further purified using Endotoxin Removing Gel before it was challenged to the host cells. The endotoxin level of rTvα-actinin 2 was 0.8 EU/ml, which is lower than 2.5 EU/ml, the value indicating contamination.
When VECs were treated with rTvα-actinin 2 (10 μg/ml), all 4 cytokines (TNF-α, IL-10, IL-12, and IL-6) were released ranging from 160±8 to 730±35 pg/ml. Comparable amount of cytokines was produced in the rTvα-actinin 2-exposed RWPE-1 with VECs.
rTvα-actinin 2 induces a tolerogenic profile in DCs by down-regulating the surface molecule and increasing IL-10 production
Since the rTVα-actinin-treated host cells produced immunosuppressive cytokines as well as pro-inflammatory cytokines, we focused on the function of tolerogenic dendritic cells (tDCs) which can induce and control immune tolerance via differentiation of regulatory T cells (Tregs).
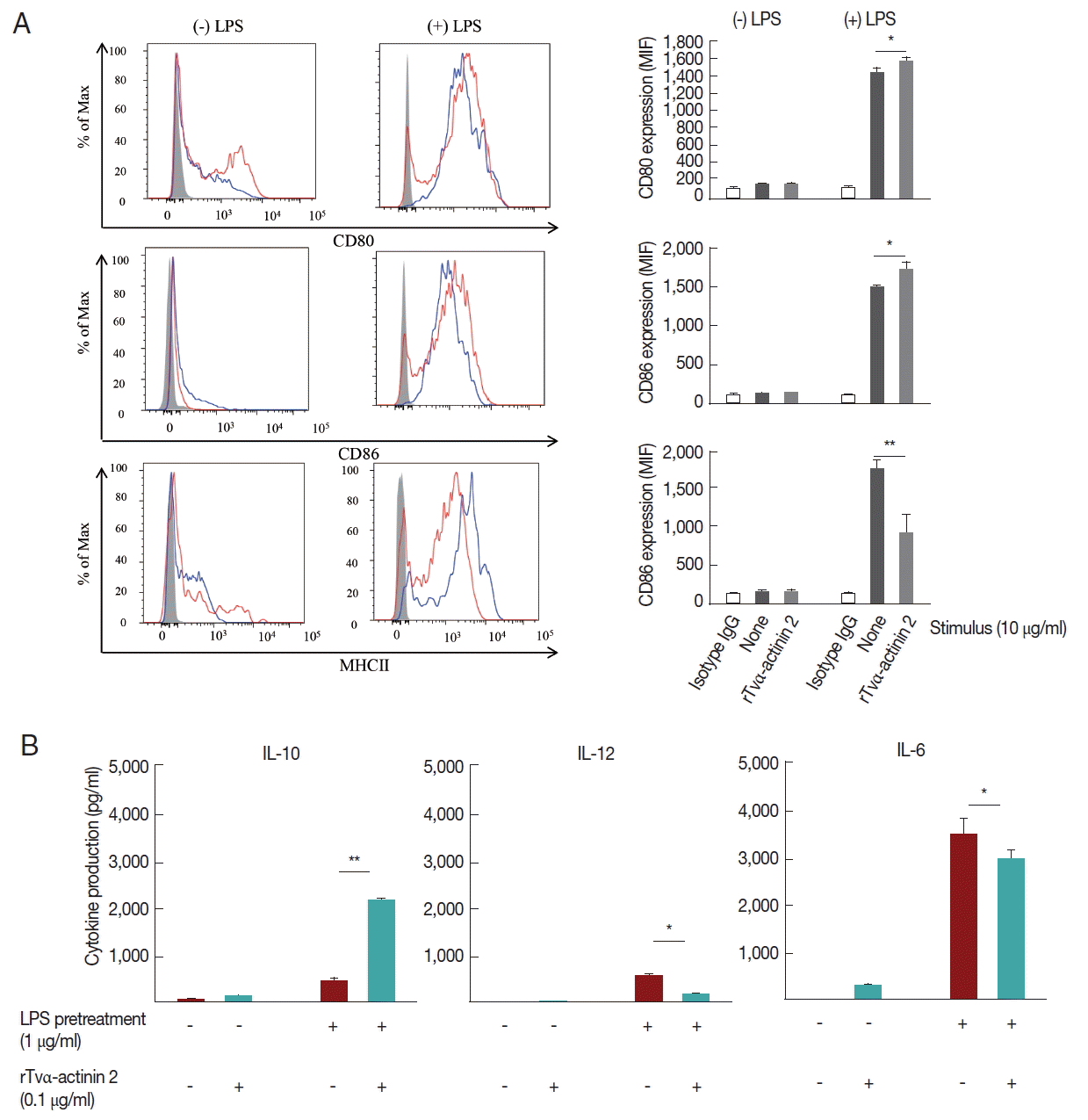
A set of mouse BMDCs was treated with LPS (1 μg/ml), and then incubated with 0.1 μg/ml rTvα-actinin 2 for 24 h. Another set of mouse BMDCs was treated with 0.1 μg/ml rTvα-actinin 2 for 24 h without LPS treatment. As a control, mouse BMDCs were treated with PBS instead of rTvα-actinin 2. The expression of DC activation markers, CD80, CD86, and MHC class II, was monitored in these cells using their specific antibodies (Fig. 4A). Each set of BMDCs was incubated with isotype IgG as a control. All 3 DC activation markers showed increased expression in response to LPS treatment. The expression of CD80 and CD86 was not significantly altered by an additional treatment with rTvα-actinin 2. Interestingly, expression of MHC II decreased dramatically [from 1,762±11 to 915±250 Mean Fluorescence Intensity (MFI)] with an additional challenge of rTvα-actinin 2. This result indicated that T. vaginalis α-actinin 2 down-regulates LPS-induced DC activation as shown by the decrease in MHC II expression.
Mouse BMDCs were stimulated with rTvα-actinin 2 (0.1 μg/ml), and then assayed for IL-10, IL-12, and IL-6 levels (Fig. 4B). While the levels of IL-12 and IL-6 in LPS-challenged DCs were decreased by rTvα-actinin 2 treatment, IL-10 production was further increased by rTvα-actinin 2 in LPS-treated DCs. IL-10 plays an anti-inflammatory role to prevent inflammatory pathologies [13].
rTvα-actinin 2 produces IL-10 in effector T lymphocytes by generation of regulatory molecules
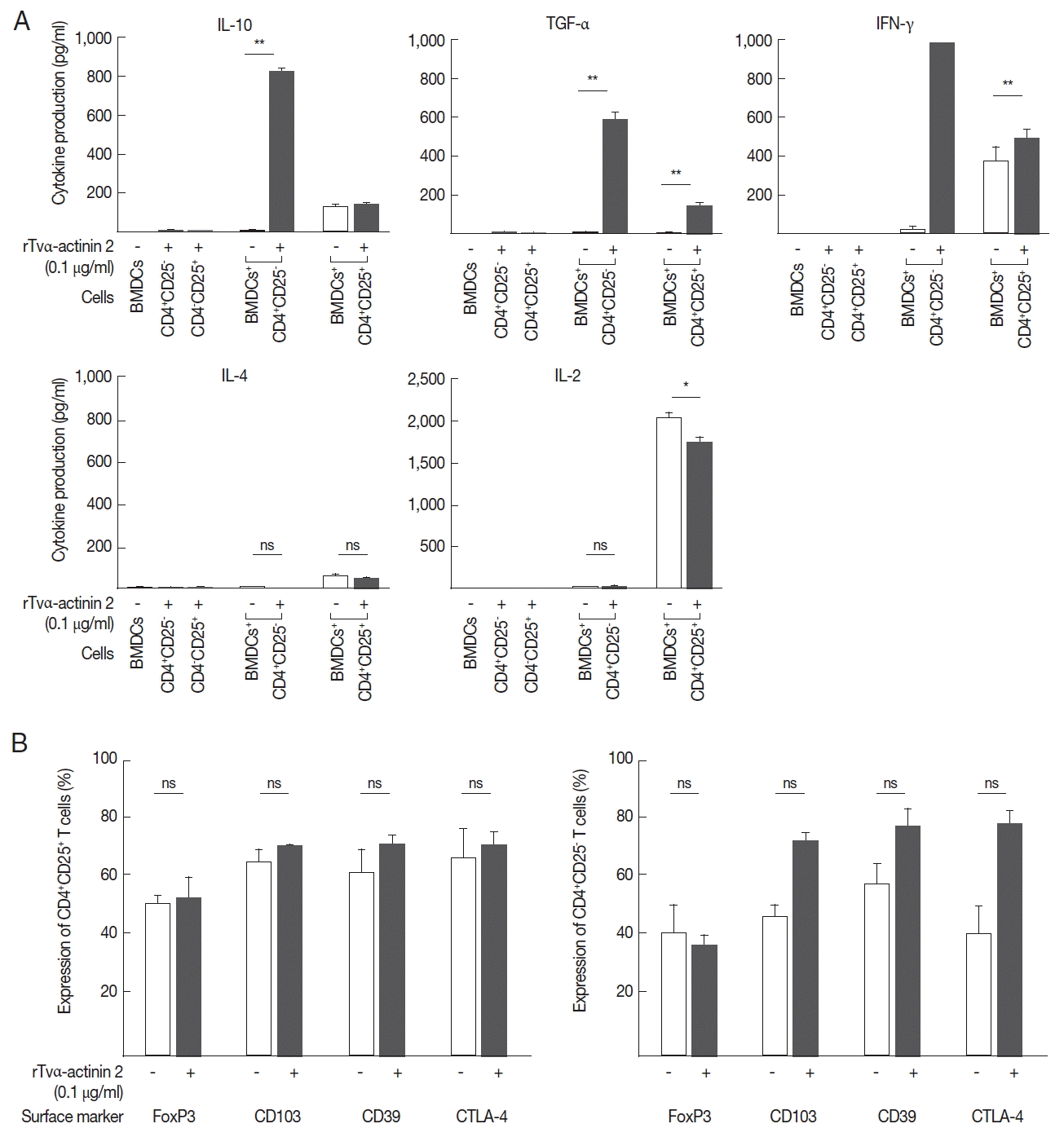
In this experiment, we examined whether IL-10 is induced in regulatory T cells upon co-incubation with rTva-actinin 2-treated DCs. The regulatory T cells isolated from the spleen and draining lymph nodes of mice were further sub-fractionated into natural Tregs (nTregs; CD4+CD25+) and inducible Tregs (iTregs; CD4+CD25−). When the obtained CD4+CD25− T cells were co-incubated with LPS-stimulated mouse BMDCs treated with rTvα-actinin 2, they secreted a high level of IL-10 (825 pg/ml) compared to the cells incubated with LPS-stimulated DCs (9 pg/ml) (Fig. 5A). In addition, these cells secreted a high level of TGF-β (585 pg/ml) and IFN-γ (1,000 pg/ml) only when they were co-incubated with LPS-stimulated DCs treated with rTvα-actinin 2. On the contrary, iTregs co-incubated with LPS-stimulated DCs treated with rTvα-actinin 2 did not release IL-4 or IL-2. On the other hand, nTregs secreted various amount of these cytokines ranging from 45 to 1,746 pg/ml when co-incubated with treated rTvα-actinin 2 of LPS-stimulated DCs. Except TGF-α, showing increased secretion after rTvα-actinin 2 treatment, nTregs demonstrated no alteration in cytokine levels in response to rTvα-actinin 2.
These Tregs were co-incubated with DCs treated only with LPS or DCs treated with LPS and rTvα-actinin 2, and the expression of FoxP3, CD103, CD39, and CTLA-4 was determined (Fig. 5B). The expression of Treg markers such as FoxP3, CD103, CD39, and CTLA-4 was not significantly changed by an additional treatment of LPS-stimulated mouse BMDCs with rTvα-actinin 2 in the CD4+CD25+ cells. In case of CD4+ CD25− cells, the expression of 3 markers, CD103, CD39, and CTLA-4, was increased upon co-incubation with rTvα-actinin 2-treated DCs compared with that in cells co-incubated with the untreated DCs. On the contrary, FoxP3 level was not changed by rTvα-actinin 2 treated DCs. This result suggests that rTvα-actinin 2 induced the Treg phenotype in effector T lymphocytes.
DISCUSSION
Since Tvα-actinin 2 was identified as an immunogenic protein reacting to anti-T. vaginalis IgGs of female patients with trichomoniasis [7], it has even been used to monitor men’s serological samples for trichomoniasis exposure and risk of prostate cancer [8]. Recently, this protein was finely mapped for epitopes reactive to patients’ sera from only females or from both males and females [9]. To be functional with respect to host interaction, this protein in the pathogen should be secreted or be present on its outermost surface. In this study, Tvα-actinin 2 was found in the secretome of T. vaginalis (Fig. 1B) and its secretion was not affected by the presence of the host cells, VECs. Due to the lack of signal sequence in Tvα-actinin 2, it should be examined how Tvα-actinin 2 is secreted from T. vaginalis. However, many proteins lacking signal sequences were found to be secreted via exocytosis [14].
In this study, we examined immune responses of the host epithelial cells, especially vaginal and prostate cells by measuring cytokine production upon incubation with T. vaginalis as well as rTvα-actinin 2 protein (Figs. 2, 3, respectively). Among the produced cytokines, IL-10 attracted our attention because it is an anti-inflammatory cytokine preventing inflammatory pathologies [15]. Therefore, we examined whether rTvα-actinin 2 plays a role in immunosuppression (Fig. 4), clearly indicating that rTvα-actinin 2 modulates DC activation by down-regulation of IL-12/up-regulation of IL-10, and inhibition of LPS-induced maturation of DCs. Secretion of IL-10 by tDCs is essential for tolerance in Treg differentiation via several different pathways [16].
Several components of T. vaginalis modulate the host immune response. Human neutrophils and macrophages stimulated with T. vaginalis produce IL-8 and the acute proinflammatory cytokine IL-6, TNF-α, and IL-1β, thereby initiate the immune response to T. vaginalis infection [10,17]. Especially, IL-6 is a critical factor of chronic inflammation in prostate cancer [18]. A recent study on interaction with prostate epithelial cells and T. vaginalis demonstrated increased production of IL-6 and epithelial–mesenchymal transition markers [19]. T. vaginalis lipophosphoglycan was shown to mediate adhesion of these parasites to VECs, and to induce a selective up-regulation of chemotactic cytokines in a TLR4-MyD88-independent manner [20]. Screening of T. vaginalis antigens with patients’ sera led to the isolation of T. vaginalis enolase which has plasminogen binding activity and whose expression is up-regulated by contact between the parasite and VECs [21]. Our results show immunosuppressive characteristics of the infection triggered by T. vaginalis. Expression of IL-12 and TNF-α was suppressed in T. vaginalis–adhesive cells, suggesting that this inhibitory mechanism is induced by T. vaginalis to evade host immunity [22]. Dying neutrophils by T. vaginalis result in the resolution of vaginal inflammation by inducing anti-inflammatory cytokine production in human macrophages [23]. In a study observing DC activation against various pathogens, formalin-fixed T. vaginalis induces only IL-10, but not IL-12 [24]. Importantly, excretory-secretory products (ESP) of T. vaginalis triggered tolerance as shown the inhibition of LPS-induced maturation of DCs [25]. Western blot analysis of T. vaginalis ESP with anti-Tvα-actinin 2 revealed that this protein is also present in the secreted fraction (Fig. 1B). Therefore, it is likely that Tvα-actinin 2 is one of the protozoan molecules functioning as an immunosuppressor during trichomoniasis, which acts both as a surface and secreted virulence factor. Interestingly, T. vaginalis homolog of macrophage migration inhibitory factor, TvMIF was discovered to induce inflammation and cell proliferation, thus leading to the progression of prostate cancer [26]. Identifying a host receptor for Tvα-actinin 2 would reveal the mechanisms underlying this protozoan molecule functions as a modulator for host immune systems.
Modulation of DC activity by rTvα-actinin 2 was mediated by Treg cells as shown in Fig. 5. IL-10 is known to initiate an anti-inflammatory feedback loop by the cooperative action with other immune cells [27]. The tDCs activated by IL-10 in peripheral tissues, obtain the ability to secrete IL-10, and then move to lymphoid organs, a site for Treg differentiation. We present evidence that Tvα-actinin 2 could generate iTregs during infection. Co-cultivation of Tvα-actinin 2 did not increase the levels of IL-10, TGF-β, and IFN-γ in nTregs (CD4+CD25+) but increased the levels of IL-10, TGF-β, and IFN-γ in CD4+ CD25− T cells. The high surface levels of CD103, CD39, and CTLA-4 on T effs (CD4+CD25−) help to contribute to tolerance of T. vaginalis infections as a consequence of IL-10 production. Additionally, IL-10 is required for the suppressive phenotype of Tregs against the inflammatory signals. IFN-γ is a key factor for the conversion of CD4+CD25− T cells into iTreg as shown in suppressed autoimmunity of the IFN-γ deficient mice [28].
Thus, our results demonstrated that rTvα-actinin 2 is responsible for immune tolerance, a characteristic of trichomoniasis by modulating the maturation and cytokine production by DCs.