INTRODUCTION
Macroorchis spinulosus is a minute intestinal trematode of the family Nanophyetidae, which is represented by large testes. Adult M. spinulosus were collected from the small intestine of mammals, infected naturally or experimentally, such as dogs, cats, mice, guinea pigs, rabbits, and albino and house rats (Ando, 1918). The metacercariae of M. spinulosus were found from crustacean intermediate hosts such as freshwater crabs, Potamon dehaani and P. obtusipes (Ando, 1918, 1921) and crayfish, Cambaroides similis (Chai et al., 1996). In recognition of one or more morphological differences, distinctive intermediate hosts and geographical segregation, new species of the genus Macroorchis, M. himizu, M. chimarrogalus and M. elongates, were proposed (Machida and Uchida, 1982; Saito et al., 1982). Recently, the morphological features of M. spinulosus were found different from the original descriptions and supplemented and redescribed in detail based on adult morphologies (Chai et al., 1996).
Information on surface ultrastructures of the growth and development of M. spinulosus in the final hosts is scarce. It is reported that the excysted metacercariae of M. spinulosus grow up rapidly to ovigerous adult flukes in the small intestine of final hosts (Woo et al., 2003). This work was carried out to get more information on developmental surface ultrastructures of M. spinulosus in the final host, and may provide knowledge on functions of the differentiated tegument in host-parasite interactions.
MATERIALS AND METHODS
The crayfish, Cambaroides similis, were caught in the streams in Macheon-myon, Hamyang-gun, Gyeongsangnam-do, Korea from October to December 1993. The crayfish were crushed with a mortar and a pestle, and washed with PBS. The M. spinulosus metacercariae were collected from the sediment under a dissecting microscope. Albino rats, Charles River Sprague-Dawley strain, 4-week-old, were fed each with 100 metacercariae.
The Rats were sacrificed at predetermined intervals, 1, 3, 5, 7, 14, and 28 days after the infection. The rats' intestine was resected out and divided into duodenum, jejunum and ileum. Intestinal contents were washed in PBS and M. spinulosus were recovered from the sediments under a dissecting microscope. The recovered flukes were washed with cold physiological saline 3 times and fixed in cold 2.5% glutaraldehyde (0.1 M phosphate buffer, pH 7.2) at 4℃. After washing with buffer, flukes were post-fixed in 1% osmium tetroxide (0.1 M phosphate buffer, pH 7.2) for 1 hr. The fixed flukes were dehydrated through a graded series of ethanol, exchanged with acetone, and critical point-dried. The dried flukes were mounted on metal stubs and coated with gold in an ion sputtering coater. The prepared fluke specimens were observed with a scanning electron microscope (ISI DS-130C, ISI Co., Tokyo, Japan) at an accelerating voltage of 10 kV.
RESULTS
Juvenile flukes
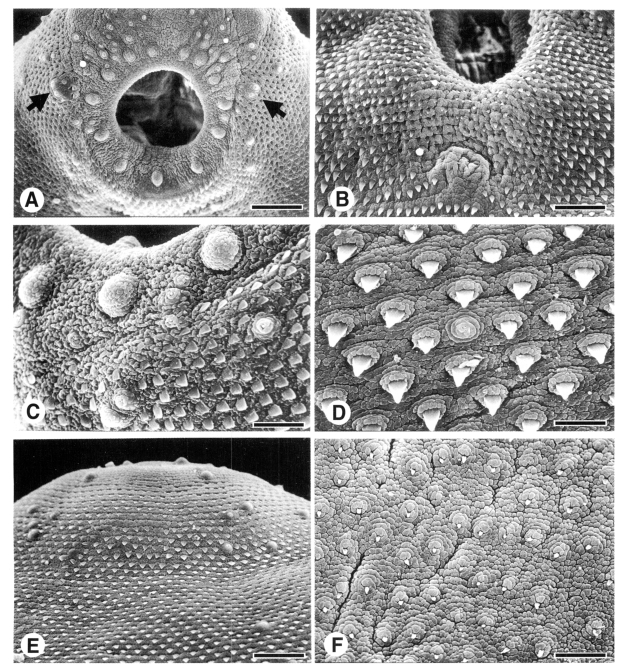
The body of 1-day-old fluke was flat and leaf-shape pointed posteriorly. The oral sucker, circular and wide open, was subterminal at anterior end of the body. The acetabulum was median at anterior two-fifths of the body (Fig. 1A). The lip of oral sucker appeared velvety and wrinkled cytoplasmic processes, and was devoid of tegumental spines. Unciliated sensory papillae swelled with wrinkled surface (Type II papillae), 14 in number, located equidistantly on the lip of oral sucker. Sensory papillae bulbous with the tegument wrinkled at base and smooth at apex and with a short cilium (Type I papillae) ornamented densely around the oral sucker and sparsely over the body surface. The Type I papillae were found on inner surface of the oral sucker. Anterior apex was devoid of tegumental spines and appeared cytoplasmic processes corrugated. On anterior outskirts of the oral sucker Type I papillae intercalated Type II papillae, looks supplementing the later ones (Fig. 1B). Surface of the acetabulum was covered with worm-like cytoplasmic processes arranged radially. The acetabulum was encircled with two circles, inner and outer, of Type II papillae. Of the outer circle, six Type II papillae located equidistantly along outer margin of the acetabulum. Of inner circle, 5-6 Type II papillae sat on anterior two-thirds of the lip (Fig. 1C). Type I papillae, 3-5 in number, were found on lateral rim of the acetabulum. Genital pore opened distantly postero-median to the acetabulum, revealing general feature of the ventral surface. On dorsal surface, Type I papillae aligned bilaterally symmetrical (Fig. 1D) and became more distant posteriorly.
The body surface, from anterior to posterior ends, was beset with tegumental spines, spade-like anteriorly and peg-like posteriorly (Figs. 1E, 1F). The tegumental spines were deployed concentric to the oral sucker on anterior half of the body. Posterior end of the body notched in V-shape with an excretory pore. Cytoplasmic processes was cobblestone-like on anterior half of the body and became slightly finer posteriorly (Figs. 1E, 1F).
In 3-day-old flukes, cytoplasmic processes on anterior apex devoid of tegumental spine were velvety. On the body surface, the cytoplasmic processes differentiated into transient shapes from cobblestones to velvet between tegumental spines.
Adult flukes
In 5-day-old flukes, the body got thicker and paremchymatous, and bulged out like a hip in posterior half. From half of the observed flukes, sperms ejaculated from the genital pore found on the ventral surface. Type I papillae concentrated on the anterior apex while very rare all over the body surface. On margin of the oral sucker, two grouped form of 2-3 Type I papillae located bilaterally symmetrically. Type II papillae got prominent contour on the lip of oral sucker (Fig. 2A). The acetabulum became thinner and less stout compared to the oral sucker. One grouped form of two Type I papillae was found anterior margin of the acetabulum. Genital opening revealed protruded as a mound devoid of tegumental spines, posterior to the acetabulum (Fig. 2B). Excretory pore, extruded buttock-like, with spine-free Y-shape area on posterior end of ventral surface. Around the excretory pore sensory papillae were not found. Tegumental spines on posterior half of the body retained its peg-like shape and density. Cytoplasmic processes were differentiated into velvety ones on the entire body surface.
In 7-day-old flukes, the surface structure retained generally as in the 5-day-old flukes. Anterior body armored heavily with tegumental spines and reduced loosely to the end of body. Tegumental spines got bigger and stouter than those of juvenile flukes. The spines revealed spade-shape with broader base on anterior body surface (Fig. 2C) and narrowed posteriorly to peg-like ones with a pointed-tip (Fig. 2D). The tegumental spines on the middle part of the ventrolateral surface of the body were wider than those on the dorsal surface. Cytoplasmic processes developed into fine velvety processes over the body surface (Figs. 2C, 2D). The swollen dome of Type II papillae, on the oral sucker and acetabulum, appeared strawberry-like covered with fine velvety cytoplasmic processes (Fig. 2C). Type I papillae elevated with spiraling tegument or with concentric circles; looked like a fried egg at top view (Figs. 2C, 2D).
In 14-day-old flukes, maximum width of the body was observed at posterior one-third level, just after the acetabulum. Whole surface featured similar to that of 7-day-old flukes. Posterior end of the body protruded out like a turret, centered with an excretory pore. Around the excretory pore was free from tegumental spines. At central part of the lip of oral sucker was spaced devoid of sensory papillae and tegumental spine, where cytoplasmic processes were wrinkled longitudinally. On anterior margin, transient to the apex, of the oral sucker, 3-4 Type I papillae intercalated the Type II papillae. On anterior surface, ventral and dorsal, of the body, some Type I papillae emerged very close, so seemed to form a grouped form.
In 28-day-old flukes, whole surface retained ultrastructure similar to that of 14-day-old flukes. The tegumental spines on posterior half of the body were decreased in their size and density. The acetabulum was wrinkled radially and equal size to the oral sucker. One Type II papilla on anterior center of outer circle flanked a Type I papilla. On the body surface, Type I papillae were most prominent observed yet, and aligned symmetrical bilaterally (Fig. 2E). Of the type I papillae, smooth dome based on 2-3 encircling rings supported with velvety cytoplasmic processes of adjacent surface. On the posteriormost surface, the tegumental spines reduced to minute in size and sparse in density. The cytoplasmic processes were finer on posterior surface by 1/2-1/3 than those on anterior surface of the body (Fig. 2F).
DISCUSSION
The cytoplasmic processes of M. spinulosus were cobblestone-like in juvenile flukes and differentiated into velvety ones in adult flukes. This developmental differentiation of cytoplasmic processes has been reported from intestinal and heterophyid trematodes. It has been considered for adult flukes that fine velvety cytoplasmic processes covering posterior half of the body is an efficient way of increasing absorptive area to provide increased nutritional demand of reproductive organs producing a large quantity of eggs (Køie, 1987; Hong et al., 1991). A reduction of width and density of the tegumental spines in posterior half of the body enhanced the increasing of absorptive surface area of M. spinulosus as in other flukes.
The peg-like single tipped tegumental spines on anterior surface of M. spinulosus retained their shape and density throughout developmental stages. In albino rats, a final host, M. spinulosus revealed habitat shift. It resided in the jejunum in juvenile stage and migrated up and established at duodenum as grew up to adult stages (Woo et al., 2003). The tegumental spines with a pointed tip may supportive for locomotion of juvenile flukes moving from one habitat to another. It is suggested from Centrocestus armatus, Metagonimus yokogawai and Neodiplostomum seoulense that less pointed tips on the juvenile flukes work more effectively for their locomotion and anchorage (Woo et al., 1998; Lee et al., 1984, 1985). Compared with single tipped spines, tegumental spines of heterophyid flukes, such as M. yokogawai and Heterophyopsis continua, increased pointed tips as growing up to adults (Lee et al., 1984; Hong et al., 1991). It was suggested that the scale-like multi-pointed tegumental spines on the anterior half surface of the body could function as an abrasive apparatus producing debris for feeding from the intestinal mucosa rather than as a locomotive apparatus (Hong et al., 1991).
Ciliated sensory papillae with a short or long cilium have been found in diverse species of trematodes (Fujino et al., 1979; Page et al., 1980). The cilium is extended through the tegument and connected to nerve fibers, so that it may sense contacts and changes encountered from environments and transmit those signals to the central nervous system. The ciliated (Type I) sensory papilla was suggested to function as tango-, rheo-, mechano-, or chemoreceptor (Lee et al., 1984; Ip and Desser, 1984; Smales and Blankespoor, 1984). In M. spinulosus, Type I papillae were concentrated around the oral sucker and increased in number as it grew up to adult fluke. The oral sucker was larger than the acetabulum in juvenile stage, but the two suckers became almost equal size in adult stage (Woo et al., 2003). The enlarged acetbulum could holdfast the matured ovigerous fluke to the intestinal mucosa and render the oral sucker to probe actively its environs for feeding (Hong et al., 1991). The Type I papillae abundant around the oral sucker are supposed to sense contacts to intestinal mucosa, flow of intestinal contents and nutrient composition (Morris and Threadgold, 1967).
Unciliated sensory papillae, similar to the Type II papillae of M. spinulosus, have been found on the oral sucker and acetabulum of several species of flukes such as H. continua, M. yokogawai, N. seoulense, Clonorchis sinensis, Echinostoma hortense and C. armatus (Fujino et al., 1979; Lee et al., 1984, 1985, 1986; Hong et al., 1991; Woo et al., 2003). Unciliated domed sensory papillae are covered with tightly packed tegument and have a bulbous extension containing a large amount of electron-dense materials (Hoole and Mitchell, 1981). From anatomical structural features, this type of sensory papillae has been suggested to be tango- and mechanoreceptive. In M. spinulosus, the oral sucker and acetabulum appeared equipped with equal number of the Type II papillae from juvenile to adult stages. Being considered with dimensions of ovigerous adult flukes, the two sucking organs of M. spinulosus may exert almost equal suctorial power upon the intestinal musoca.
On dorsal surface of M. spinulosus, the Type I papillae appeared arranged in linear symmetry, which found similar to those of the ciliated papillae of H. continua and M. yokogawai (Lee et al., 1984; Hong et al., 1991). It is suggested that the Type I papillae sense contacts and pressure of dorsal surface to the intestinal mucosa.