INTRODUCTION
Freshwater snails are known to host a wide variety of parasites. Many digenetic trematode species, especially the Echinostomatidae, require 1 or 2 snail hosts to complete their life cycles. Over 60 species of echinostomatid flukes are distributed worldwide [1]. Most species mainly parasitize avian hosts such as poultries and migratory birds, and sometimes in mammals including humans. Among them, at least 20 species belonging to 9 genera have been reported to cause human infections throughout the world and also constitute public health problems especially in Southeast Asia and the Far East [2,3]. The zoonotic potential of echinostomes is associated with the ingestion of raw snails, fishes, and amphibians that harbor the infective stage, metacercariae [4].
Echinostomiasis cases have been commonly found in north and northeastern parts of Thailand [5-7]. More than 50% of the stool samples collected in northern Thailand residents contained echinostome eggs [8]. Eating raw snails and tadpoles was identified as an important mode of transmission [9]. Moreover, various species of freshwater snails have been reported to play a dual role of the first and second intermediate hosts of echinostomes in Thailand, namely, Indoplanorbis spp., Gyraulus spp., Lymnaea spp., Pila spp., Viviparus spp., Filopaludina spp. and Bithynia spp. [10,11].
During the examination of larval stages of trematodes, freshwater snails were found to be infected with a larval stage of an echinostome bearing 37 collar spines that resembles members of Echinostoma revolutum group. There have been reports of echinostome infections in the intestine of various definitive hosts such as birds and mammals, including humans in Thailand [6,7,12,13], but little has been reported on intermediate hosts. Therefore, the present study was carried out for species identification and determination of the prevalence and intensity of echinostome metacercariae in freshwater snails collected from Chiang Mai province, Thailand, and also to provide epidemiological information on larval trematodes of freshwater snails that will help developing a prevention strategy for public health promotion.
MATERIALS AND METHODS
Study area
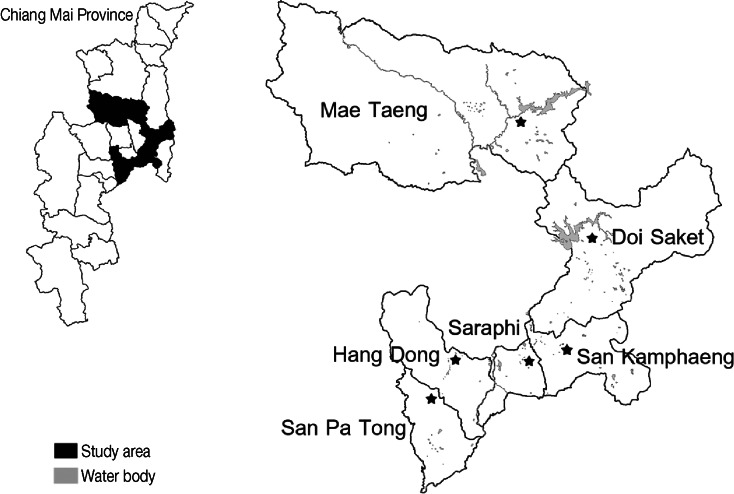
Snails were collected in Chiang Mai province, Thailand, during October 2011 to April 2012. The study areas occupied over 6 districts of Chiang Mai province; Doi Saket, Hang Dong, Mae Taeng, San Kamphaeng, San Pa Tong, and Saraphi (Fig. 1). Freshwater snails were collected in both stagnant and running water bodies such as the pond, rice paddy, irrigation canal, stream, and river. The number of investigated snails, the habitat type, and coordinates of particular localities were recorded and detailed in Table 1.
Snail sampling and identification
Snail samples were collected using a simple random sampling approach. The snails were found on the rock, rough sand, dried leaves, and on aquatic plants in all habitat types. They were collected from diverse habitats by handpicking and scooping methods. At least 2 different habitats were investigated in each of the districts. Live snails from each area were placed in separate containers with water from the same habitat and transported to the laboratory. Live snails were kept in the laboratory in aquaria containing dechlorinated tap water and aerated at room temperature until they were examined. Snail species were identified according to the morphological features described by Brandt [14].
Snail samples collected
A total of 2,914 freshwater snail specimens were collected from different sampling sites in 6 districts of Chiang Mai province. On the basis of the shell morphology, the snail samples were classified into 12 species. They included 198 Clea helena, 16 Eyriesia eyriesi, 220 Lymnaea auricularia rubiginosa, 16 Adamietta housei, 144 Thiara scabra, 462 Melanoides tuberculata, 300 Tarebia granifera, 100 Bithynia funiculata, and 244 Bithynia siamensis siamensis. Of the Filopaludina spp., 542 belonged to Filopaludia doliaris, 122 were Filopaludia sumatrensis polygramma, and 550 were Filopaludina martensi martensi.
Collection of the metacarcariae
Larval trematode infections in snails were studied by the crushing method. They were isolated using a sharp pin, gently covered with a cover slip, and observed under a light microscope. The presence of a head collar with 37 spines was highly indicative of E. revolutum group. The numbers of metacercariae were recorded. Freshly isolated metacercariae were used for morphological observations and experimental infection to various definitive hosts.
Identification of metacercariae
Freshly isolated 37-collar spined metacercariae were fed to hamsters (Mesocricetus auratus) and domestic chicks (Gallus gallus domesticus). Infected animals were sacrificed and necropsied at days 15 and 20 post-infection, and adult flukes were collected from their intestines. The collected worms were compressed, fixed in 5% formalin, stained with aceto-carmine, dehydrated in ascending grades of alcohols, cleared in xylene, and mounted in Permount for permanent slides.
Freshly isolated metacercariae and permanent slides of adult worms were examined under a light microscope. The shape, size, and cyst wall structure were used for characterization of the metacercariae. For adult worms, the size and shape of the body and organs, number and arrangement of collar spines, and the shape and size of eggs were employed as the morphological parameters.
RESULTS
Morphology of the metacercariae and adults
Metacercaria
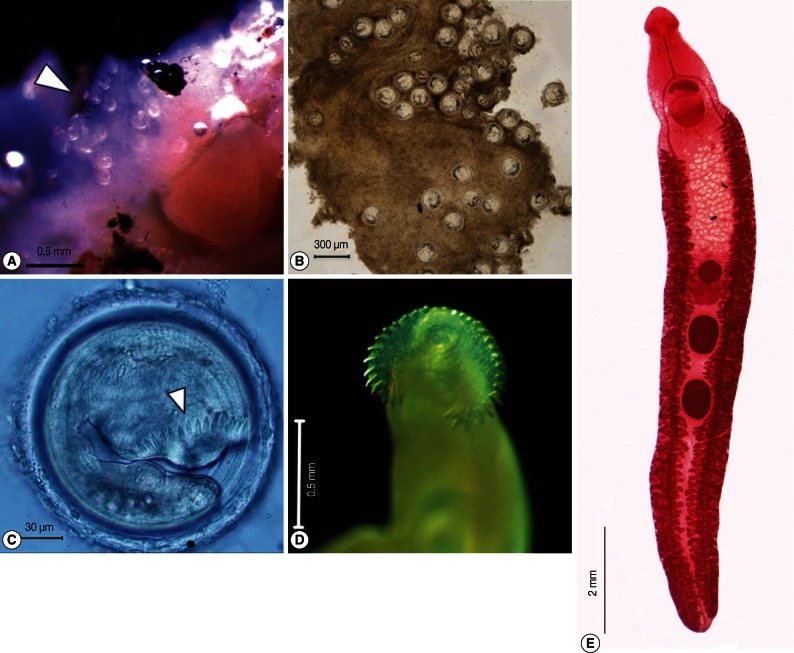
Metacercariae, folded within a transparent cyst (Fig. 2C), were spherical, 136.0-195.0 µm in diameter, with a bilayered wall (n=30). Cyst wall consisted of outer, transparent layer, about 4.4-10.5 µm thick, and inner, opaque layer, about 0.7-2.7 µm thick. Collar spines, 37 in total number, were presented in both fresh and fixed specimens. Excretory granules and suckers were visible. Excretory bladder was filled with small concretions. Cysts were found clumped together in the pericardial sac of the snails, with each cyst enveloped by thin connective tissues of the snail host origin (Fig. 2A, B).
Adult
Most adult worms were recovered from the duodenum of hamsters, and from the rectum and colon of chicks. Adults at day 20 post-infection (n=20) were elongated, 3.40-5.30 mm (4.35 mm in average) cm in length, dorso-ventrally flattened, somewhat attenuated at both ends, and ventrally slightly bent (Fig. 2E). Tegument of anterior body armed with numerous small spines which extended to the posterior end of ventral sucker. Head crown distinct, well developed, with ventral ridge (Fig. 2D). There are 37 collar spines arranged in a semicircle around the collar, double row, and dorsally uninterrupted, with spine arrangement of 5 corner spines on each side, 6 laterals on each side in a single row; 15 dorsal spines in a double row. Oral sucker ventro-subterminal, spherical or subspherical, and prepharynx absent or very shot. Pharynx well developed, muscular, elongate-oval, esophagus long. Intestinal bifurcation anterior to ventral sucker; intestinal ceca blind and extending almost to posterior extremity. Ventral sucker spherical, muscular, very large, located middle of body and protruded ventrally. Testes 2, tandem, beginning at mid-hind body, elongate-oval, slightly lobed or slightly irregular in outline. Cirrus sac muscular, well developed, elongate-oval, antero-dorsal to ventral sucker, median and immediately preacetabular. Cirrus unarmed and unclear in length. Genital pore median, preacetabular, followed by genital atrium. Ovary spherical or nearly oval, compact, at or anterior to midbody. Mehlis' gland located between the ovary and anterior testis. Uterus moderately developed, with intercecal loops between ovary and ventral sucker. Eggs numerous, 72.5-120×40-82.5 µm (n=20) in size. Eggs operculated, elliptical, yellowish, and thin-shelled. Vitellaria follicular, forming 2 lateral fields and extending from short distance posterior to ventral sucker to the posterior end of body. Excretory vesicles not observed, excretory pore terminal.
Infection status of snails with E. revolutum metacercariae
Total 656 of 2,914 snails examined were infected with E. revolutum metacercariae. The overall infection rate and the number of metacercariae recovered from snail species are shown in Table 2. Throughout the course of our survey, a total of 7,486 metacercariae were recovered from 7 snail species, including C. helena, E. eyriesi, B. funiculata, B. siamensis siamensis, F. doliaris, F. sumatrensis polygramma, and F. martensi martensi. The remaining 5 species, i.e., L. auricularia rubiginosa, A. housei, T. scabra, M. tuberculata, and T. granifera were found not to be infected with these metacercariae.
The overall prevalence of infection was 22.5% (656/2,914). The infection rate varied from 0% to 58.7%. Most metacercariae were recovered from Filopaludina spp., and the least from C. helena (5.1%, 10/198). A total of 7,486 E. revolutum metacercariae were found in the pericardial sac of infected snails (Fig. 2A, B), with a mean intensity of 11.4 per snail. The intensity of infection ranged from 1.2 to 16 metacercariae per snail. The most heavily infected species of snails were F. doliaris (58.7%), B. funiculata (44%), F. sumatrensis polygramma (41%), and F. martensi martensi (38.5%).
On the other hand, in some snail species, including C. helena, E. eyriesi, B. funiculata, B. siamensis siamensis, F. doliaris, F. sumatrensis polygramma, and F. martensi martensi, there were mixed infections with several types of cercariae. They included monostome cercariae, distome cercariae, xiphidiocercariae, gymnocephalus cercariae, and furcocercous cercariae. There were found as mixed infections in the gastrointestinal organs of the infected snails. Moreover, mixed infections with unidentified metacercariae were found in B. siamensis siamensis.
DISCUSSION
Several studies on the infection of digenean larvae in their snail hosts have been reported from Thailand [15-17]. The studies revealed many described and undescribed digenean species. The present study was carried out to observe the natural infection of the metacercariae of E. revolutum from freshwater snails in Chiang Mai province, Thailand. We detected metacercariae of E. revolutum in 7 snail species. The result of this study indicated that these snails act as the second intermediate hosts of this parasite. In addition, in C. helena, E. revolutum metacercariae were found for the first time in Thailand. For the experimental infection, metacercariae were force-fed to hamsters and domestic chicks, and adult flukes were successfully recovered from their small intestines. The worms characteristically revealed a prominent head collar with 37 collar spines arranged in a double row. In this study, they were identified as E. revolutum (Fröelich, 1802) Looss, 1899 based on the morphology, morphometrics, host-parasite relationships, and geographic distribution.
However, some difficulty was reported in identifying the species, because many different species with similar morphologies were reported among the 37-collar spined echinostomes, the so-called 'revolutum' group, and taxonomic problems exist among those species. Ten species of Echinostoma were listed as valid within the revolutum group by Fried and Graczyk [18]; E. caproni Richard, 1964, E. trivolvis (Cort, 1914), E. paraensei Lie and Basch, 1967, E. revolutum (Frölich, 1802), E. friedi Toledo et al., 2000, E. miyagawai Ishii, 1932, E. echinatum (Zeder, 1803), E. parvocirrus Nassy and Dupouy, 1988, E. luisreyi Maldonado et al., 2003, and E. jurini (Skvortzov, 1924). Review of the Echinostoma species in the revolutum group in terms of morphological features, morphometrics, host-parasite relationships, and geographic distribution [3,18-28] revealed that our specimens resembled most closely E. revolutum and E. jurini. Of these, our specimens were more compatible to E. revolutum.
E. revolutum differs from E. jurini by its definitive hosts. E. jurini have only mammals as the definitive hosts, whereas E. revolutum take both birds and mammals as the definitive host [18,22]. Also in the present study, E. revolutum could be infected to both hamsters and domestic chicks. Moreover, Kanev et al. [22] presented that E. revolutum takes lymnaeid snails as the first intermediate host, and molluscs, tadpoles, fish, and freshwater turtles as the second intermediate host. E. jurini have viviparid snails as the first intermediate host, and molluscs, frogs, and freshwater turtles as the second intermediate host. E. jurini has been reported from Europe and possibly in Asia. It is suggested that E. jurini occur in Asia where viviparid snail hosts are distributed. In our study, hamsters and domestic chicks were both successfully infected, and the second intermediate hosts were Filopaludina spp., E. eryresi (viviparid snails), Bithynia spp., and C. helena. The first intermediate host for our flukes may be the same or different species of these snails, although this was not confirmed in this study. These results were highly compatible with E. revolutum rather than E. jurini.
There were many reports of human infections with the Echinostomatidae in North and Northeast of Thailand [7]. Six species were included; Echinostoma malayanum, Hypoderaeum conoideum [29], Echinostoma ilocanum, Echinochasmus japonicus, E. revolutum, and Episthmium caninum [30-32]. However, the intermediate hosts of these trematodes, including the snail, fish, and tadpole have been little studied. Several studies have been undertaken on the occurrence of digenean larvae in the snail hosts in northeast, central, and southern Thailand [15-17]. The studies reported many already described and undescribed digenean species which varied in their larval stages. However, few have been studied and reported in northern Thailand, especially in Chiang Mai province.
Our study is a progressive report of freshwater snail distribution and infection status with E. revolutum metacercariae in Chiang Mai province. A total of 2,914 snail samples comprising of 12 species were collected. They consisted of 5 families, including Viviparidae, Bithyniidae, Buccinidae, Thiaridae, and Lymnaeiidae. As shown by the results (Table 2), 4 viviparid snails (F. doliaris, F. sumatrensis polygramma, F. martensi martensi, and E. eyriesi), 2 bithyniid snails (B. funiculata and B. siamensis siamensis) and 1 buccinid snail (C. helena) were found to be infected with E. revolutum metacercariae. The highest prevalence was seen in viviparid snails, Filopaludina spp. (38.5-58.7%) and B. funiculata (44%). Meanwhile, 3 snail species showed lower prevalences, E. eryresi (12.5%), B. siamensis siamensis (8.2%), and C. helena (5.1%). On the other hand, thiarid snails (A. housei, T. scabra, M. tuberculata, and T. granifera) and lymnaeid snails (L. auricularia rubiginosa) were not found to be infected with E. revolutum metacercariae. The metacercariae were found chiefly in the pericardial sac of the snails. Esteban and Antoli [4] similarly reported that the Echinostomatidae metacercariae were found in the pericardial sac and kidney region of the snail host.
According to Brandt [14] who studied on non-marine aquatic mollusca of Thailand, many Filopaludina spp. were found to harbor several kinds of cercariae and metacercariae of the Echinostomatidae. However, the larvae were not identified to the genus and species level. For bithyniid snails, B. funiculata and B. siamensis siamensis are important intermediate hosts of Opisthorchis viverrini [14,33], while in this study they were found to harbor E. revolutum metacercariae. With regard to C. helena, they appeared to be infected with E. revolutum metacercariae in this study, although they had never been considered to be of medical importance previously [11,14,15,19,28,33]. Hence, detection of E. revolutum metacercariae in C. helena is for the first time so far as the literature are concerned. In addition, the present study also confirmed that Filopaludina spp., E. eyriesi, and Bithynia spp. act as the second intermediate hosts of E. revolutum under natural conditions, which is the first report in Chiang Mai province.
In the present study, E. revolutum metacercariae showed a broad range of host specificity because they could infect various species of freshwater snails. Viewing from the high prevalence and high intensity of metacercarial infections in the snail, it is possible that the surveyed area is one of the high risk areas. The finding of E. revolutum in Chiang Mai province also raises an interesting question about its distribution throughout the northern part of Thailand, its adaptation toward local snail species, and its ubiquity in this area. In addition, the results in this study provide information concerning the existence and biological characteristics of E. revolutum in Thailand. Further, it is hoped that the present study might be helpful to design public health prevention and control strategies for human echinostomiasis.
Conclusively, the present study confirmed for the first time that freshwater snails in Chiang Mai province are heavily infected with E. revolutum metacercariae and their life cycle is actively maintained in the study area. Moreover, this study confirmed C. helena as a new second intermediate host of E. revolutum. The discovery of this fluke in this area is important for public health control and for monitoring its dispersion because E. revolutum can be easily transmitted to humans if they consume raw or partially cooked snails.