Adenosine 5'-triphosphate-sensitive potassium channels (KATP channels) are thought to regulate various cellular functions such as secretion as well as muscular and neural excitability by linking the cell's metabolic state to its membrane potential (Noma, 1983; Bernardi et al., 1993). KATP channels were originally discovered in the heart (Noma, 1983) and found later in various tissues, including pancreatic-cells (Rorsman et al., 1985), smooth and skeletal muscles (Standen et al., 1989), the brain (Ashford et al., 1988), pituitary glands (Bernardi et al., 1988), and the kidney (Hunter et al., 1988). It has been discovered that the KATP channels are comprised of two kinds of subunits: sulphonylurea receptors (SUR) which belong to the ATP-binding cassette family and inwardly-rectifying K+ channel (Kir) subunits which forms the potassium ion-selective pore. The channels are proposed to form a complex of SUR1 and Kir6.2 in pancreatic cells (Sakura et al., 1995; Inagaki et al., 1996), and SUR2A and Kir6.2 in cardiac and skeletal muscles (Inagaki et al., 1996). In particular, several Kir6.2 genes have been identified in human, mouse, rat and rabbit tissues, showing high mRNA levels in the heart, skeletal muscle, bladder and gut. Therefore, the KATP channel of the helminth worm is thought to be important in the cellular communication and metabolic pathway. However, the information on the study of the KATP channel for the helminth worm has been scare until now.
To add useful information for ion channel of the helminth worm, the gene encoding a pore-forming subunit of the liver fluke in the adult worm Clonorchis sinensis (CsKir6.2) was isolated and characterized. In addition, the gene expression level of a CsKir6.2 in the adult worm was investigated using a Northern blot analysis.
The total RNA was isolated from the adult of C. sinensis by guanidium isothiocyanate lysis and CsCl centrifugation at 32,000 × g for 16 hr at 20℃. A cDNA library was constructed using SMART cDNA library synthesis kit (Clontech., Palo Alto, CA, USA). Briefly, the first-strand cDNAs were synthesized using PowerScript reverse transcriptase, SMART IV oligonucleotide and a modified oligo (dT) primer (CDS III/3' PCR primer). The second-strand cDNAs were synthesized using 50 × Advantage 2 polylmerase Mix, 5' PCR primer, and a CDS/3' PCR primer. The resulting cDNA was digested with Sfi I, and fractionated using Chroma Spin-400. The cDNAs larger than 0.5 kb were pooled (data not shown). After digestion with Sfi I and size fractionation, the cDNA was ligated into the Sfi I-digested λTriplEx2 vector using T4 DNA ligase. The ligated cDNAs were packaged in Gigapack III gold packaging extract (Stratagene, La Jolla, CA, USA). To obtain a partial fragment of a specific probe, 1 gm of total RNA from the adult worm C. sinensis was reverse-transcribed using an oligo d(T)15 primer and Moloney Murine Leukemia Virus (M-MuLV) reverse transcriptase (Life Technologies, Gaithersburg, MD, USA). Two degenerate oligonucleo-tides (Kir6.2F, 5-TGACCT GGCSCCCRGCGA-3 and Kir6.2R, 5-CRTCKGC CAGGTAGGAGGT-3 S, G+C; R, A+G; K, G+T) were designed and synthesized according to the highly conserved regions of various human and rabbit ATP-sensitive inwardly-rectifying potassium channels, termed Kir6.2. The resulting cDNA was subjected to PCR amplification in a 50 µl reaction mixture containing 5 µl of RT-PCR product, 5 µl of 10 × Taq polymerase buffer, 4 µl of 2.5 mM dNTPs, 25 pM of each primers and 5 units of Taq polymerase. The reaction was subjected to 35 cycles of denaturation at 95℃ for 30 sec, annealing at 55℃ for 1 min, and extension at 72℃ for 30 sec, and one at 72℃ for 5 min in a thermal cycler (GeneAmp PCR system 9700, Perkin-Elmer). PCR with these primers and the first strand cDNA from the total RNA amplified an approximately 620 bp cDNA fragment, which then was sequenced by direct sequencing. The PCR fragment was showed the significant sequence homology with Kir 6.2 family (data not shown). An adult worm C. sinensis cDNA library of approximately 100,000 independent plaques was screened by plaque hybridization employing 620 bp partial fragment, which was amplified by RT-PCR. Hybridization to the 32P-labeled probe was done overnight at 42℃. The membrane was washed with high stringency and exposed to Kodak X-AR film. Positive clones were isolated and the lambda DNA was purified by use of a Qiagen Lambda kit (Qiagen, Valencia, CA). The cDNA insert was cloned into a pGEM-T easy vector (Promega) by PCR, using λTriplEx Sequencing primer set (Clontech) and advanced Taq polymerase with proofreading capacity (Clontech). The recombinant plasmid in the bacterial culture was purified with a Qiagen plasmid minikit (Qiagen). The nucleotide sequence was determined by the Dideoxy Chain Termination method using a Sequenase kit (ABI Prism Dye Terminator Cycle Sequencing Core Kit, Perkin Elmer) and an automated DNA sequencer (Applied Biosystems model 377A; Perkin Elmer). The determined nucleotide sequence and deduced amino acid sequence were analyzed with Basic Local Alignment Search Tool (BLAST) in National Center for Biotechnology Information (NCBI) and ExPASy Molecular Biology Server (http://www.expasy.ch)
The one clone containing the largest insert (1,189 bp in length) revealed a 999 bp complete open reading frame (ORF) coding for a protein of 333 amino acids. The 3'-untranslated region was contained poly (A)+ tail (Fig. 1). This sequence has been deposited in the GenBank database with the accession number AY277926. A hydrophobicity plot of the Kir6.2 revealed two putative transmembrane domains as found in other Kir channels. In addition, a GFG-motif was found in the putative pore-forming loop. The GFG-motif was showed in only both Kir6.1 and Kir6.2 (Inagaki et al., 1996). Although Kir6.2 shows a high degree of homology with Kir6.1, the intracellular N-terminal and C-terminal regions and an extracellular region between the first transmembrane region and the pore region (H5) do not. Therefore, the nucleotide sequence of CsKir6.2 is more similar to other known Kir6.2 than to other ion channel subunits. However, the CsKir6.2 has more unusual structural features than do other Kir6.2 genes (Fig. 2). The CsKir6.2 is smaller than Kir6.2 subunits of other species, containing a lacked 57 amino acids sequence in the N-terminal region.
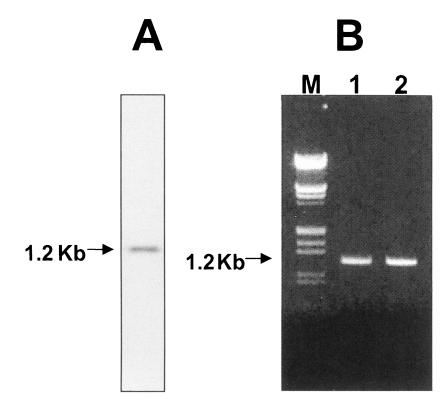
In the case of ruling out a lacked sequence region, a comparison of the amino acid sequence for CsKir6.2 with the Kir6.2 subunits of other species in the database showed a high identity to the proteins of human, mouse, rabbit, guinea pig and rat (Fig. 2). There was a difference of 13, 14, 6, 20, and 13 amino acids between the sequences of C. sinensis and the guinea pig, human, mouse, rat, and rabbit, respectively. A Northern blot was used to investigate the expression of Kir6.2 in C. sinensis. A single RNA transcript of approximately 1 kb in size were weakly expressed (Fig. 3A). In order to reveal the cause of a lacked-sequence structure in the Cskir6.2, it was amplified against genomic DNA using CsKir6.2F (5'- ATGCTGTCCCGAAAGGGCAT-3') and CsKir6.2R (5'-TCAGGACAAGGAATCTGGAG-3') primers. Samples were denatured at 95℃ for 2 min, followed by 35 cycles of 94℃ for 30 sec, 58℃ for 30 sec and 72.5℃ for 1 min. A 5 min final extension was performed at 72.5℃. PCR products were analyzed on 1% agarose gel. The nucleotide sequence was determined by dideoxynucleotide chain termination method using a sequenase kit (ABI PRISM Dye Terminator Cycle Sequencing Core Kit) and an automated DNA sequencer. Amplicon of PCR was identical to the cDNA in size (Fig. 3B) and nucleotide sequence (data not shown). Therefore, the lacked-sequence structure of CsKir6.2 is thought to be a peculiar characteristic of C. sinensis and is not isoform.
In this study, a pore-forming subunit of the KATP channel (Kir6.2 gene) of C. sinensis was cloned. This is the first report on the cloning of the Kir6.2 gene from trematodes. These results demonstrate that Kir6.2 encodes an inwardly-rectifying KATP channel protein which is expressed in adult C. sinensis. Generally, the KATP channel consists of a pore forming subunit (Kir6.2) and a sulphonylurea receptor (SUR). Both subunits are required to form a functional KATP channel. Therefore, further study is needed to address the exact functions of KATP channel by co-expressing Kir6.2 and SUR after the SUR gene is cloned in C. sinensis.