Anisakid Larvae from Anchovies in the South Coast of Korea
Article information
Abstract
Anisakiasis (anisakidosis) refers to a foodborne zoonosis caused by ingesting raw or undercooked marine fish or cephalopods infected with anisakid larvae. The present study was performed to investigate the prevalence of anisakid larvae in anchovies (Engraulis japonica) purchased from 2 local markets in Gyeongsangnam-do, the Republic of Korea (=Korea), during 2018–2019. Anchovies were transported to our laboratory and examined by pepsin-HCl artificial digestion technique followed by microscopic observations and molecular analyses. The overall prevalence of anisakid larvae was 19.5% (39/200), from which a total of 51 larvae (av. 1.3 larvae/infected anchovy) were recovered. Sequencing of the larvae targeting the ITS region, including ITS1, 5.8S rRNA, and ITS2 genes confirmed the species of larvae as Anisakis pegreffii (54.9%; 28/51), Hysterothylacium sinense (23.5%; 12/51), and Hysterothylacium aduncum (21.5%; 11/51). The results suggested that anchovies could be a potential source of human anisakiasis in Korea.
Anisakiasis refers to a foodborne zoonosis caused by infection of people with larval stages of anisakid nematodes belonging to the families Anisakidae or Raphidascarididae, including the genera Anisakis, Pseudoterranova, and Contracaecum [1]. The disease may occur when people ingest raw marine fish or cephalopods, which contain third-stage larvae in their viscera or muscle. The first reported anisakiasis was 11 cases of previously healthy adults who experienced violent abdominal colic after ingestion of marine fish in the Netherlands [2]; larval anisakid nematodes were found deeply buried in their gut wall. After then, anisakiasis has been reported from almost all over the world. Over 90% of the total anisakiasis cases (about 20,000) were reported from Japan, followed by the Netherlands, Germany, France, Spain, and the Republic of Korea (=Korea) [1]. The clinical symptoms of invasive anisakiasis include severe abdominal pain, nausea, and vomiting, and the larvae can be removed at gastrointestinal endoscopy or by surgery [1]. In some cases of invasive anisakiasis, the larvae rarely penetrate other sites such as the throat or visceral organs [1,3]. To prevent this infection, marine fish needs to be immediately eviscerated after fishery and be cooked at temperatures greater than 60°C or frozen at −20°C for at least 24 hr prior to consumption [1].
People in Korea like to consume raw or lightly cooked or salted fish. Consequently, since Kim et al. [3] recorded the first case of human anisakiasis in Korea, a total of 645 anisakiasis cases have been reported until 2015 [4]. Among 203 clinical cases, the majority (165 cases; 81.3%) was due to Anisakis type I larvae [4]. Based on morphological analyses, these Anisakis type I larvae were diagnosed as Anisakis simplex [5–7]. However, 25 out of 26 larvae (96.2%) extracted from human patients during 2000–2013 in Korea were confirmed to be Anisakis pegreffii using molecular analyses [8]. More recently, all 20 larvae (100%) extracted from patients by endoscopy in a health check-up center in 2017–2019 were also molecularly confirmed to be A. pegreffii [9]. The larvae of A. simplex and A. pegreffii are morphologically indistinguishable (Anisakis type I), and therefore molecular analyses were required to determine the precise species [10].
Several studies have been performed on the prevalence of anisakid larvae in various marine fishes in Korea [11–15]. However, the infection status of anchovies (Engraulis japonica) has seldom been reported except in a study by Song et al. [11], which used the morphological method for species identification. In the present study, we investigated the infection status of anchovies purchased from 2 markets in the south coast of Korea with anisakid larvae using both morphological and molecular methods. The internal transcribed spacer (ITS) region, including the ITS1, 5.8S rRNA, and ITS2 genes, were sequenced for molecular studies.
A total of 200 anchovies (100 from each area) were purchased from 2 local fish markets located in a middle (area A) and a western part (area B) of Gyeongsangnam-do, Korea (50 km apart from each other). The area A was surveyed in October 2018 and the area B was studied in May 2019. The anchovies were stated to have been caught near the coast of the South Sea, Korea. In total, 51 anisakid larvae were collected from 200 anchovies, and all the larvae collected were molecularly analyzed.
To isolate the larvae from anchovies, the pepsin-HCl artificial digestion method was used. The procedure was as it follows. Identification of the fish species and measurement of the size and weight of each fish. Grinding the fish in a mortar with pestle. Transferring the flesh of the fish into a beaker and pour artificial gastric juice (8 ml HCl + 6 g pepsin 1: 10,000 + 1,000 ml of 0.85% saline). Mixing the mixture well and keeping it at 37°C for 2 hr with occasional stirring. Filtering it through a 1×1 mm mesh. Adding 0.85% saline and letting it stand until sediment came down. Discarding the supernatant carefully not to throw away the sediments. Repeating the washes until the supernatant became clear. Transferring the sediment into a Petri dish and examination under a stereomicroscope.
Anisakid larvae collected from the sediment were washed in water, cleared in lactophenol solution, and observed under an optical microscope (Leica D2500, Leica Microsystems, Wetzlar, Germany). Morphologically, third-stage larvae of Anisakis type I and 2 Hysterothylacium spp. (H. sinense and H. aduncum) were identified.
In order to obtain a molecular (species-specific) diagnosis, the total genomic DNA (gDNA) was extracted using the DNeasy Blood and Tissue kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Molecular identification was carried out by sequencing of the ITS (ITS1, 5.8 subunit rRNA, and ITS2) genes. Amplification of the nuclear ribosomal region was performed using the ITS A (5′-GTC GAA TTC GTA GGT GAA CCT GCG GAA GGA TCA-3′) and ITS B (5′-GCC GGA TCC GAA TCC TGG TTA GTT TCT TTT CCT-3′) [16] with 2× MasterMix (MGmed, Seoul, Korea), containing 10 pmol of each primer. The mixture was undergone initial denaturation at 95°C for 10 min, followed by 35 cycles of denaturation at 95°C for 30 sec, annealing at 55°C for 30 sec, and extension at 72°C for 80 sec with a final extension at 72°C for 5 min. Automated DNA sequencing experiments on ITS PCR products were performed at Macrogen Co. (Seoul, Korea).
Anisakid larvae were detected in 39 (19.5%) out of 200 anchovies examined (Fig. 1A, B), and a total of 51 larvae were collected (av. 1.3 larvae/infected anchovy) (Table 1). The larvae were divided into 3 different morphological types. Larvae of Anisakis type I (A. pegreffii) (Group 1) had a boring tooth at its anterior end, a long ventriculus, and a mucron at its caudal end (Fig. 2A–C). Larvae of Hysterothylacium sinense (Group 2) had a large boring tooth at its anterior end, a small ventriculus with a long intestinal cecum and a long ventricular appendage, but not a structure like a mucron at its caudal end (Fig. 2D–F). Larvae of Hysterothylacium aduncum (Group 3) had a small boring tooth at its anterior end, a small ventriculus with a long intestinal cecum, and a long ventricular appendage, and a mucron at its posterior end (Fig. 2G–I).

Anchovy purchased from a local market in Gyeongsangnam-do. (A) Whole fish. (B) Sashimi of the anchovy. Scale bar=1 cm.

Microscopic images of third-stage larvae of Anisakis pegreffii (A–C), Hysterothylacium sinense (D–F), and Hysterothylacium aduncum (G–I). Scale bar=0.1 mm. (A) Anterior extremity of A. pegreffii larva showing a boring tooth (BT). (B) Middle part showing a long ventriculus (V). (C) Posterior extremity with a mucron (M). (D) Anterior end of Hysterothylacium sinense larva with a boring tooth (BT). (E) Middle part showing a small ventriculus (V). (F) Posterior extremity showing its caudal end without a mucron. (G) Anterior extremity of Hysterothylacium aduncum larva with a small boring tooth (BT). (H) Middle part showing a long intestinal cecum (IC; arrow indicates its terminal portion). (I) Posterior extremity showing its caudal end with a mucron (M).
The species-specific identification of the anisakid larvae was supported by sequencing of the ITS region (Fig. 3). Group 1 larvae (Anisakis type I; 28/51, 54.9%) showed 100% identity with A. pegreffii (AB196671) and 99.5% with A. simplex (AB277822). Group 2 larvae (12/51, 23.5%) showed 100% identity with H. sinense (KX817295), 79.9% with H. aduncum (KX110074), and 60.3% with A. pegreffii (AB196671). Group 3 larvae (11/51, 21.5%) showed 100% identity with H. aduncum (KX110074), 79.9% with H. sinense (KX817295), and 63.4% with A. pegreffii (AB196671). The prevalence of anisakid larvae in anchovies purchased from 2 surveyed areas was a little different from each other; it was higher in area A (25/100; 25.0%) than in area B (14/100; 14.0%) (Table 1).
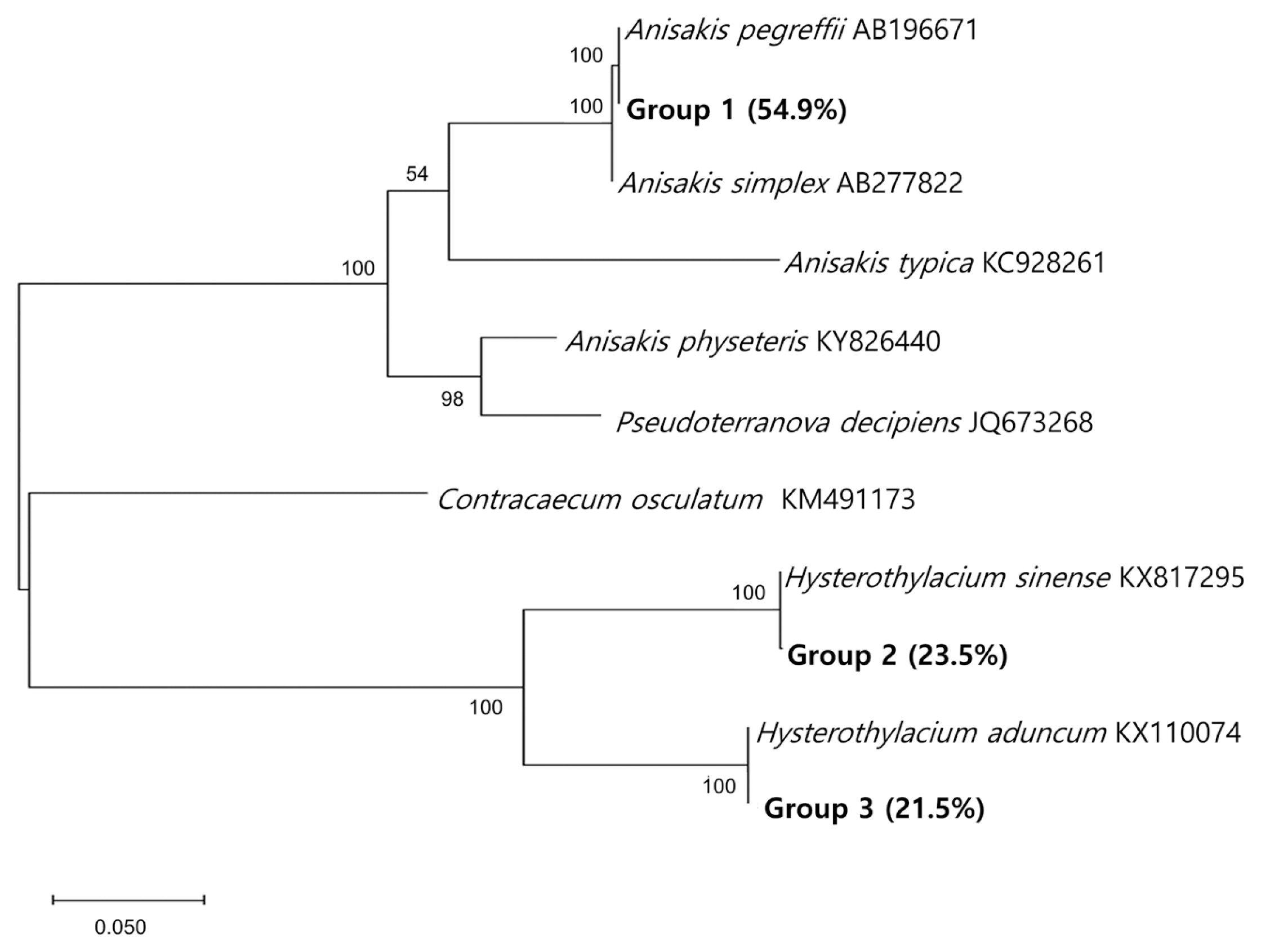
A phylogenetic tree of anisakid nematodes based on sequences of the ITS region. This tree was constructed from a representative selection of sequences available in GenBank using the maximum-likelihood method employing Tamura-nei model of nucleotide substitution with 1,000 bootstrap replications. Scale bars indicate nucleotide substitutions per site.
The economic condition of Korea has developed remarkably over several decades, and the trends of parasite diseases have changed noticeably. Nowadays, foodborne parasites, including liver and intestinal flukes and anisakid larvae, have become the major groups of epidemiological significance, whereas, in the past, soil-transmitted helminthiases were ranked as the most prevalent parasitic diseases [1].
A. pegreffii was originally reported from a Mediterranean monk seal in 1955 by Campana-Rouget and Biocca [17]. The third-stage larvae of A. pegreffii are found in various species of marine fish and cephalopods and morphologically classified as Anisakis type I, the same type as A. simplex. Since the larvae of A. pegreffii and A. simplex are almost indistinguishable each other, molecular methods, including PCR-RFLP and nucleotide sequencing, were applied for species identification [18–23]. The main geographic habitat of A. pegreffii was reported to be the Mediterranean Sea and the Southern Atlantic Ocean [24]. The first case of human anisakiasis caused by A. pegreffii was reported in Italy [18]. Later, studies using molecular methods have found that this species also occurs frequently in Korea and Japan [15]. Subsequently, human infections have been found in Japan, Italy, and Korea [8,9,19–22]. In Korea, a total of 35 A. pegreffii infections have been molecularly diagnosed, including 15 cases in 2015 [8] and 20 cases in 2019 [9].
In our study, 3 species of anisakid larvae were isolated from anchovies caught from the south coast of Korea. Among 51 larvae isolated, 54.9% (28/51) were molecularly confirmed to be A. pegreffii. According to Sohn et al. [4], 1.3% of anisakiasis patients (4/311) reported in Korea recalled that they had eaten anchovies. Therefore, anchovies are suspected as one of the potential sources of human anisakiasis in Korea.
The genus Hysterothylacium Ward and Magath, 1917 (Anisakidae) comprises of more than 60 species [25]. The larval stages of these worms mostly parasitize the viscera and tissues of various fish and invertebrate hosts which act as paratenic or intermediate hosts [26,27]. The adult worms of Hysterothylacium sinense Li, An and Zhang, 2007 were discovered from the intestine of marine fishes caught off the Yellow Sea, China [25], and their third-stage larvae were found from the cinnamon flounder (Pseudorhombus cinnamoneus) caught from the Yellow Sea, China [28]. The presence of adult Hysterothylacium aduncum (Rudolphi, 1802) Deardorff and Overstreet, 1981 was reported mainly in temperate and cold waters, including the North-Eastern Atlantic, Seas of the North of Europe, the Mediterranean Sea, Adriatic Sea, Pacific and Atlantic waters of North America, waters of Australia, and the southwest Atlantic Ocean [29]. Various species of fish caught from the south coast of Korea, including the sea eels (Astroconger myriaster) and chub mackerel (Scomber japonicus), were reported to be infected with H. aduncum larvae [9,12–14]. Our study first demonstrated the presence of H. aduncum and H. sinense larvae in anchovies using molecular analysis.
Among the species of Hyterothylacium, human infections have been known only in H. aduncum; 2 cases from Japan and Spain [30,31]. In these reports, the specific identification of the larvae was performed only by morphological methods [30,31]. Thus, further (molecular) studies are required to determine whether H. aduncum can truly infect humans. In addition, the possibility of human infection with H. sinense also needs to be elucidated.
The prevalence of anisakid larvae in anchovies purchased from 2 different areas (middle and western parts of Gyeongsangnam-do) was different by locality; 25.0% in middle (area A) vs 14.0% in western parts (area B) (Table 1). Based on the fact that both areas belong to the South Sea and the distance between the areas is not so far (approximately 50 km), it seems that the locality where the anchovies were caught did not affect much difference in the prevalence. Rather, season (October in area A and May in area B) of fishery might have been a more important factor for the difference. Previous studies on various fish species have described seasonal variations in the prevalence of anisakid larvae, including A. simplex, Contracaecum sp., and H. aduncum [32]. Further studies are needed to confirm seasonal tendencies in the prevalence of anisakid larvae in anchovies caught in the South Sea.
ACKNOWLEDGMENT
This study was supported by a grant from the Ministry of Food and Drug Safety, the Republic of Korea, in 2017–2019 (17162MFDS034).
Notes
CONFLICT OF INTEREST
We have no conflict of interest related to this work.
