Immune Response of BALB/c Mice toward Putative Calcium Transporter Recombinant Protein of Trichomonas vaginalis
Article information
Abstract
Trichomoniasis is a common sexually transmitted infection caused by Trichomonas vaginalis, which actually does not exist a vaccine for control or prevention. Thus, the identification of new and potent immunogens in T. vaginalis, which can contribute to the development of a vaccine against this parasite, is necessary. Therefore, the aim of this work was to evaluate the potential of a recombinant Transient Receptor Potential-like channel of T. vaginalis (TvTRPV), as a promising immunogen in BALB/c mice. First, TvTRPV was cloned and expressed as a recombinant protein in Escherichia coli BL21 cells and purified by nickel affinity. Next, BALB/c mice were immunized and the antibody levels in mice serum and cytokines from the supernatant of macrophages and from co-culture systems were evaluated. Recombinant TvTRPV triggered high levels of specific total IgG in sera from the immunized mice. Also, a statistically significant increase of cytokines: IL-1β, IL-6, and TNF-α after stimulation with the corresponding antigens in vitro, was identified. Moreover, co-cultures using CD4+ T cells from immunized mice were able to identify higher levels of IL-10 and IFN-γ. These results were useful to validate the immunogenicity of TvTRPV in BALB/c mice, where IL-10-IFN-γ-secreting cells could play a role in infection control, supporting the potential of TvTRPV as a promising target for vaccine against T. vaginalis.
Trichomoniasis is the most common non-viral sexually transmitted infection in the world, caused by the protozoan parasite Trichomonas vaginalis with an incidence of 276 million new cases each year [1]. Trichomoniasis can increase the risk of getting or spreading other sexually transmitted infections, such as human immunodeficiency virus (HIV) or human papilloma virus (HPV) [2,3]. With an increasing incidence of infection and a possible non-effective treatment, the prevention of this disease with a vaccine would clearly be desirable [4]. Until now, work on the development of T. vaginalis vaccines is still mainly focused on the screening of parasite-derived immunogens capable of inducing both humoral and cellular immune responses [5,6].
Some microbial surface proteins are considered as potential immunogens. Specifically, ion transporters from diverse microorganisms have been reported that they may be suitable targets for development of live attenuated vaccines, exhibiting superior protective immunity compared with commercial vaccines [7,8]. In addition, it has been described that components of ionic transporters can induce antibodies that promote specific immune responses rather than inhibiting ionic transport [9,10].
Transient Receptor Potential (TRP) family of ion channels serve as cellular sensors for a wide spectrum of physical and chemical stimuli [11]. Prole and Taylor examined several protozoan parasite genomes and found that only T. vaginalis had a unique homologue of TRPV channels (TvTRPV), which was predicted to exist on membrane surface of this parasite [12]. Some members of the TRP family have been shown to be associated with inflammatory processes and immune responses [13]. Recently, it was identified that immunization of mice with a variety of antigens expressing TRPA1 channels resulted in the generation of monoclonal antibodies that could act as selective antagonists [14].
Taking into account the immunogenic potential described in some ionic transporters, the association of some members of the TRP family with immune f location of TvTRPV in T. vaginalis, as well as its important role in different cellular processes, TvTRPV shows potential to be an immunogenic protein and the present study reports the humoral and cellular immune response of BALB/c mice immunized with the recombinant TvTRPV protein from T. vaginalis.
For this work, total RNA isolation from T. vaginalis trophozoites and cDNA synthesis were carried out as reported previously [15]. TvTRPV gene (Genbank Accession: XP_001296819) was amplified from cDNA by PCR using specific primers and cloned into the pCold-II (Takara, Otsu, Shiga, Japan) prokaryotic expression vector, by insertion at BamHI and HindIII restriction sites. Transformed E. coli BL21 (DE3) (Promega, Madison, Wisconsin, USA) cells were grown in LB at 37°C and protein expression was induced at OD600 of 0.4 with 1 mM isopropyl-β-D-1-thiogalactoside (IPTG) (Promega, Madison, Wisconsin, USA) for 24 hr. Culture conditions were optimized to achieve a recombinant protein concentration equal to approximately 1 mg/ml culture medium. After induction, the protein was purified from the soluble fraction using His-spin protein miniprep kit (Zymo Research, Irvine, California, USA) following the manufacturer’s instructions, except that all steps were performed at 4°C and increased to 5 washes prior the elution step.
Once obtained the recombinant protein, 30 male BALB/c mice were randomly divided in groups of 5 and were immunized subcutaneously: 3 experimental groups were immunized with recombinant TvTRPV at doses of 50, 100, and 200 μg/kg (TvTRPV50, TvTRPV100, and TvTRPV200, respectively), and 2 control groups, 1 immunized with Incomplete Freund’s Adjuvant (IFA) (Santa Cruz Biotechnology, Dallas, Texas, USA) and a second unimmunized control (C−). Two booster injections were given in 2-week interval.
Blood was collected from the mice by tail vein puncture before each immunization and to analyze the humoral response to anti-recombinant TvTRPV we measured antigen-specific IgG antibody levels in sera by enzyme-linked immunosorbent assay (ELISA). Microtiter plates were coated with the recombinant protein (1 μg/ml, 100 μl/well) overnight at 4°C in carbonate buffer (0.05 M carbonate-bicarbonate, pH 9.6). The non-specific sites were blocked with blocking buffer (Peprotech, London, UK) for 2 hr at room temperature. Serum samples from mice were diluted (1:25) in diluent buffer 1X (Peprotech), were loaded to each well and then incubated for 2 hr at room temperature. Anti-mouse IgG conjugated to HRP (Santa Cruz Biotechnology) were diluted (1:1,500), added to each well and incubated for 2 hr at room temperature. Later, ABTS was added and incubated for 30 min in the dark at room temperature. Immediately, the absorbance value of wells was read using an ELISA reader (Bio-Rad iMark, California, USA) at a wavelength of 450 nm. Triplicate biological replicates of samples from each experimental group were evaluated.
We also examined whether recombinant TvTRPV induces macrophages to secrete proinflammatory cytokines by antigen stimulation peritoneal macrophages (MØs), which were isolated from mice as described previously [16]. MØs were seeded at 1×105 cells/ml/well in 24-well plate and incubated with recombinant TvTRPV protein at 1, 10, and 100 μg/ml in complete medium (DMEM supplemented with 10% FBS and 1% penicillin-streptomycin) for 48 hr (37°C, 5% CO2). Macrophages without treatment were used as negative control (C−) and activated macrophages with LPS (1 μg/ml) were employed as a positive control. The maturation of macrophages was confirmed by microscopic monitoring the change in shape by the increment of the cytoplasmic volume and granularity.
We also evaluated the antigenic specific cellular immune response using CD4+ T cells which were co-incubated with mice macrophages treated with recombinant TvTRPV. For this coculture system, mice were euthanized 4 weeks after the last boost and CD4+ T-cells were isolated from mouse spleens as described previously [17]. Cell suspension was prepared in 90 μl of isolation buffer per 1×107 total cells. Resuspended CD4+ T-cells were purified by negative selection by using CD4+ T-cells isolation kit (Miltenyi Biotec, Bergisch Gladbach, Germany) according to the manufacturer’s instructions. CD4+ T cells were incubated with macrophages previously obtained, in the following proportions: 1 CD4+ T cell for each 2 macrophages (1:2). Cells were grown in this co-culture system for 72 hr (37°C, 5% CO2) in a 24-well plate for IFN-γ production.
Cell culture medium of treated and activated macrophages (for IL-1β, IL-6, and TNF-α detection) and co-culture systems of macrophages and CD4+ T cells (for IFN-γ and 1L-10 evaluation) were analyzed through commercial ELISA kits according to the manufacturer’s instructions (Peprotech). Microtiter plates were coated with capture antibody (1 μg/ml, 100 μl/well) overnight at 4°C for IL-1β, IL-6, TNF-α, IFN-γ and 1L-10 evaluation. The non-specific sites were blocked with blocking buffer (1% BSA in PBS) for 1 hr at room temperature. Serial dilutions of recombinant cytokines were used as standard curve in each case, as recommended by the manufacturer. The cytokines present in the supernatants were detected by adding biotinylated avidin-HRP conjugated to detection antibody (Peprotech) and incubated overnight. The absorbance values at 490 nm were determined using an ELISA plate reader (Bio-Rad iMark) and the cytokines concentration was determined by comparison of the absorbance values in the corresponding standard curve.
The present study was approved by the ethics committee of the Campus of Agricultural and Biological Sciences (CB-CCBA-M-2016–005) at Autonomous University of Yucatan, Mexico, and conducted according to the guidelines outlined in the declaration of Helsinki.
A 711 bp segment corresponding to the TvTRPV gene was amplified from cDNA, cloned into pCold-II expression vector and expressed as a polyhistidine fusion protein, observed as a band about 27.4 kDa in an SDS-PAGE only from soluble fraction after 24 hr of induction. The amount of purified recombinant TvTRPV was estimated as 0.7 mg/ml (Fig. 1).

Prokaryotic expression and purification of recombinant TvTRPV. 0 hr, E. coli BL21 not induced; 24 hr, induction time after adding 1 mM IPTG; IF, insoluble fraction of induced cells; SF, soluble fraction of induced cells; W1 and W2, washings E1 and E2, TvTRTPV protein obtained by elution; MW, molecular weight marker.
The humoral response to anti-recombinant TvTRPV show that antigen-specific IgG antibody response was significantly higher in the mice immunized with recombinant TvTRPV (TvTRPV50, TvTRPV100, and TvTRPV200 groups) at 2 weeks after the first immunization, compared to the control groups (IFA and C−) (Table 1).
We detected high levels of IgG in mice immunized with recombinant TvTRPV (TvTRPV50, TvTRPV100, and TvTRV200 groups), which showed a concentration-dependent increase following booster immunizations (week 4 and 6, respectively). Although TvTRPV50, TvTRPV100, and TvTRPV200 groups showed a gradual increase in the production of IgG antibodies at the different times evaluated, we did not observe statistical differences between the 3 groups, except for TvTRPV200 group that showed a significant increase (P <0.05) after the first and second booster immunizations with respect to the IgG levels detected in IFA group (Table 1). These results suggest that in BALB/c mice, recombinant TvTRPV stimulates a high IgG response. The data also suggest that the booster immunization of the highest dose enhances the immune response induced by this recombinant protein.
We also examined whether recombinant TvTRPV induces macrophages to secrete proinflammatory cytokines. For this purpose, macrophages were stimulated in vitro with 1, 10, and 100 μg/ml of recombinant protein. LPS-stimulated macrophages were used as a positive control, and unstimulated macrophages as negative control. The obtained results showed that macrophages incubated with different doses of recombinant TvTRPV stimulated the production of IL-1β, IL-6, and TNF-α, showing a concentration-dependent increase of the recombinant TvTRPV in the secretion of these proinflammatory cytokines. The levels of IL-1β and IL-6 detected in recombinant TvTRPV-stimulated macrophages displayed a significant increase with respect to the negative control and the levels of TNF-α detected in macrophages stimulated with the highest dose of the recombinant protein (100 μg/ml) presented statically significant difference with respect to the unimmunized control (C−) (Fig. 2). These results indicate that the recombinant TvTRPV is recognized by antigen-presenting cells, such as macrophages, with the subsequent stimulation in the secretion of proinflammatory cytokines.
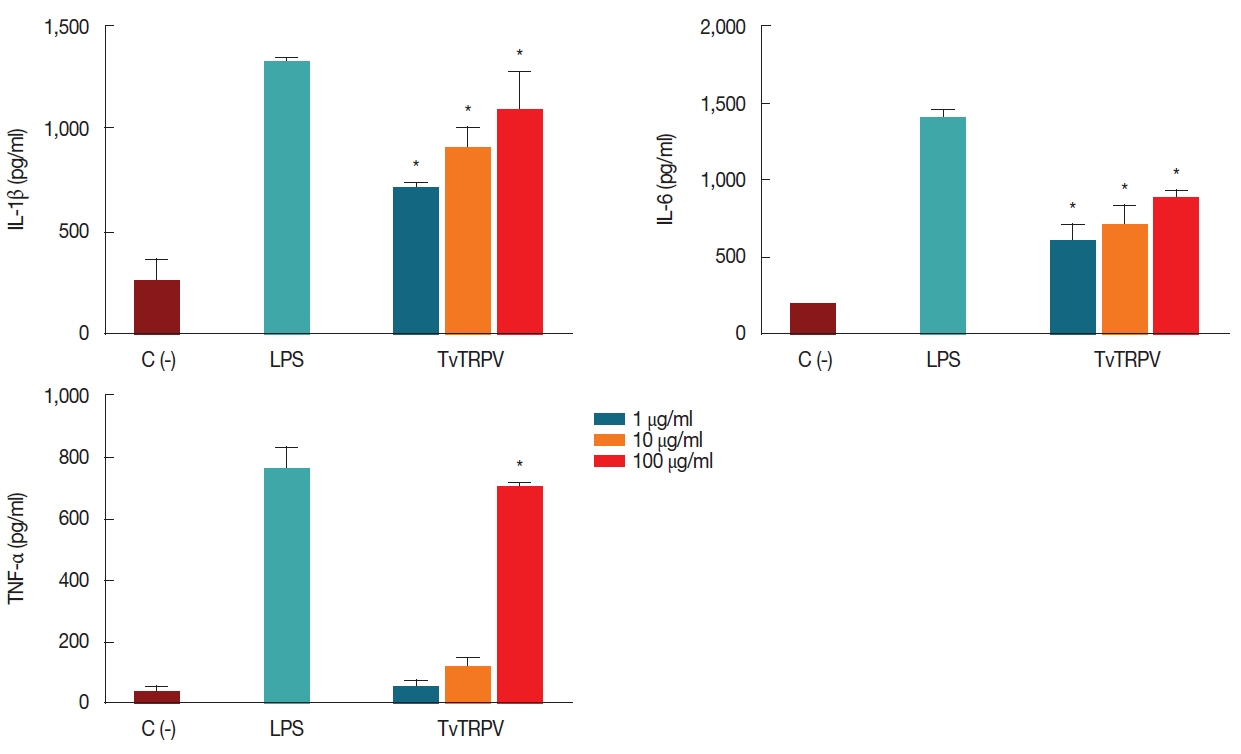
Effect of recombinant TvTRPV on the macrophages 1L-1β, IL-6 and TNF-α production. Each bar represents the mean value±standard deviation (SD). Significance was accepted by comparison with the unimmunized control: (C-), (Dunnett, *P <0.05). Macrophages stimulated with LPS (1 μg/ml) were used as positive controls for cytokine production.
On the other hand, when the obtained CD4+ T cells were co-incubated with mice macrophages treated with recombinant TvTRPV, they secreted detectable levels of IFN-γ and IL-10. In vitro treatment of CD4+ T cells from recombinant TvTRPV-immunized mice stimulated with macrophages previously treated with different doses (1, 10, and 100 μg/ml) of this recombinant protein triggered significantly higher amounts of IFN-γ when compared to the level of this cytokine produced by CD4+ T cells stimulated with macrophages without treatment (C−) (Fig. 3). Also, the levels of IL-10 detected in CD4+ T cells from recombinant TvTRPV-immunized mice with the highest doses (100 and 200 μg/kg) and that were stimulated with macrophages treated with 100 μg/ml of recombinant TvTRPV were significantly higher in comparison with those produced by CD4+ T cells co-cultured with untreated macrophages (Fig. 3). These results showed that recombinant TvTRPV was able to induce a broad range of cytokines such as of IL-1β, IL-6, TNF-α, IL-10, and IFN-γ. These latter deserve particular mention, since were produced by CD4+ T cells, a regulatory population previously described and named IL-10-IFN-γ-secreting CD4+ T cells.
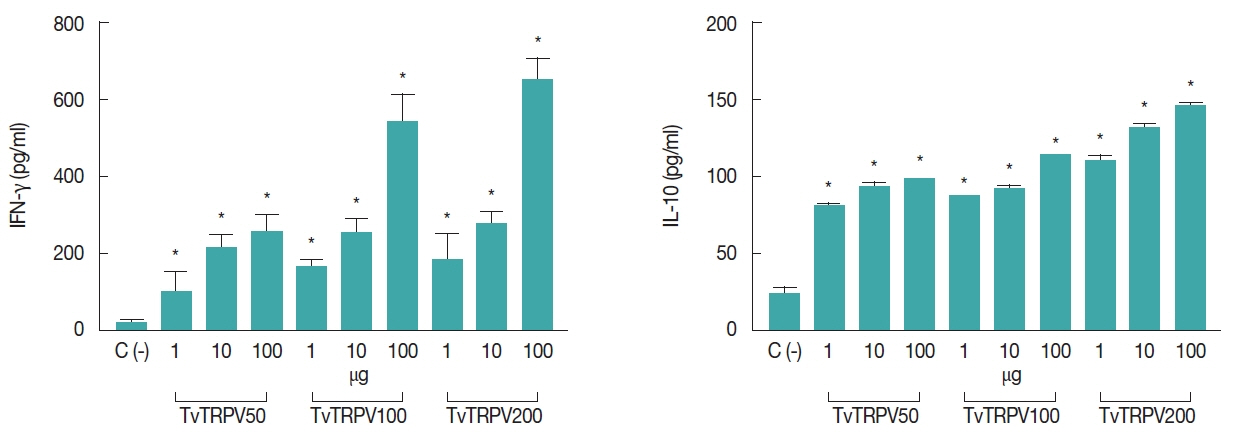
IFN-γ and IL-10 levels detected in co-culture system. Purified CD4+ T cells from immunized mice with different doses of recombinant TvTRPV (TvTRPV50, TvTRPV100, and TvTRPV200) stimulated with macrophages treated with different doses of recombinant TvTRPV (1, 10, and 100 μg). Co-culture systems were set up with 1:2 CD4+ T-cell:MØ. Each bar represents the mean value±standard deviation (SD). Significance was accepted by comparison with CD4+ T cells stimulated with macrophages without treatment: C (-), (Dunnett, *P<0.05).
To evaluate the immunological potential of a protein, the study of the humoral immune response is one of the most important parameters. In this work, recombinant TvTRPV derived from T. vaginalis triggered the production of IgG antibodies against this protein. Our results showed significantly higher IgG levels in mice immunized with the highest dose of recombinant TvTRPV protein (200 μg/kg) in all the times evaluated. In addition, the low and intermediate doses (50 and 100 μg/kg) were capable to stimulate the production of IgG antibodies. These results are consistent with previous reports, in which T. vaginalis isolates were used to produce whole-cell vaccines and higher levels of IgG were detected in serum of mice after vaccination compared with controls [18,19].
Although the protective role of antibodies during T. vaginalis infection is unclear and there is little evidence that antibodies are effective in the elimination of parasites in vivo [1], a previous report in which mice were immunized with low and high doses of recombinant α-actinin subunits, showed IgG antibody titers related to protective effect. These results suggest that a protective effect against T. vaginalis could possibly be related, at least in part, to a humoral response [5]. However, further studies are required to determine if the antibodies produced against recombinant TvTRPV provide a protective effect against the infection caused by T. vaginalis.
On the other hand, different components of the innate immune response could be participating in defense mechanisms against T. vaginalis. In trichomoniasis it has been suggested that TNF-α, IL-1β (known chemotactic mediators involved in inflammation), and IL-6 produced by macrophages stimulated with T. vaginalis may play a crucial role in vivo as mediators of neutrophilic tissue recruitment at the infected sites in women [20]. In this study it was demonstrated that recombinant TvTRPV was able to stimulate effector functions of murine macrophages, finding a significant increase in the levels of IL-1β, IL-6 and TNF-α, compared with non-stimulated macrophages, which, interestingly, coincides with the reports mentioned above, suggesting that TvTRPV protein from T. vaginalis could play a critical role in the initiation of the inflammatory process during the infection establishment.
These results are in agreement with previous reports in which both Trypanosoma cruzi and Leishmania major have been shown to stimulate the increased secretion of cytokines such as TNF-α, IL-6, among others, from mice macrophages [21,22]. Besides humoral and innate response, the cellular immune response is also an important index for detection of immunogenic potential of proteins. For this purpose, we determined the level of Th1/Th2 cell related cytokines (IFN-γ and IL-10) in the supernatant of co-culture systems using CD4+ T cells from spleen following immunization and re-stimulation in vitro. CD4+ T-helper cells may play a prominent role in parasite elimination, parasite persistence or pathological events during T. vaginalis infection [1,23].
Our findings showed that recombinant TvTRPV-activated macrophages promote the secretion of significantly higher levels of IFN-γ and IL-10 in co-cultures with CD4+ T cells from mice immunized with this recombinant protein, than those from the unimmunized control. It has been demonstrated that IFN-γ increase macrophage-mediated cytotoxicity against T. vaginalis and also it is suggested that participate in the elimination or suppression of proliferation of T. vaginalis by stimulation of nitric oxide production as one of the effectors [24], while IL-10 is known to initiate an anti-inflammatory feedback loop by the cooperative action with other immune cells [25].
The obtained results demonstrate that recombinant TvTRPV after being recognized by macrophages and presented to the T cells clones producing IFN-γ and IL-10, suggesting that cellular response against this protein could be related to a protective immunity against the protozoan parasite. The characteristics and mechanisms of antibodies and cytokines induced by this protein would provide valuable knowledge for alternative therapeutics against trichomoniasis. Therefore, further experiments are necessary in order to elucidate the molecular mechanisms by which TvTRPV exerts a cellular and humoral response in mice and if these have a protective effect on the infection caused by T. vaginalis.
ACKNOWLEDGMENT
This work was undertaken as part of a research project supported by Grant 237990 (to J.C. Torres-Romero) from Consejo Nacional de Ciencia y Tecnología (CONACYT), México. T. Oliveros-Mendoza was a scholarship recipient from CONACYT.
Notes
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
