Infections of Two Isthmiophora Species (Digenea: Echinostomatidae) in Wild Mammals from Republic of Korea with Their Morphological Descriptions
Article information
Abstract
Isthmiophora hortensis (Digenea: Echinostomatidae) is a dominant echinostome in animal reservoir hosts and humans in the Republic of Korea (Korea). We intended to investigate the infection status with this echinostome species in the several species of wild animals and describe the morphological characteristics in the faunistic view point. A total of 175 animal carcasses belonging to 3 families, i.e., Canidae, Felidae and Mustelidae, were collected from the southern regions of Korea from March 2010 to July 2017. Isthmiophora spp. worms were recovered from the small intestines of each animal under a stereomicroscope after washing of intestinal contents. Isthmiophora hortensis was recovered from 4 species of wild carnivores, i.e., Nyctereutes procyonoides (3/107: 2.8%), Mustela sibirica (11/31: 35.5%), Meles lucurus (2/3: 33.3%) and Martes flavigula (1/2: 50%). The other 3 carnivores comprising stray dogs, cat and leopard cat were negative for I. hortensis infection (0/2, 0/10 and 0/12, respectively). Specimens obtained from the Lutra lutra (6/8: 75%) were identified as a distinct species, I. inermis, by morphological comparison. Isthmiophora inermis has thinner body, elongate testes and different anterior limits of vitelline fields. Detailed morphological descriptions and comparisons with the morphological characteristics are provided. Conclusively, it was confirmed for the first time that 3 species of mustelid mammals, i.e., M. sibirica, M. lucurus, and M. flavigula, are to be the new definitive hosts of I. hortensis in Korea. Additionally, I. inermis is to be newly added in the Korean echinostome fauna.
INTRODUCTION
Isthmiophora hortensis (Asada, 1926) (Digenea: Echinostomatidae) is an echinostome trematode species characterized by the possession a collar with 27 spines and has medical and veterinary importance. Human infection with I. hortensis has been reported frequently in East Asian countries such as China, Japan and the Republic of Korea (=Korea) [1].
Due to its public health importance, the life cycle of the species has been studied extensively. Freshwater snails of the family Lymnaeidae Rafinesque, 1815 are known to serve as first intermediate hosts [2–4], while freshwater fishes and frogs have been reported as second intermediate hosts [3,5–10]. Additionally, a wide range of small mammals are known as respective definitive hosts, such as the rat (reported in Japan by Asada, 1926) [21], black-striped field mouse Apodemus agrarius (Pallas), Manchurian reed vole Microtus fortis pelliceus Thomas, domestic mouse Mus musculus L., brown rat Rattus norvegicus (Berkenhout), black rat R. rattus L., Daurian pika Ochotona dauurica (Pallas), domestc dog Canis lupus familiaris L., domestic cat Felis catus L., Japanese weasel Mustela itatsi Temminck (=Lutreola itatsi itatsi) and raccoon Procyon lotor L. [12–25]. Recently, Sohn et al. reported two additional species, namely, the raccoon dog Nyctereutes procyonoides (Gray) and wild boar Sus scrofa L., to the list of known definitive hosts of I. hortensis [23].
Ishtmiophora inermis (Fuhrmann, 1904) is one of 6 species belonging to the genus Isthmiophora. This species is morphologically very similar to I. hortensis, but has thinner body, elongate testes and different anterior limits of vitelline fields [26]. Moreover, this species has a somewhat different host range than I. hortensis. Unlike the hosts of I. hortensis as we mentioned above, this species has been found mainly in otters i.e., Lutra sp., L. lutra and Lontra canadensis (Schreber, 1777) [18,27–32]. Eurasian otter has been found in Korea as well, but the distribution of I. inermis is still unknown until present study.
Considering the life cycle of Isthmiophora flukes and their known hosts, wild mammals and especially carnivorous or omnivorous animals such as mustelids seem to be suitable hosts for I. hortensis. However, their infection status in Korea is not well known. Therefore, the present study aimed to investigate the infection status of I. hortensis for wild mammals in Korea and to determine the range of its definitive hosts. The present study provides reports on yellow-throated marten Martes flavigula (Boddaert), Aisan badger Meles lucurus (Hodgson) and Siberian weasel M. sibirica Pallas as new host records of I. hortensis, and presents a detailed overview of its infection status in Korea. In addition, we include detailed morphological characteristics of I. inermis, found from the intestines of Eurasian otter Lutra lutra, as a new member of the Korean parasite fauna.
MATERIALS AND METHODS
Collecting specimens
Carcasses of wild carnivores were collected from March 2010 to July 2017. A total of 175 wild carnivores belonging to three families comprising Canidae, Felidae and Mustelidae were collected and examined. Collection, identification and necropsy of the host animals were conducted with the support of several wild animal rescue centers or wildlife related institutes, namely, the Wildlife Center of Chungbuk (WCC), Cheongju-si, Chungcheongbuk-do, Korea, the Chungnam Wild Animal Rescue Center (CNWARC), Yesan-gun, Chungcheongnam-do, Korea, the Korean National Park Service Species Restoration Technology Institute (KNPSSRTI), Gurye-gun, Jeollanam-do, Korea and the Conservation Genome Resource Bank for Korean Wildlife (CGRB), Seoul, Korea, as well as small-scale college laboratories. All of the examined hosts were collected from the Korean peninsula.
Examined hosts included stray dogs C. lupus familiaris L. (n=2), Korean raccoon dog N. procyonoides koreensis (Gray) (n=107), stray cats F. cactus L. (n=10), Amur leopard cats Prionailurus bengalensis euptilurus (Elliot) (n=12), Siberian weasels M. sibirica (n=31), Eurasian otters L. lutra L. (n=8), Asian badgers M. lucurus (n=3) and yellow-throated martens M. flavigula (n=2). Most of the animals were dead during rescue/care in the wildlife rescue center, or found on the road as a carcass.
The visceral organs were extracted from each dead body, and the lumen was exposed by a longitudinal incision. Isthmiophora spp. were recovered from the visceral organs using the filtration method [36]. Parasite numbers were then recorded and recovered specimens were stored in fixative agents (70% ethanol or 10% neutral buffered formalin) for further research. Detailed information concerning infection status and locality of positive cases is provided in Table 1.
Morphological identification
Identification of species was based on the key to the species of Isthmiophora provided by Kostadinova and Gibson [26]. Two species, I. hortensis and I. intermis, were distinguished using morphological observations. Based on the morphological condition of specimens and the representativeness among the morphological differences, 12 I. hortensis and 12 I. inermis were chosen for morphological observation. Specimens of I. hortensis (host code no. B417) was obtained from a M. sibirica collected from Cheongju (city) in 2011 and I. inermis (host code no. B1330) was obtained from a L. lutra collected from Cheongju in 2015. They were stained with Semichon’s acetocarmine and observed under light microscopy on a mounted slide in Permount (Fisher Scientific) [33]. The voucher specimens were deposited at the National Institute of Biological Resources (NIBR), Incheon Metropolitan City, Korea and the Parasite Resource Bank (PRB), Cheongju-si, Chungcheongbuk-do, Korea.
Abbreviations used in the present study are as follows: BL: body length; BW: maximum body width; CL: collar length; CW: collar width; NCS: number of collar spines; OSL: oral sucker length; OSW: oral sucker width; PL: prepharynx length; PHL: pharynx length; PHW: pharynx width; ESL: esophagus length; CSL: cirrus sac length; CSW: cirrus sac width; SVL: seminal vesicle length; SVW: seminal vesicle width; VSL: ventral sucker length; VSW: ventral sucker width; OVL: ovary length; OVW: ovary width; MEL: Mehlis’ gland length; MEW: Mehlis’ gland width; ATL: anterior testis length; ATW: anterior testis width; PTL: posterior testis length; PTW: posterior testis width; EL: egg length; EW: egg width; FO: forebody length (distance from the anterior end to the anterior margin of the ventral sucker); U: uterine field length (distance from the anterior margin of the ovary to the posterior margin of the ventral sucker); and T: post-testicular field length. The size and proportions of standard structures relative to body length were calculated as described by Kostadinova [34]. Measurements are shown in Table 2.
RESULTS
Among the 175 hosts examined, 23 animals were positive for Isthmiophora flukes (13%). Isthmiophora flukes were found in all mustelid hosts and included Siberian weasel (11/31, 35.5%), Asian badger (2/3, 33.3%), Eurasian otter (6/8, 75%) and yellow-throated marten (1/2, 50%). Furthermore, only 3 of the 107 raccoon dogs (2.8%) were infected with Isthmiophora flukes. Dogs, cats, and leopard cats were negative for these flukes. However, the flukes that were found in these animals are thought to be I. hortensis. Moreover, morphological examination revealed that all the Isthmiophora flukes found in Eurasian otters were I. inermis. Detailed information concerning infection status and locality of positive cases is provided in Table 1.
Descriptions in the present study: Family Echinostomatidae Looss, 1899, Genus Isthmiophora Luhe, 1909, Isthmiophora hortensis (Asada, 1926) Kostadinova and Gibson, 2002
Host: Martes flavigula, Meles lucurus, Mustela sibirica, Nyctereutes procyonoides
Localities: Chungcheongbuk-do (Cheongju-si), Chungcheong-nam-do (Boryeong-si, Cheongyang-gun, Hongseong-gun, Sejong-si, Taean-gun and Yesan-gun) and Ulsan (Ulju-gun); Republic of Korea.
Description (Fig. 1, n=12): Body thin, foliated, dorso-ventrally flattened. Maximum body width at the level of ovary. Forebody short to long (FO/BL=12–19%). Tegumental spine present, densely distributed in forebody but less dense in hind body. Collar reniform, small. Ventral lappets poorly developed. Collar spines 27 in number, angle spines aligned in double row (2 ventral and 2 dorsal), lateral spines in single row, and dorsal spines in double row. Oral sucker subterminal, subspherical, muscular. Prepharynx present. Pharynx muscular, elongate-oval. Oesophagus short. Intestinal bifurcation in front of ventral sucker. Caeca blind, narrow, overlapped by vitelline follicles, almost reaching posterior end. Ventral sucker subspherical, well-developed, muscular.
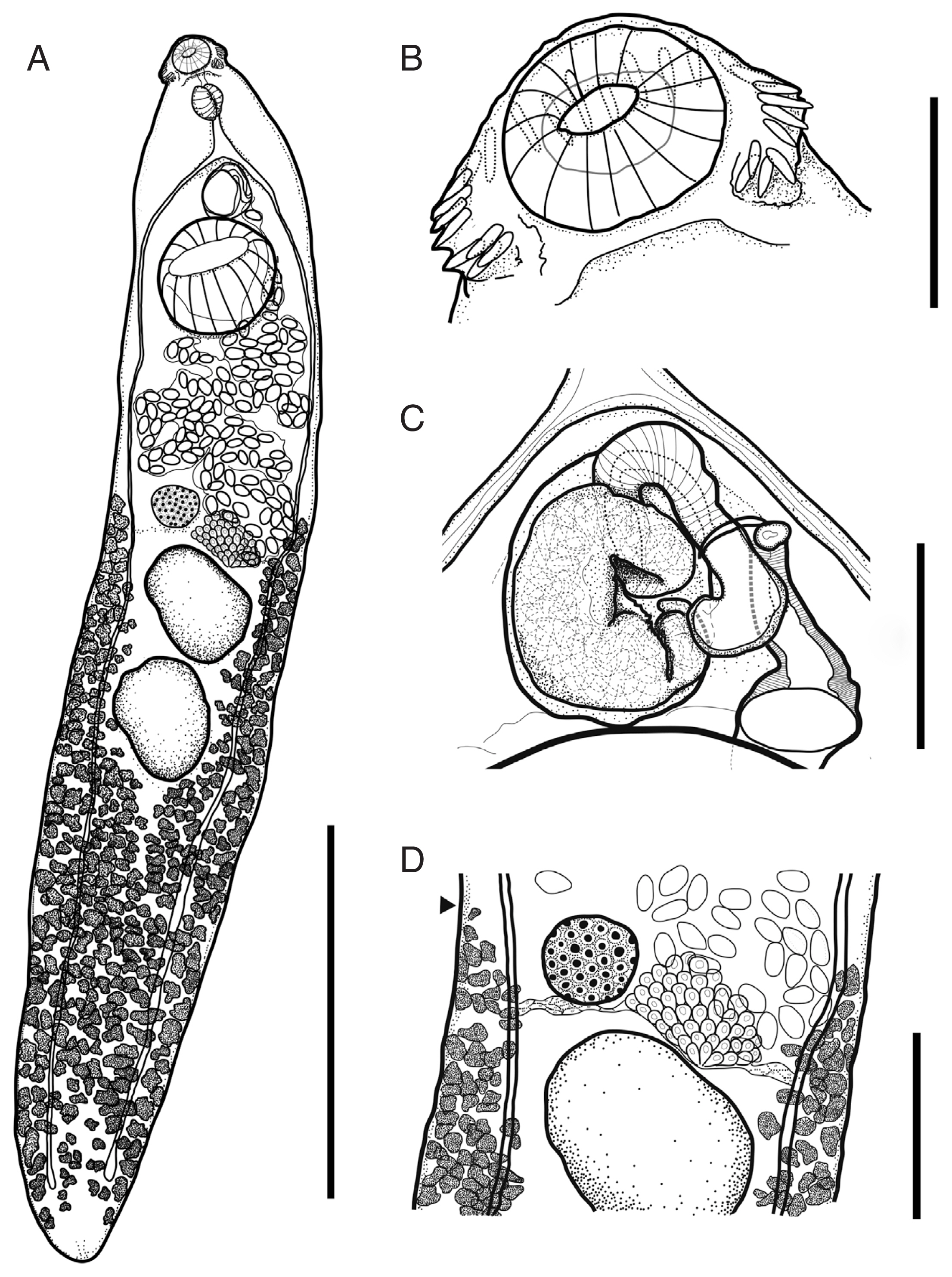
Descriptions of Isthmiophora hortensis (Asada, 1926) recovered from a Siberian weasel Mustela sibirica Pallas. (A) Entire worm, (B) Head collar. (C) Terminal genitalia, (D) Body part showing the anterior distribution of vitelline follicles (black arrowhead) Scale bars: (A) 2 mm. (B–C) 200 μm. (D) 500 μm.
Testes 2, tandem, large, oval to irregular shaped, located in equatorial region of body. Post testicular region long (T/BL=36–47%). Cirrus sac oval, muscular, located antero-dorsal to ventral sucker, included elongated seminal vesicle with curved pars prostatica. Cirrus tubular, armed with spines. Genital pore located posterior to intestinal bifurcation.
Ovary spherical to oval, dextral to median, close to ventral sucker (U/BL=5–15%). Mehlis’ gland large, sub-median, contiguous but slightly behind ovary. Uterus intercaecal, short. Eggs oval, yellowish. Vitelline follicles small, distributed from the level of ovary to posterior extremity, occupy each side of the body and overlapped at post testicular field. Excretory vesicle Y-shaped.
Isthmiophora inermis (Fuhrmann, 1904) Kostadinova and Gibson, 2002
Host: Lutra lutra
Localities: Chungcheongbuk-do (Cheongju-si and Chungju-si), Chungcheongnam-do (Yesan-gun), and Jeollanam-do (Gurye-gun); Republic of Korea
Descriptions (Fig. 2, n=12): Body thin, elongated and cylindrical (BW/BL=10–12%). Maximum body width at level of anterior testis or ventral sucker. Forebody short (FO/BL=11–17%). Tegumental spine obscure.
Collar weakly developed, armed with 27 spines. Ventral lappets poorly developed. Oral sucker subterminal, subspherical, muscular. Prepharynx present. Pharynx muscular, elongate-oval. Oesophagus relatively short, bifurcated slightly at anterior region of the ventral sucker. Ventral sucker large, subspherical, muscular, well-developed.
Testes 2, tandem, elongated, oval shaped. Post testicular region very long (47–55%). Cirrus sac located between intestinal bifurcation and ventral sucker, oval, muscular, includes elongated, curved seminal vesicle with convoluted and long pars prostatica. Cirrus tubular. Genital pore located posterior to intestinal bifurcation.
Ovary spherical, entire, dextral, located somewhat in spaces anterior to anterior testis. Mehlis’ gland well-developed, median. Uterus short (U/BL=6–10%), filled with eggs. Metraterm muscular. Vitelline follicles well-developed, covering region starting from posterior end to near anterior border of anterior testis.
DISCUSSION
Morphological observations of stained slide specimens representing the two groups of Isthmiophora flukes indicated that the characteristics of specimens matched well with the diagnostic characteristics of the genus Isthmiophora [26,34]. All specimens had 27 collar spines and exhibited the characteristics regarded as the major attributes of the genus Isthmiophora as noted by Kostadinova and Gibson [26]. Observations revealed a slightly greater anterior position of the testes (T/BL=36–55%), armed cirrus, small head collar with not well-developed ventral lappets, longer oral spines than aboral, short forebody (FO/BL=11–17%) and uterus (U/BL=5–14%), and large eggs.
According to the differential keys of Isthmiophora suggested by Kostadinova and Gibson [26], our specimens were identified as comprising two Isthmiophora species, I. hortensis and I. inermis. All specimens collected from animal hosts investigated in this study, with the exception of Eurasian otters, had morphological characteristics identical to those of I. hortensis, which was the only species reported previously in Korea.
I. hortensis is regarded as very similar morphologically to I. melis, and the only difference between the two species is the position of cirrus sac (located between intestinal bifurcation and ventral sucker or antero-dorsal to the ventral sucker). Recently, Sohn et al. investigated body indices of I. hortensis obtained from 4 species of terrestrial mammals, namely, A. agrarius, F. cactus, N. procyonoides and S. scrofa, and compared results with those of experimentally infected albino rats (metacercariae were obtained from the Korean dark sleeper, Odontobutis platycephala Iwata & Jeon) in Korea [23]. They also noted that I. hortensis has a high similarity with I. melis and that it is very difficult to distinguish the two species, which can only be accomplished using a morphological aspect such as body indices. Our results also indicate that body indices are very similar to those of I. melis; however, our observations indicate that the position of the cirrus sac is similar to that reported by Kostadinova and Gibson [26].
I. inermis can be distinguishable from I. hortensis by its slender body, rather elongated testes and anterior limits of the vitelline fields at the level of the anterior testis [26]. These features can also be checked in our specimens collected from L. lutra. Additionally, morphometric comparisons supported the species identifications (Table 2). Observations showed that specimens of both species did not differ much since measurements of size overlapped for most body parts. However, the measurements support the distinction of species based on differences in the ratios of body parts. In comparison with I. hortensis, I. inermis had a slender body, BW/BL: 9–12% (11%) and longer post-testicular fields, T/BL: 47–55% (50%). The BW and T ratio relative to the BL of I. hortensis were 11–19% (15%) and 36–47% (41%), respectively (Table 2).
The specimens collected from Lutra lutra were all identified as I. inermis, which had never been reported before as a member of the Korean parasite fauna. Isthmiophora inermis was primarily described as Euparyphium inerme in a Lutra sp. from Java, Indonesia [27], but Kostadinova and Gibson [26] considered this species as a member of the genus Isthmiophora and transferred it from the genus Euparyphium to Isthmiophora. As in the present study, I. inermis has been mainly detected from otters (Lutra sp., and L. lutra) in Eurasian countries such as China, Indonesia (Java), the Russian Far East and Vietnam [18,27–31]. Additionally, several suspicious findings of this species were recorded from North America [32]. The only known case of a non-mustelid host involved dogs in India [35]. Our results suggest that the species could be found along with the distribution of its main host L. lutra, and Korea is a new location for its distribution.
The generic conception of the genus Isthmiophora as re-defined by Kostadinova and Gibson [26] provides insights into the life history and host range of species identified in this study by comparison with other members of the genus. Generally, they have been known to use mammals as definitive hosts [26,34]. Among the known host species, mustelids represent a large portion of their host diversity. With the exception of two species having 29 collar spines, I. lukjanovi Chertkova, 1971 [36] and I. citellicola Kadenatsii in Skrjabin & Bashkirova, 1956 [37], which were only reported from Gazella subgutturosa and Citellus pygmaeus, the other 4 species are known to use at least one species of mustelid. The type species I. melis was reported from over 10 species of mustelids [29], and otters are known to act as definitive hosts of I. inermis [27–32]. A North American species, I. beaveri, was reported from a mustelid host, Neovison vison (Schreber) (=Mustela vison), and also from experimentally infected ferrets [26,38,39]. Several authors have noted the importance of mustelids as hosts of Isthmiophora flukes [13,40,41].
Isthmiophora hortensis has been known to use mammals as its definitive host. It is our understanding that the host list includes 11 animals belong to 6 families as follows: Canidae (Canis lupus and N. procyonoides), Felidae (F. catus), Ochotonidae (Ochotona dauurica), Muridae (A. agrarius, R. norvegicus, R. rattus, fortisM. pellceus, M. musculus), Mustelidae (M. itatsi), Procyonidae (P. lotor) and Suidae (S. scrofa) in Korea, China, Japan, Vietnam and the Russian Far East [12,14–25,29]. However, only one species of mustelid, the Japanese weasel M. itatsi, has been reported as a host of I. hortensis [13].
As with other Isthmiophora species that have 27 collar spines, I. hortensis was also found to use mustelids as its definitive hosts. In the present study, we found I. hortensis from 3 species of mustelids, i. e., M. sibirica, M. lucurus and M. flavigula. Furthermore, their infection rates seem relatively higher than those for other host species reported previously in Korea. Seo et al. [14] examined a total of 624 rodents comprising 5 species (A. agrarius, R. norvegicus, R. rattus, M. musculus and M. fortis) and a species of shrew, Crocidura lasiura Dobson (=C. russula) from the northern region of the Republic of Korea. They found I. hortensis from 15 rodents and the maximum number of infections was 134 from R. rattus. An additional survey was conducted inside a campus located in Seoul by Seo et al. [15]. They found only 4 positive rats among 325 rats (R. norvegicus). Follow-up studies of rodent species were then conducted by several authors, but the infection rates were not very high and measured 16/170 (rats) by Seo et al. [16], 1/34 (R. rattus) by Ahn and Ryang [42], and 6/236 (A. agrarius) by Sohn et al. [23]. The number of infections from the 3 studies was estimated as 1 to 65. Cho et al. [17] found I. hortensis from another host (dog) with an infection rate of 3.9% (4/102). Analysis of cats showed similar results.
Two surveys were conducted for helminth parasites of stray cats in Korea. I. hortensis was found in 5 of 438 (1.1%) cats in Busan, and 6 of 400 (1.5%) cats near 5 major river basins. The numbers of infection were 1–485 and 1–59, respectively [21,25]. Although the number of animals examined in the present study was not large, the infection rate and number of infections in mustelid hosts were relatively high (Table 1). On the other hand, our investigation of the infection status of the raccoon dog revealed results similar to those of a previously reported case of a dog (2.8 vs. 3.9%, respectively) and a smaller infection number (1–5 vs. 2–32). These results indicate that mustelid hosts act as predominant hosts of I. hortensis in wild carnivores within Korea.
The existence of additional species of Isthmiophora in Korea may prompt a revision of previously believed information on I. hortensis. The classical identifications of echinostome metacercariae depend only on the number of collar spines [9]. This resulted in little impetus previously to consider distinguishing 2 Isthmiophora species that had the same number of collar spines (27–28) using the classical morphological identification. If fishes can mediate the metacercariae of I. inermis, then previously reported data may include the misidentification of I. inermis. Indeed, a consideration of the habitat and eating habits of L. lutra suggests strongly that freshwater fishes are the main intermediate host of I. inermis rather than other intermediate hosts. Lutra lutra has been known to use freshwater fishes as its main prey. Furthermore, Choe et al. [43,44] recorded cases of fluke species in Korea, Pegosomum bubulcum [43] and Chaunocephalus ferox [44], which had 27 collar spines and were thought to use freshwater fishes as intermediate hosts. Recently, Sohn and Na [45] found unidentified echinostomatid metacercariae that are morphologically very similar with those of I. hortensis metacercariae from freshwater fishes caught from Junam-jeosuji (reservoir) and Woopo-neup (swamp) in Gyeongsangnam-do, Korea. In this situation, clarification of the intermediate host of I. inermis is considered essential before metacercariae investigation. In addition, certainty is needed regarding differential diagnosis between metacercariae that have 27 collar spines, and such investigations should involve molecular analysis. Human infections of I. inermis are also an issue that can’t be overlooked, and their potential for human infection needs to be investigated.
In conclusion, the results of the present study revealed the infection status of I. hortensis in various wild mammals and the existence of an additional Isthmiophora species, I. inermis, in Eurasian otters, in Korea. We provided their detailed morphometric comparisons, and showed the morphological differences between two species. However, our study is limited that the identification is based only morphological observations. DNA analysis should be conducted in near future for better understanding about the differences between Isthmiophora species and also the taxonomic aspects. As with other Isthmiophora species that have 27 collar spines, Isthmiophora hortensis appeared to have a significant relationship with mustelid hosts, and we added three more species to the host list of Isthmiophora hortensis. We also showed the relatively high prevalence of I. hortensis infection in mustelid hosts. Considering the present results and ecology of mustelid hosts, they seem to play an important role as dominant hosts in the life cycle of I. hortensis in the Korean ecosystem.
ACKNOWLEDGMENTS
The authors are grateful to Dr. Youngsun Lee (Gyeonggi Northern Livestock and Veterinary Service, Yangju, Korea) and Mr. Joon Seok Lee (Chungnam Wild Animal Rescue Center, Yesan, Korea), for their assistance with sample collections. This work was supported by a grant from the National Institute of Biological Resources (NIBR), which is funded by the Ministry of Environment (MOE) of the Republic of Korea (NIBR no. 2015-02-001) and the Parasite Resource Bank of Korea of the National Research Resource Center (2012-0000037) of the Republic of Korea.
Notes
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest with this article.


